Articles
- Page Path
- HOME > Endocrinol Metab > Volume 38(6); 2023 > Article
-
Original ArticleThyroid Phospholipase C-γ as a Potential Therapeutic Target for Graves’ Orbitopathy
 Keypoint
Keypoint
A study investigated the role of phospholipase C-gamma, or PLC-γ, role in Graves' orbitopathy, or GO, and the effects of the PLC-specific inhibitor U73122 on GO orbital fibroblasts. Elevated PLCG1 and PLCG2 mRNA expression levels were found in GO tissues. U73122 reduced the levels of proinflammatory molecules and phosphorylated kinases in GO fibroblasts, suggesting its therapeutic potential. This highlights PLC-γ's role in GO pathogenesis and U73122 as a promising treatment target. -
Tae Hoon Roh1
 , Min Kyung Chae2, Jae Sang Ko2, Don O. Kikkawa3, Sun Young Jang4
, Min Kyung Chae2, Jae Sang Ko2, Don O. Kikkawa3, Sun Young Jang4 , Jin Sook Yoon2
, Jin Sook Yoon2
-
Endocrinology and Metabolism 2023;38(6):739-749.
DOI: https://doi.org/10.3803/EnM.2023.1780
Published online: November 21, 2023
1Department of Medicine, Yonsei University College of Medicine, Seoul, Korea
2Department of Ophthalmology, Severance Hospital, Institute of Vision Research, Yonsei University College of Medicine, Seoul, Korea
3Division of Oculofacial Plastic and Reconstructive Surgery, Department of Ophthalmology, Shiley Eye Institute, University of California San Diego, La Jolla, CA, USA
4Department of Ophthalmology, Soonchunhyang University Bucheon Hospital, Soonchunhyang University College of Medicine, Bucheon, Korea
- Corresponding authors: Sun Young Jang. Department of Ophthalmology, Soonchunhyang University Bucheon Hospital, Soonchunhyang University College of Medicine, 170 Jomaru-ro, Wonmi-gu, Bucheon 14584, Korea Tel: +82-32-621-6718, Fax: +82-32-621-5018, E-mail: ysyat01@naver.com
- Jin Sook Yoon. Department of Ophthalmology, Severance Hospital, Institute of Vision Research, Yonsei University College of Medicine, 50-1 Yonsei-ro, Seodaemun-gu, Seoul 03722, Korea Tel: +82-2-2228-3570, Fax: +82-2-312-0541, E-mail: yoonjs@yuhs.ac
Copyright © 2023 Korean Endocrine Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 1,641 Views
- 97 Download
ABSTRACT
-
Background
- Phospholipase C-γ (PLC-γ) plays a crucial role in immune responses and is related to the pathogenesis of various inflammatory disorders. In this study, we investigated the role of PLC-γ and the therapeutic effect of the PLC-specific inhibitor U73122 using orbital fibroblasts from patients with Graves’ orbitopathy (GO).
-
Methods
- The expression of phospholipase C gamma 1 (PLCG1) and phospholipase C gamma 2 (PLCG2) was evaluated using polymerase chain reaction in GO and normal orbital tissues/fibroblasts. The primary cultures of orbital fibroblasts were treated with non-toxic concentrations of U73122 with or without interleukin (IL)-1β to determine its therapeutic efficacy. The proinflammatory cytokine levels and activation of downstream signaling molecules were determined using Western blotting.
-
Results
- PLCG1 and PLCG2 mRNA expression was significantly higher in GO orbital tissues than in controls (P<0.05). PLCG1 and PLCG2 mRNA expression was significantly increased (P<0.05) in IL-1β, tumor necrosis factor-α, and a cluster of differentiation 40 ligand-stimulated GO fibroblasts. U73122 significantly inhibited the IL-1β-induced expression of proinflammatory molecules, including IL-6, IL-8, monocyte chemoattractant protein-1, cyclooxygenase-2, and intercellular adhesion molecule-1 (ICAM-1), and phosphorylated protein kinase B (p-Akt) and p38 (p-p38) kinase in GO fibroblasts, whereas it inhibited IL-6, IL-8, and ICAM-1, and p-Akt and c-Jun N-terminal kinase (p-JNK) in normal fibroblasts (P<0.05).
-
Conclusion
- PLC-γ-inhibiting U73122 suppressed the production of proinflammatory cytokines and the phosphorylation of Akt and p38 kinase in GO fibroblasts. This study indicates the implications of PLC-γ in GO pathogenesis and its potential as a therapeutic target for GO.
- Graves’ orbitopathy (GO), also known as thyroid eye disease and thyroid-associated ophthalmopathy, is an autoimmune inflammatory disorder of the eye orbit associated with Graves’ disease [1]. Although GO pathophysiology is yet to be fully elucidated, the role of thyrotropin receptor (TSHR)- and insulinlike growth factor-1 receptor (IGF-1R)-autoantibody-stimulated orbital fibroblasts in the disease pathogenesis is well-known [2-4]. Current evidence indicates that the inflammatory response is perpetuated and amplified via complex interactions among orbital fibroblasts and infiltrated inflammatory cells [4,5]. During the process, activated orbital fibroblasts can differentiate into adipocytes and produce excessive hyaluronan, a hydrophilic glycosaminoglycan [1,6]. Consequent enlargement of extraocular muscles and an increase in orbital connective tissues can cause various clinical complications, including proptosis, periorbital edema, diplopia, and compressive optic neuropathy [4,6]. To decrease the orbital inflammatory response, which is a major etiology of GO, glucocorticoids have been used as a mainstay treatment [7]; however, concerns regarding systemic side effects and treatment failures indicate the need for developing new effective therapeutic agents [7].
- Phospholipase C (PLC) is an enzyme that catalyzes phosphatidylinositol 4,5-bisphosphate hydrolysis into diacylglycerol and inositol triphosphate, which mediates the activation of protein kinase C (PKC) and Ca2+ signaling [8]. Mammalian PLC enzymes are categorized into six subtypes (β, γ, δ, ε, ζ, and η) based on their structures [8]. PLC-γ and PLC-β are recognized as primary PLCs that are directly activated by extracellular stimuli, and their strong association with various human diseases has been widely reported [9]. PLC-γ has two isozymes, PLC-γ1 and PLC-γ2, which differ from other PLC subtypes because of their unique γ-specific array (γSA) structure [10]. γSA plays a crucial role in conferring specific functions, such as regulating PLC-γ activity and facilitating interactions with various signaling molecules and receptor tyrosine kinase [10]. PLC-γ1 is widely expressed and regulates cellular growth and differentiation, whereas PLC-γ2 is expressed mainly in immune cells [8]. PLC-γ is a key regulator of mediating immune responses and engages in various signaling pathways, including those associated with T-cell receptors, B-cell receptors, and toll-like receptors (TLRs) [11-13]. Recent studies have revealed that aberrant PLC-γ activation and gene expression induce inflammatory conditions such as autoinflammation and PLC-γ2-associated antibody deficiency and immune dysregulation [14], rheumatoid arthritis [15], viral inflammation [16], and different cancer types [17]. Moreover, animal experiments revealed that gain-of-function mutations in phospholipase C, gamma 2 (Plcg2) could induce severe inflammatory reactions and autoimmunity [18,19] and the protective and anti-inflammatory effects of PLC-γ2 deficiency against inflammatory arthritis [20,21].
- In the context of its potential role in various inflammatory and immune responses, prospects of PLC-γ as a therapeutic target are currently being explored, and the anti-inflammatory effect of PLC inhibition has been mainly reported in cell experiments [16,22,23]. However, no studies have been conducted to investigate the relationship between PLC-γ and GO. Therefore, in this study, we investigated the role of PLC-γ in the pathogenesis of GO using the PLC-specific inhibitor U73122.
INTRODUCTION
- Reagents
- The reagents used in the study were obtained from the following sources: U73122 (catalog number 1268, Tocris Bioscience, Minneapolis, MN, USA); Dulbecco’s modified Eagle’s medium/Nutrient Mixture F-12 (DMEM/F12, 1:1), penicillin, and streptomycin (Welgene, Gyeongsan, Korea); fetal bovine serum (FBS) (Gibco, Thermo Fisher Scientific, Waltham, MA, USA); 3-(4,5-dimethyl-thiazol-2-yl)-2,5-diphenyl-tetrazolium bromide (MTT) (catalog number 475989, Sigma-Aldrich, St. Louis, MO, USA); recombinant human interleukin (IL)-1β (catalog number 201-LB) and tumor necrosis factor (TNF)-α (catalog number 210-TA, R&D systems, Minneapolis, MN, USA); recombinant human cluster of differentiation 40 ligand (CD40L) (catalog number ALX-850-064-KI01, Enzo Life Sciences, Farmingdale, NY, USA); antibodies for phosphorylated protein kinase B (p-Akt) (catalog number 9271), total Akt (t-Akt) (catalog number 9272), phosphorylated extracellular signal-regulated kinase (p-ERK) (catalog number 9101), t-ERK (catalog number 9102), p-p38 mitogen-activated protein kinase (MAPK) (catalog number 9211), t-p38 (catalog number 9212), phosphorylated c-Jun N-terminal kinase (p-JNK) (catalog number 9251), t-JNK (catalog number 9252), IL-6 (catalog number 12153), IL-8 (catalog number 94407), monocyte chemoattractant protein-1 (MCP-1, also known as C–C motif chemokine ligand 2 [CCL2]) (catalog number 2027), cyclooxygenase-2 (COX-2) (catalog number 4842), and intercellular adhesion molecule-1 (ICAM-1) (catalog number 4915, Cell Signaling Technology, Danvers, MA, USA); and anti-β-actin antibody (catalog number sc-47778, Santa Cruz Biotechnology, Santa Cruz, CA, USA).
- Subjects and cell culture
- The orbital adipose/connective tissue specimens were harvested from orbital decompression performed on 15 patients with GO having proptosis (12 females and three males; age, 24 to 62 years). During the surgery, all patients were in a stable euthyroid state and did not receive any steroid or radiation therapy for ≤3 months before surgery. The control tissue samples were obtained from 14 healthy control subjects who underwent blepharoplasty, all without thyroid disease and GO (10 females and four males; age, 24 to 69 years). Written informed consent was obtained from all the participants of the study. The protocol of this study, including specimen acquisition, was approved by the Institutional Review Board of Soonchunhyang Hospital, Soonchunhyang University College of Medicine (2020-04-006) and Severance Hospital, Yonsei University College of Medicine (4-2022- 0272). The work was done in accordance with the ethical principles for medical research involving human subjects outlined in the Declaration of Helsinki. The detailed clinical characteristics of all the participants are presented in Supplemental Table S1.
- Orbital fibroblasts were cultured as described in a previous study [24]. Briefly, the tissue explants were minced and placed directly in DMEM/F12 (1:1 ratio) containing 20% FBS, 100 U/mL penicillin, and 100 μg/mL streptomycin. After cell growth was confirmed, the cultured monolayers were passaged serially with the trypsin/ethylenediaminetetraacetic acid solution. The cells were cultured in a humidified 5% CO2 incubator at 37°C. The cell strains used in this study were stored in liquid N2 until utilized, and cells from the third–seventh passages were used for experiments.
- Cell viability assay
- To determine the non-toxic concentration of U73122 for treating the cells, orbital fibroblasts from patients with GO were seeded into 24-well culture plates (1×105 cells/well) and treated with different concentrations of U73122 (control, 0.1, 0.3, 0.5, 1, and 5 μM) for 24 and 48 hours. Thereafter, the treated cells were washed and incubated with the MTT solution (5 mg/mL) for 3 hours at 37°C, then solubilized in ice-cold isopropanol. The absorbance of the dye was measured using a microplate reader (VersaMax, Molecular Devices, Sunnyvale, CA, USA) at 560 nm with background subtraction at 630 nm, based on which the corresponding cell viability was assessed.
- Reverse transcription-quantitative polymerase chain reaction
- To assess phospholipase C gamma 1 (PLCG1) and phospholipase C gamma 2 (PLCG2) mRNA levels in each orbital sample, Precellys 24 tissue homogenizer (catalog number P000669-PR 240-A, Bertin Instruments, Montigny-le-Bretonneux, France) and Precellys lysing kit (catalog number KT03961-007.2, Bertin Instruments) were used for homogenizing the tissue samples. The total RNA was extracted using TRIzol (catalog number 15596026, Invitrogen, Carlsbad, CA, USA), and complementary DNA (cDNA) was synthesized from 1 μg of the isolated RNA according to the manufacturer’s protocol (catalog number 74104, Qiagen, Valencia, CA, USA). The cDNA was amplified with SYBR Green polymerase chain reaction (PCR) Master Mix (catalog number RR420A, Takara Bio, Kusatsu, Japan) in a QuantStudio 3 real-time PCR thermocycler (catalog number A28136, Applied Biosystems). The PCR conditions were set as follows: 50°C for 2 minutes and 95°C for 10 minutes; followed by 40 cycles of 95°C for 15 seconds and 60°C for 1 minute; followed by 95°C for 15 seconds, 60°C for 1 minute, and 95°C for 15 seconds. All PCRs were performed in triplicate, and the primer sequences used are listed in Table 1. The glyceraldehyde3-phosphate dehydrogenase level was used to normalize the results, and the 2− ΔΔCq method was used to present the results as the relative fold changes in the threshold cycle value [25].
- Western blotting assay
- For the Western blotting assay, orbital fibroblasts were washed with phosphate-buffered saline to remove any attached reagents and lysed in lysis buffer as described previously [24]. The cell lysates were centrifuged for 15 minutes at 14,000 rpm and the pellets were discarded. The protein concentrations in the supernatant fractions were quantified using the bicinchoninic acid assay, and the extracted proteins were denatured and resolved by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis. Subsequently, the electrophoresed proteins were transferred onto nitrocellulose membranes (Millipore Corporation, Billerica, MA, USA), treated with primary antibodies (1:1,000 dilution), and incubated overnight at 4°C. The membranes were blocked with 5% skim milk and incubated with horseradish peroxidase-conjugated secondary antibody for 1 hour at room temperature; the immunoreactive bands were visualized using chemiluminescence (Thermo Fisher Scientific). The relative protein amount in each band was quantified through densitometry using the ImageJ software version 1.54 (National Institutes of Health, Bethesda, MD, USA) and normalized to that of β-actin in the same sample.
- Statistical analysis
- At least three strains from three different individuals were used in the experiments and each experiment was performed in triplicates. The Mann-Whitney U test and Kruskal-Wallis test were performed to analyze the nonparametric data. Post hoc analysis was performed using Dunn’s multiple comparison tests for pairwise comparisons between conditions. The Kolmogorov-Smirnov test was used to analyze the data that were not normally distributed. The experimental results in this study are expressed as mean±standard deviation. For statistical analyses, the SPSS version 22 (IBM, Armonk, NY, USA) was used, and a P<0.05 indicated statistical significance.
METHODS
- PLCG1 and PLCG2 mRNA expression in GO and normal orbital tissues
- The transcript levels of PLCG1 and PLCG2 were quantitatively compared using reverse transcription‑quantitative polymerase chain reaction (RT-qPCR) in orbital tissues from patients with GO (n=15; age, 39.13±10.88 years) and healthy control (n=14; age, 44±13.70 years) subjects. The analysis revealed significantly higher expression of both PLCG1 and PLCG2 in GO tissues than in normal orbital tissues (Fig. 1). The mean expression of PLCG1 in GO orbital tissues was 2.1-fold higher than that in normal tissues, whereas the mean expression of PLCG2 was 4.2-fold higher than that in normal tissues.
- IL-1β, TNF-α, and CD40L upregulated PLCG1 and PLCG2 mRNA expression in GO fibroblasts
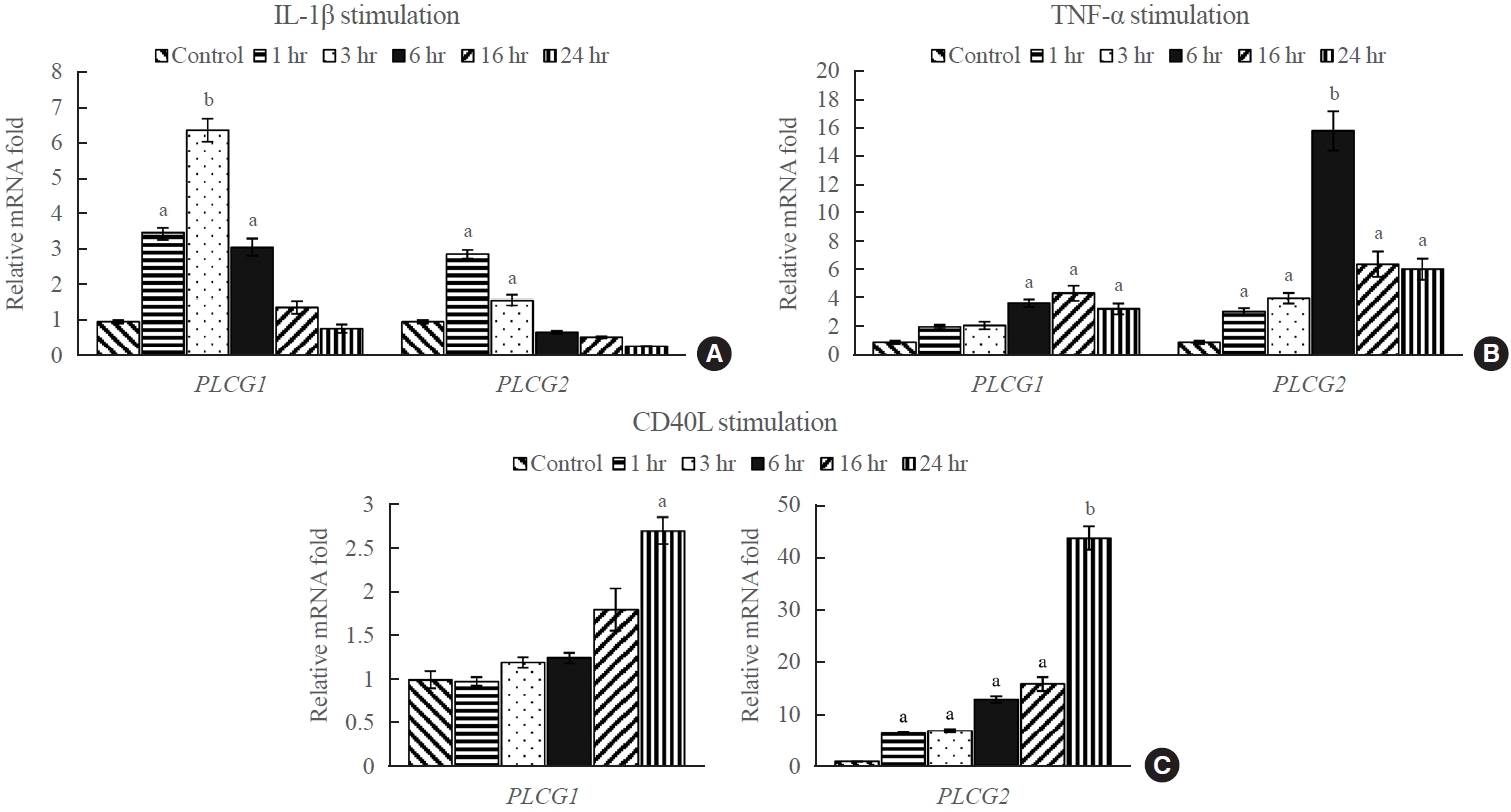
- To investigate the effect of external stimuli on PLCG1 and PLCG2 mRNA expression, the transcript levels of PLCG1 and PLCG2 in GO fibroblasts were evaluated after treatment with various stimulants, including IL-1β (10 ng/mL), TNF-α (10 ng/mL), and CD40L (1 μg/mL). Quantitative analysis using RT-qPCR was performed at 1, 3, 6, 16, and 24 hours after treatment. The results indicated a significant increase in the expression of PLCG1 and PLCG2 following treatment with IL-1β, TNF-α, and CD40L compared with that the untreated controls (Fig. 2). Specifically, PLCG1 mRNA expression exhibited the highest values after 3 hours of IL-1β treatment, 16 hours of TNF-α treatment, and 24 hours of CD40L treatment, with approximate fold increases of 6.4, 4.5, and 2.7, respectively, compared with the corresponding control groups. Contrastingly, PLCG2 mRNA expression exhibited the highest increase after 1 hour of IL-1β treatment, 6 hours of TNF-α treatment, and 24 hours of CD40L treatment, with approximate fold increases of 2.9, 16, and 44, respectively, compared with the corresponding control groups. Unlike treatments with IL-1β and TNF-α, CD40L treatment exhibited a gradual increase in the expression of PLCG1 and PLCG2 throughout the experimental period. The increase was especially prominent in PLCG2 mRNA expression, exhibiting an increase of more than 40-fold compared with the corresponding control at 24 hours of treatment (P<0.01).
- Effect of U73122 on cell viability
- We determined the non-toxic concentrations of U73122 on orbital fibroblasts using the MTT assay. Orbital fibroblasts from GO were treated with U73122 (0.1 to 5 μM) to determine its effect on viability for 24 and 48 hours. Cell viability was expressed as a percentage relative to the respective control. Results obtained from treatment with 1 μM or lower concentrations of U73122 indicated that cell viability did not significantly decrease regardless of the time (Supplemental Fig. S1). Based on these results, the maximal non-toxic concentration of U73122 in orbital fibroblasts was considered at 1 μM for 48 hours, and this concentration was used for subsequent experiments.
- U73122 inhibits PLCG1 and PLCG2 expression
- The effect of U73122 on PLCG1 and PLCG2 mRNA expression was assayed through RT-qPCR. GO fibroblasts were treated with 1 μM of U73122 for 3, 24, and 48 hours. The results indicated that U73122 significantly decreased the transcript levels of PLCG1 and PLCG2 in GO fibroblasts regardless of time (Supplemental Fig. S2). The average transcriptional inhibitory effect of U73122 on the PLCG1 gene was highest at 48 hours after treatment, whereas it was highest at 24 hours after treatment for PLCG2.
- U73122 inhibits IL-1β-induced expression of proinflammatory molecules
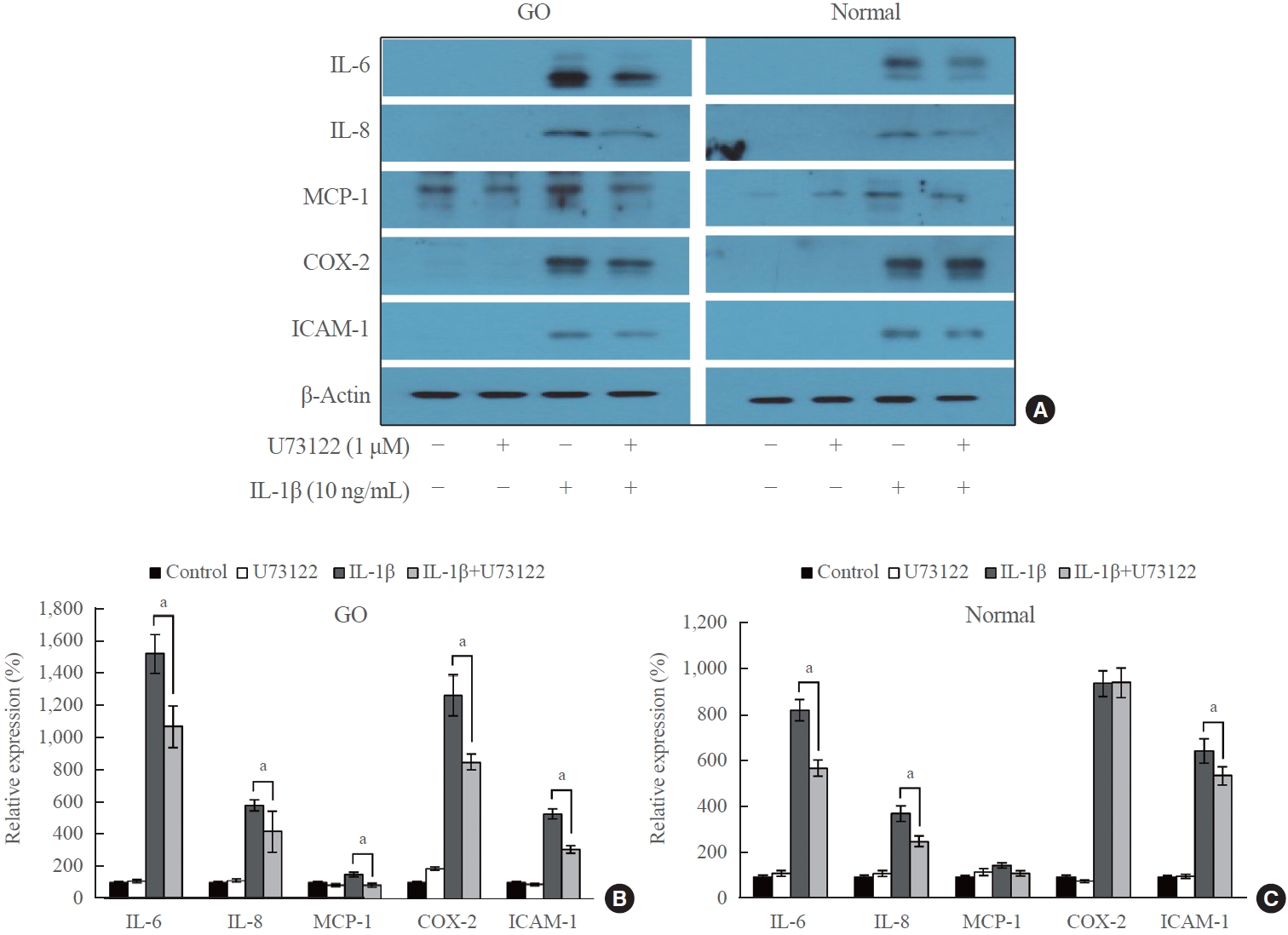
- Western blot analysis was performed to investigate the effect of U73122 on the protein expression of IL-1β-induced proinflammatory molecules (IL-6, IL-8, MCP-1, COX-2, and ICAM-1) in orbital fibroblasts. GO and normal orbital fibroblasts were treated with U73122 (1 μM) with or without IL-1β stimulation (10 ng/mL) for 48 hours (Fig. 3). The results indicated an increase in proinflammatory molecules in both GO and normal fibroblasts after 48 hours of IL-1β treatment. Notably, IL-6 expression in GO cells exhibited the highest increase, with a 15.2-fold increase by IL-1β induction, followed by a 12.6-fold increase in COX-2 expression. The expression of all IL-1β-induced molecules, including IL-6 and COX-2, significantly decreased in GO fibroblasts treated with U73122. In normal cells, IL-1β induced the highest increase in COX-2 expression by 9.4-fold, followed by an 8.3-fold increase in IL-6. However, the IL-1β-induced expression of MCP-1 and COX-2 was not significantly attenuated by U73122 in normal orbital fibroblasts, whereas the IL-1β-induced expression of IL-6, IL-8, and ICAM-1 was significantly diminished.
- Effect of U73122 on the phosphorylation of signaling molecules
- To investigate the signaling pathways affected by U73122, Western blotting analysis was performed on many downstream signaling kinases in GO and normal orbital fibroblasts. GO and normal orbital fibroblasts were treated with U73122 (1 μM) with or without IL-1β (10 ng/mL) for 1 hour (Fig. 4). Following treatment with IL-1β, the phosphorylated forms of signaling molecules involved in the inflammatory response, including Akt and MAPKs (ERK, p38, and JNK), were increased in both GO and normal orbital fibroblasts. When U73122 was treated for 1 hour, the expression of p-Akt induced by IL-1β significantly decreased in GO and normal cells. Moreover, U73122 significantly suppressed IL-1β-induced p-p38 expression in GO fibroblasts and significantly inhibited IL-1β-induced p-JNK expression in normal fibroblasts. However, no significant inhibitory effect of U73122 was observed on other signaling molecules.
RESULTS
- Based on the pathogenesis of GO, we investigated the roles of PLC-γ in orbital tissues and the molecular pathways and interactions involved using primary cultures of orbital fibroblasts. PLCG1 and PLCG2 were overexpressed in GO tissues compared with normal orbital tissues. The production of proinflammatory cytokines, which is closely associated with the development of GO, was upregulated after IL-1β treatment in GO fibroblasts but was significantly suppressed after treatment with U73122, a well-known PLC inhibitor. To the best of our knowledge, this is the first study to show that PLC-γ mediates the inflammatory reactions in GO fibroblasts.
- PLC-γ has been implicated in various human diseases and modulates the immune system by activating the downstream of various cell surface receptors [10,17,26]. The T-cell receptor complex recruits IL-2-inducible T-cell kinase (ITK) to activate PLC-γ1 [27]; similarly, the B-cell receptor recruits kinases, such as spleen tyrosine kinase (Syk) and Bruton’s tyrosine kinase (BTK), which activate PLC-γ2 [26]. The activation of PLC-γ ultimately leads to downstream signaling that activates transcription factors, including the nuclear factor of activated T-cells (NFAT), activator protein-1 (AP-1), and nuclear factor κB (NF-κB) [28,29]. NF-κB, a ubiquitous transcription factor, plays a fundamental role in mediating inflammatory and immune responses, including CD40-mediated interactions between orbital fibroblasts and T-cells [30,31]. CD40 is a transmembrane protein that is overexpressed in GO fibroblasts, and the CD40–CD40L interaction plays a crucial role in perpetuating orbital inflammation [32]. In this study, we found that PLCG1 and PLCG2 mRNA expression was higher in GO tissues than in control, and their expression was significantly increased by stimulation with IL-1β, TNF-α, and CD40L in GO fibroblasts. Notably, PLCG2 mRNA expression increased more than 40-fold with 1 μg/mL of CD40L, suggesting that PLC-γ may play a crucial role in orbital inflammation.
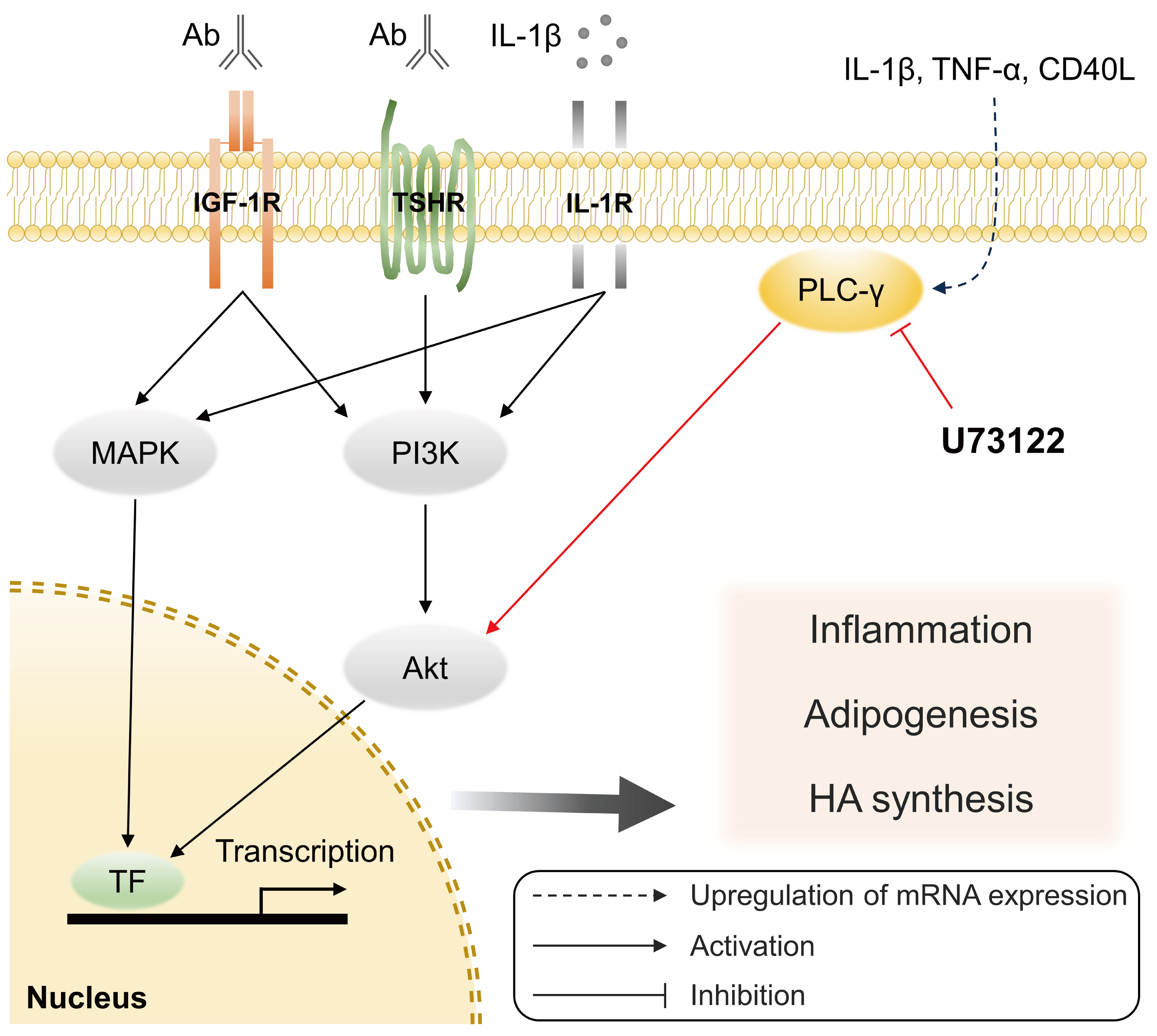
- The phosphatidylinositol 3-kinase (PI3K) signaling, which is essential for the development, activation, and differentiation of T- and B-cells [33], plays a crucial role in the pathogenesis of GO [34,35]. The kinase Akt is a key PI3K effector, and the PI3K-Akt signaling pathway activated in GO fibroblasts by stimulation of TSHR and IGF-1R is associated with adipogenesis and hyaluronan production [34-36]. Our previous study showed that the production of proinflammatory cytokines and adipogenesis induced by treating IL-1β in GO fibroblasts was inhibited by the specific PI3Kδ inhibitor, idelalisib [37]. Furthermore, studies have suggested that PLC-γ downstream signaling pathway interacts with the PI3K-Akt pathway in several cell types [38,39]. Using U73122 in Balb 3T3 fibroblasts, Amin et al. [40] showed that the PLC-PKC signaling cascade involving PLC-γ was required for IL-1β-induced Akt activation. Similarly, we showed that the phosphorylation of Akt and MAPK was increased in both GO and control fibroblasts after IL-1β treatment, and inhibiting PLC-γ via U73122 treatment significantly suppressed the expression of p-Akt and p-p38 in GO fibroblasts. This suggests that PLC-γ may play an important role in activating the PI3K-Akt pathway, which is triggered by surface receptors on orbital fibroblasts, including the TSHR and IGF-1R (Fig. 5).
- Previous studies have suggested that PLC-γ plays an important role in the innate immune system. Triggering receptor expressed on myeloid cells 1 (TREM1) is a receptor expressed on neutrophils and monocytes/macrophages, and its downstream signaling increases inflammation [41]; PLC-γ-induced calcium mobilization and activation of transcription factors are involved in this pathway [41]. Further, Aki et al. [13] showed that PLC-γ2 is required to induce normal production of TNF-α and IL-6 when stimulated with peptidoglycan (a ligand for TLR2) and lipopolysaccharide (LPS) (a ligand for TLR4) using bone marrow-derived macrophages. In the context of GO, the levels of macrophage infiltration in the orbital adipose tissue were significantly higher in patients with GO than in normal controls, which exhibited a significant positive correlation with the CCL2 mRNA expression [42]. Moreover, our previous study revealed that TLR2 and TLR4 expression was significantly higher in GO tissues than in controls and TLR2 blockade with antibodies significantly decreased the release of proinflammatory cytokines [43]. These findings indicate that PLC-γ activation may increase the inflammatory response in GO via the innate and adaptive immune system.
- The aminosteroid U73122 is the most widely used PLC inhibitor, and several previous studies have used U73122 as a PLC-γ inhibitor [16,39,40,44]. In this study, we showed that U73122 significantly inhibited the mRNA expression of PLCG1 and PLCG2 in GO fibroblasts. Although the underlying precise mechanism is yet to be elucidated, these findings are consistent with a previous study that indicated that U73122 affects both PLC activity and gene expression [45]. The blockade of PLC and Ca2+ accumulation by U73122 inhibits LPS-induced mRNA expression of proinflammatory cytokines and NF-κB nuclear translocation in human gingival fibroblasts [23]. An in vivo study showed that LPS-induced cardiac Tnf expression and myocardial dysfunction were significantly suppressed in heterozygous phospholipase C, gamma 1 (Plcg1) knockout mice and U73122-treated wild-type mice [46]. These results indicate that the treatment with U73122 leads to the blockade of inflammation both in vitro and in vivo. Our study also showed that U73122 treatment on GO fibroblasts significantly suppressed the IL-1β-induced expression of proinflammatory molecules, suggesting that the inhibition of PLC-γ can potentially exhibit a therapeutic effect by modulating the inflammatory response in GO.
- However, there are some limitations in the study. The absence of reported therapeutic applications or clinical trials involving U73122 in human clinical studies leads to insufficient pharmacological information. Moreover, U73122 may not be highly specific for PLC because it can inhibit Ca2+ pumps [47], potassium channels [48], and enzymes, including 5-lipoxygenase [49], in various cells. Based on the evidence that U73122 is less selective for PLC inhibition, the results using U73122 to analyze the roles and the molecular mechanisms of PLC-γ should be carefully interpreted. Inhibitors that can reduce off-target effects and directly modulate PLC-γ enzymes should be developed. Additionally, in this study using orbital fibroblasts, the interactions with T-cells, B-cells, and innate immune cells related to GO pathogenesis were not sufficiently considered. While orbital fibroblasts may not completely encompass the inflammatory response seen in patients with GO, we did simulate the environment using IL-1β, a proinflammatory cytokine that is highly associated with GO pathogenesis [24,50].
- To conclude, in this study, we showed that U73122, which is used to inhibit PLC-γ, exhibits anti-inflammatory effects on orbital fibroblasts in patients with GO. We postulate that the inhibition of PLC-γ can be a potential candidate for GO therapy. Further in vivo studies using animal models and clinical trials are required to elucidate the specific mechanism of targeting PLC-γ in the pathogenic mechanism of GO.
DISCUSSION
Supplementary Material
Supplemental Table S1.
Supplemental Fig. S1.
Supplemental Fig. S2.
-
CONFLICTS OF INTEREST
Don O. Kikkawa is a former consultant of Horizon Therapeutics. Remaining authors have no potential conflict of interest to declare.
-
AUTHOR CONTRIBUTIONS
Conception or design: S.Y.J., J.S.Y. Acquisition, analysis, or interpretation of data: T.H.R., M.K.C., J.S.K., J.S.Y. Drafting the work or revising: T.H.R., D.O.K. Final approval of the manuscript: T.H.R., M.K.C., J.S.K., D.O.K., S.Y.J., J.S.Y.
Article information
-
Acknowledgements
- This work was supported by the National Research Foundation of Korea (NRF) grant funded by the government of Korea (MSIT) (No. 2021R1F1A1046652). This study was also supported by the Student Research Bursary of Yonsei University College of Medicine (2023) and Soonchunhyang University Research Fund. A portion of this study was presented in an abstract form at the 129th Annual Meeting of the Korean Ophthalmological Society, April 7 to 8, 2023, Busan, Republic of Korea.


- 1. Wiersinga WM. Advances in treatment of active, moderateto-severe Graves’ ophthalmopathy. Lancet Diabetes Endocrinol 2017;5:134–42.ArticlePubMed
- 2. van Steensel L, Dik WA. The orbital fibroblast: a key player and target for therapy in graves’ ophthalmopathy. Orbit 2010;29:202–6.ArticlePubMed
- 3. Tsui S, Naik V, Hoa N, Hwang CJ, Afifiyan NF, Sinha Hikim A, et al. Evidence for an association between thyroidstimulating hormone and insulin-like growth factor 1 receptors: a tale of two antigens implicated in Graves’ disease. J Immunol 2008;181:4397–405.ArticlePubMedPMCPDF
- 4. Dik WA, Virakul S, van Steensel L. Current perspectives on the role of orbital fibroblasts in the pathogenesis of Graves’ ophthalmopathy. Exp Eye Res 2016;142:83–91.ArticlePubMed
- 5. Lehmann GM, Feldon SE, Smith TJ, Phipps RP. Immune mechanisms in thyroid eye disease. Thyroid 2008;18:959–65.ArticlePubMedPMC
- 6. Bahn RS. Graves’ ophthalmopathy. N Engl J Med 2010;362:726–38.ArticlePubMedPMC
- 7. Taylor PN, Zhang L, Lee RW, Muller I, Ezra DG, Dayan CM, et al. New insights into the pathogenesis and nonsurgical management of Graves orbitopathy. Nat Rev Endocrinol 2020;16:104–16.ArticlePubMedPDF
- 8. Kadamur G, Ross EM. Mammalian phospholipase C. Annu Rev Physiol 2013;75:127–54.ArticlePubMed
- 9. Yang YR, Follo MY, Cocco L, Suh PG. The physiological roles of primary phospholipase C. Adv Biol Regul 2013;53:232–41.ArticlePubMed
- 10. Koss H, Bunney TD, Behjati S, Katan M. Dysfunction of phospholipase Cγ in immune disorders and cancer. Trends Biochem Sci 2014;39:603–11.ArticlePubMed
- 11. Nakamura Y, Fukami K. Regulation and physiological functions of mammalian phospholipase C. J Biochem 2017;161:315–21.ArticlePubMed
- 12. Bae YS, Lee JH, Choi SH, Kim S, Almazan F, Witztum JL, et al. Macrophages generate reactive oxygen species in response to minimally oxidized low-density lipoprotein: tolllike receptor 4- and spleen tyrosine kinase-dependent activation of NADPH oxidase 2. Circ Res 2009;104:210–8.PubMed
- 13. Aki D, Minoda Y, Yoshida H, Watanabe S, Yoshida R, Takaesu G, et al. Peptidoglycan and lipopolysaccharide activate PLCgamma2, leading to enhanced cytokine production in macrophages and dendritic cells. Genes Cells 2008;13:199–208.PubMed
- 14. Zhou Q, Lee GS, Brady J, Datta S, Katan M, Sheikh A, et al. A hypermorphic missense mutation in PLCG2, encoding phospholipase Cγ2, causes a dominantly inherited autoinflammatory disease with immunodeficiency. Am J Hum Genet 2012;91:713–20.ArticlePubMedPMC
- 15. Afroz S, Giddaluru J, Vishwakarma S, Naz S, Khan AA, Khan N. A comprehensive gene expression meta-analysis identifies novel immune signatures in rheumatoid arthritis patients. Front Immunol 2017;8:74.ArticlePubMedPMC
- 16. Zhu L, Yuan C, Ding X, Xu S, Yang J, Liang Y, et al. PLC-γ1 is involved in the inflammatory response induced by influenza A virus H1N1 infection. Virology 2016;496:131–7.ArticlePubMed
- 17. Mandal S, Bandyopadhyay S, Tyagi K, Roy A. Recent advances in understanding the molecular role of phosphoinositide-specific phospholipase C gamma 1 as an emerging oncodriver and novel therapeutic target in human carcinogenesis. Biochim Biophys Acta Rev Cancer 2021;1876:188619.ArticlePubMed
- 18. Yu P, Constien R, Dear N, Katan M, Hanke P, Bunney TD, et al. Autoimmunity and inflammation due to a gain-of-function mutation in phospholipase C gamma 2 that specifically increases external Ca2+ entry. Immunity 2005;22:451–65.ArticlePubMed
- 19. Abe K, Fuchs H, Boersma A, Hans W, Yu P, Kalaydjiev S, et al. A novel N-ethyl-N-nitrosourea-induced mutation in phospholipase Cγ2 causes inflammatory arthritis, metabolic defects, and male infertility in vitro in a murine model. Arthritis Rheum 2011;63:1301–11.ArticlePubMed
- 20. Cremasco V, Graham DB, Novack DV, Swat W, Faccio R. Vav/phospholipase Cgamma2-mediated control of a neutrophil-dependent murine model of rheumatoid arthritis. Arthritis Rheum 2008;58:2712–22.PubMedPMC
- 21. Cremasco V, Benasciutti E, Cella M, Kisseleva M, Croke M, Faccio R. Phospholipase C gamma 2 is critical for development of a murine model of inflammatory arthritis by affecting actin dynamics in dendritic cells. PLoS One 2010;5:e8909.ArticlePubMedPMC
- 22. Zhu L, Yuan C, Ma Y, Ding X, Zhu G, Zhu Q. Anti-inflammatory activities of phospholipase C inhibitor U73122: inhibition of monocyte-to-macrophage transformation and LPSinduced pro-inflammatory cytokine expression. Int Immunopharmacol 2015;29:622–7.ArticlePubMed
- 23. Zhou Z, Xi R, Liu J, Peng X, Zhao L, Zhou X, et al. TAS2R16 activation suppresses LPS-induced cytokine expression in human gingival fibroblasts. Front Immunol 2021;12:726546.ArticlePubMedPMC
- 24. Yoon JS, Lee HJ, Choi SH, Chang EJ, Lee SY, Lee EJ. Quercetin inhibits IL-1β-induced inflammation, hyaluronan production and adipogenesis in orbital fibroblasts from Graves’ orbitopathy. PLoS One 2011;6:e26261.ArticlePubMedPMC
- 25. Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 2001;25:402–8.ArticlePubMed
- 26. Jackson JT, Mulazzani E, Nutt SL, Masters SL. The role of PLCγ2 in immunological disorders, cancer, and neurodegeneration. J Biol Chem 2021;297:100905.ArticlePubMedPMC
- 27. Paul S, Schaefer BC. A new look at T cell receptor signaling to nuclear factor-κB. Trends Immunol 2013;34:269–81.ArticlePubMedPMC
- 28. Fu G, Chen Y, Yu M, Podd A, Schuman J, He Y, et al. Phospholipase C{gamma}1 is essential for T cell development, activation, and tolerance. J Exp Med 2010;207:309–18.PubMedPMC
- 29. de Gorter DJ, Vos JC, Pals ST, Spaargaren M. The B cell antigen receptor controls AP-1 and NFAT activity through Ras-mediated activation of Ral. J Immunol 2007;178:1405–14.ArticlePubMedPDF
- 30. Sempowski GD, Rozenblit J, Smith TJ, Phipps RP. Human orbital fibroblasts are activated through CD40 to induce proinflammatory cytokine production. Am J Physiol 1998;274:C707–14.ArticlePubMed
- 31. Liu Z, Liu Y, Liu M, Gong Q, Shi A, Li X, et al. PD-L1 inhibits T cell-induced cytokines and hyaluronan expression via the CD40-CD40L pathway in orbital fibroblasts from patients with thyroid associated ophthalmopathy. Front Immunol 2022;13:849480.ArticlePubMedPMC
- 32. Hwang CJ, Afifiyan N, Sand D, Naik V, Said J, Pollock SJ, et al. Orbital fibroblasts from patients with thyroid-associated ophthalmopathy overexpress CD40: CD154 hyperinduces IL-6, IL-8, and MCP-1. Invest Ophthalmol Vis Sci 2009;50:2262–8.ArticlePubMedPMC
- 33. So L, Fruman DA. PI3K signalling in B- and T-lymphocytes: new developments and therapeutic advances. Biochem J 2012;442:465–81.ArticlePubMedPMCPDF
- 34. Kumar S, Nadeem S, Stan MN, Coenen M, Bahn RS. A stimulatory TSH receptor antibody enhances adipogenesis via phosphoinositide 3-kinase activation in orbital preadipocytes from patients with Graves’ ophthalmopathy. J Mol Endocrinol 2011;46:155–63.ArticlePubMedPMC
- 35. Draman MS, Zhang L, Dayan C, Ludgate M. Orbital signaling in Graves’ orbitopathy. Front Endocrinol (Lausanne) 2021;12:739994.ArticlePubMedPMC
- 36. Zhao P, Deng Y, Gu P, Wang Y, Zhou H, Hu Y, et al. Insulinlike growth factor 1 promotes the proliferation and adipogenesis of orbital adipose-derived stromal cells in thyroidassociated ophthalmopathy. Exp Eye Res 2013;107:65–73.ArticlePubMed
- 37. Ko J, Kim JY, Lee EJ, Yoon JS. Inhibitory effect of idelalisib, a selective phosphatidylinositol 3-kinase δ inhibitor, on adipogenesis in an in vitro model of Graves’ orbitopathy. Invest Ophthalmol Vis Sci 2018;59:4477–85.ArticlePubMed
- 38. Li D, Shatos MA, Hodges RR, Dartt DA. Role of PKCα activation of Src, PI-3K/AKT, and ERK in EGF-stimulated proliferation of rat and human conjunctival goblet cells. Invest Ophthalmol Vis Sci 2013;54:5661–74.ArticlePubMedPMC
- 39. Wu D, Peng F, Zhang B, Ingram AJ, Kelly DJ, Gilbert RE, et al. EGFR-PLCgamma1 signaling mediates high glucoseinduced PKCbeta1-Akt activation and collagen I upregulation in mesangial cells. Am J Physiol Renal Physiol 2009;297:F822–34.PubMed
- 40. Amin AR, Ichigotani Y, Oo ML, Biswas MH, Yuan H, Huang P, et al. The PLC-PKC cascade is required for IL-1beta-dependent Erk and Akt activation: their role in proliferation. Int J Oncol 2003;23:1727–31.PubMed
- 41. Colonna M. TREMs in the immune system and beyond. Nat Rev Immunol 2003;3:445–53.ArticlePubMedPDF
- 42. Chen MH, Chen MH, Liao SL, Chang TC, Chuang LM. Role of macrophage infiltration in the orbital fat of patients with Graves’ ophthalmopathy. Clin Endocrinol (Oxf) 2008;69:332–7.ArticlePubMed
- 43. Han SY, Choi SH, Shin JS, Lee EJ, Han SH, Yoon JS. High-mobility group box 1 is associated with the inflammatory pathogenesis of Graves’ orbitopathy. Thyroid 2019;29:868–78.ArticlePubMed
- 44. Glassford J, Soeiro I, Skarell SM, Banerji L, Holman M, Klaus GG, et al. BCR targets cyclin D2 via Btk and the p85alpha subunit of PI3-K to induce cell cycle progression in primary mouse B cells. Oncogene 2003;22:2248–59.ArticlePubMedPDF
- 45. Lo Vasco VR, Leopizzi M, Di Maio V, Della Rocca C. U73122 reduces the cell growth in cultured MG-63 ostesarcoma cell line involving phosphoinositide-specific phospholipases C. Springerplus 2016;5:156.PubMedPMC
- 46. Peng T, Shen E, Fan J, Zhang Y, Arnold JM, Feng Q. Disruption of phospholipase Cgamma1 signalling attenuates cardiac tumor necrosis factor-alpha expression and improves myocardial function during endotoxemia. Cardiovasc Res 2008;78:90–7.PubMed
- 47. Macmillan D, McCarron JG. The phospholipase C inhibitor U-73122 inhibits Ca(2+) release from the intracellular sarcoplasmic reticulum Ca(2+) store by inhibiting Ca(2+) pumps in smooth muscle. Br J Pharmacol 2010;160:1295–301.ArticlePubMedPMC
- 48. Klose A, Huth T, Alzheimer C. 1-[6-[[(17beta)-3-methoxyestra-1,3,5(10)-trien -17-yl]amino]hexyl]-1H-pyrrole-2,5-dione (U73122) selectively inhibits Kir3 and BK channels in a phospholipase C-independent fashion. Mol Pharmacol 2008;74:1203–14.ArticlePubMed
- 49. Feisst C, Albert D, Steinhilber D, Werz O. The aminosteroid phospholipase C antagonist U-73122 (1-[6-[[17-beta-3-methoxyestra-1,3,5(10)-trien-17-yl]amino]hexyl]-1H-pyrrole-2, 5-dione) potently inhibits human 5-lipoxygenase in vivo and in vitro. Mol Pharmacol 2005;67:1751–7.ArticlePubMed
- 50. Chen B, Tsui S, Smith TJ. IL-1 beta induces IL-6 expression in human orbital fibroblasts: identification of an anatomicsite specific phenotypic attribute relevant to thyroid-associated ophthalmopathy. J Immunol 2005;175:1310–9.PubMed
References
Figure & Data
References
Citations


 KES
KES

 PubReader
PubReader ePub Link
ePub Link Cite
Cite