Articles
- Page Path
- HOME > Endocrinol Metab > Volume 38(6); 2023 > Article
-
Original ArticleCalcium & bone metabolism Higher Plasma Stromal Cell-Derived Factor 1 Is Associated with Lower Risk for Sarcopenia in Older Asian Adults
 Keypoint
Keypoint
In a study of older adults in South Korea, lower plasma SDF-1 levels were significantly associated with sarcopenia and reduced muscle mass. Higher SDF-1 levels correlated with better skeletal muscle mass and gait speed. The research suggests that circulating SDF-1 could serve as a biomarker for muscle health in the elderly, reflecting its anabolic effects on muscle observed in the clinical setting. -
Sunghwan Ji1*
 , Kyunggon Kim2,3*
, Kyunggon Kim2,3* , So Jeong Park2, Jin Young Lee2, Hee-Won Jung1, Hyun Ju Yoo4, Il-Young Jang1, Eunju Lee1, Ji Yeon Baek1
, So Jeong Park2, Jin Young Lee2, Hee-Won Jung1, Hyun Ju Yoo4, Il-Young Jang1, Eunju Lee1, Ji Yeon Baek1 , Beom-Jun Kim5
, Beom-Jun Kim5
-
Endocrinology and Metabolism 2023;38(6):701-708.
DOI: https://doi.org/10.3803/EnM.2023.1783
Published online: October 18, 2023
1Division of Geriatrics, Department of Internal Medicine, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
2Asan Institute for Life Sciences, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
3Department of Digital Medicine, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
4Department of Convergence Medicine, Asan Institute for Life Sciences, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
5Division of Endocrinology and Metabolism, Department of Internal Medicine, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
- Corresponding authors: Beom-Jun Kim. Division of Endocrinology and Metabolism, Department of Internal Medicine, Asan Medical Center, University of Ulsan College of Medicine, 88 Olympic-ro 43-gil, Songpa-gu, Seoul 05505, Korea Tel: +82-2-3010-5876, Fax: +82-2-3010-6962, E-mail: umkbj0825@amc.seoul.kr
- Ji Yeon Baek. Division of Geriatrics, Department of Internal Medicine, Asan Medical Center, University of Ulsan College of Medicine, 88 Olympic-ro 43-gil, Songpa-gu, Seoul 05505, Korea Tel: +82-2-3010-0020, Fax: +82-2-3010-6962, E-mail: complete_flow@naver.com
- *These authors contributed equally to this work.
Copyright © 2023 Korean Endocrine Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 1,732 Views
- 77 Download
ABSTRACT
-
Background
- Despite the protective effects of stromal cell-derived factor 1 (SDF-1) in stimulating muscle regeneration shown in experimental research, there is a lack of clinical studies linking circulating SDF-1 concentrations with muscle phenotypes. In order to elucidate the role of SDF-1 as a potential biomarker reflecting human muscle health, we investigated the association of plasma SDF-1 levels with sarcopenia in older adults.
-
Methods
- This cross-sectional study included 97 community-dwelling participants who underwent a comprehensive geriatric assessment at a tertiary hospital in South Korea. Sarcopenia was defined by specific cutoff values applicable to the Asian population, whereas plasma SDF-1 levels were determined using an enzyme immunoassay.
-
Results
- After accounting for sex, age, and body mass index, participants with sarcopenia and low muscle mass exhibited plasma SDF-1 levels that were 21.8% and 18.3% lower than those without these conditions, respectively (P=0.008 and P=0.009, respectively). Consistently, higher plasma SDF-1 levels exhibited a significant correlation with higher skeletal muscle mass index (SMI) and gait speed (both P=0.043), and the risk of sarcopenia and low muscle mass decreased by 58% and 55% per standard deviation increase in plasma SDF-1 levels, respectively (P=0.045 and P=0.030, respectively). Furthermore, participants in the highest SDF-1 tertile exhibited significantly higher SMI compared to those in the lowest tertile (P=0.012).
-
Conclusion
- These findings clinically corroborate earlier experimental discoveries highlighting the muscle anabolic effects of SDF-1 and support the potential role of circulating SDF-1 as a biomarker reflecting human muscle health in older adults.
- Sarcopenia, a term used to describe progressive decline in skeletal muscle mass and function due to the imbalance between protein breakdown and synthesis, has received heightened attention in the field of geriatrics, particularly over the past decade [1,2]. The pathophysiology of sarcopenia is complex, arising from biological changes in muscle structure, disruptions in hormone levels, and external factors like inadequate energy intake or reduced physical activity [3,4]. While sarcopenia has traditionally been considered an inevitable consequence of aging, there has been growing recognition that increasing knowledge about muscle metabolism makes skeletal muscle deterioration possibly reversible and thus sarcopenia is now perceived as a disease to be overcome [4,5]. Importantly, sarcopenia is intricately tied to negative outcomes, such as falls, frailty, and diminishing functionality [6,7], all of which compromise the ability of older adults to maintain their independence. Consequently, early detection of individuals prone to sarcopenia with reliable biomarkers and prevention of this condition through effective intervention are essential to enhance overall health and well-being during the later stages of life.
- Stromal cell-derived factor 1 (SDF-1), also referred to as C-X-C motif chemokine ligand 12 (CXCL12), exerts its biologic effects by interacting with the C-X-C chemokine receptor type 4 (CXCR4) or CXCR7 present on target cells [8,9]. In addition to its primary function as a chemoattractant that influences cell migration and homing [10], SDF-1 play a pivotal role in numerous physiological processes, such as angiogenesis, hematopoiesis, tissue regeneration, and immune modulation [11-13]. Importantly, a growing body of evidence indicates that SDF-1 is implicated in muscle metabolism as well. In detail, in vivo experiments using genetically engineered mice demonstrated that the signaling pathway of SDF-1-CXCR4 takes place within the limb mesenchyme during the early stages of development, directly contributing to the formation of muscles of appropriate size [14]. Furthermore, both CXCR4 and CXCR7 are enriched in muscle fibers [15,16], and SDF-1 is known to promote not only the myogenic differentiation but the migration of satellite cells and myoblasts [16-19]. However, despite the protective effects of SDF-1 in stimulating muscle regeneration shown in experimental research, there is a lack of clinical studies linking circulating SDF-1 concentrations with muscle phenotypes. Therefore, in order to elucidate the role of SDF-1 as a potential biomarker reflecting human muscle health, we investigated the association of plasma SDF-1 levels with sarcopenia, as defined by all the recommended diagnostic criteria, in a cohort of older Asian adults.
INTRODUCTION
- Study participants
- This cross-sectional study was conducted among a group of older adults living in the community in South Korea. These individuals underwent a comprehensive functional assessment at the outpatient geriatric clinic of Asan Medical Center, a teaching hospital in Seoul, between March 2019 and February 2021. Their visits to the clinic were either for managing chronic conditions like osteoporosis, osteoarthritis, hyperlipidemia, and hypertension, or for evaluating common nonspecific symptoms associated with aging, such as fatigue and loss of appetite. None of the participants were admitted to hospitals or nursing homes; they were all able to move around independently, with or without walking aids. Individuals with conditions like end-stage renal failure requiring dialysis, symptomatic heart failure, or a life expectancy of less than 1 year due to cancer were not included in the study. All 97 eligible participants agreed to have their blood samples collected and were enrolled in the research after providing written informed consent. The Institutional Review Board of Asan Medical Center (IRB no. 2020-0259) approved the study, and it adhered to the ethical guidelines for human experimentation outlined in the Declaration of Helsinki.
- Evaluation of sarcopenia
- Experienced nurses conducted interviews and analyzed medical records to gather demographic information and medical histories. Body composition, including muscle mass, were assessed using bioelectrical impedance analysis (InBody S10, InBody, Seoul, Korea) at frequencies of 1, 5, 50, 250, 500, and 1,000 kHz. Appendicular skeletal muscle mass (ASM), representing the combined muscle mass of the upper and lower extremities, was calculated. Additionally, the skeletal muscle mass index (SMI) was computed by dividing ASM by the square of the individual’s height (kg/m2). Handgrip strength of the dominant arm was measured with a hand dynamometer (Patterson Medical, Warrenville, IL, USA) [20]. Participants were instructed to sit comfortably, bend their elbows at a 90º angle, and grasp the dynamometer as firmly as possible. Two measurements of grip strength were taken, with at least one minute between measurements, and the maximum value was recorded. Gait speed was determined over a 4-m distance, and the time needed to complete five chair stands was documented [21]. The short physical performance battery (SPPB) was administered, encompassing assessments of repeated chair stands, standing balance, and gait speed [22].
- The 2019 consensus guidelines from the Asian Working Group for Sarcopenia were employed to establish the definition of sarcopenia [23]. Sarcopenia was diagnosed when individuals presented with low muscle mass combined with weak muscle strength and/or poor physical performance. Low muscle mass was characterized by an SMI <7.0 kg/m2 for men and <5.7 kg/m² for women. Weak muscle strength was indicated by a handgrip strength of less than 28 kg for men and less than 18 kg for women. Poor physical performance was identified as a gait speed below 1.0 m/sec, a completion time of 12 seconds or more for the five-time chair stand test, or an SPPB score of 9 points or less.
- Measurement of plasma SDF-1
- Blood samples were drawn from the antecubital veins of all participants after an overnight fast. Subsequently, the samples were centrifuged at 3,000 rpm for 5 minutes at 4°C, and the supernatant was carefully separated to eliminate cellular components. Samples exhibiting signs of hemolysis or clotting were excluded from the analysis. The plasma samples were then stored at –80°C until the measurement of plasma SDF-1 concentrations was performed using the Quantikine enzyme-linked immunosorbent assay (ELISA) reagent (Cat. No. DSA00; R&D Systems, Minneapolis, MN, USA; Aviscera Bioscience Inc., Santa Clara, CA, USA), following the manufacturer’s instructions. The ELISA kit had a lower limit of detection of 47 pg/mL, with intra-assay and inter-assay coefficients of variation both below 3.9% and 13.4%, respectively.
- Statistical analysis
- Data were displayed as the mean±standard deviation (SD), as well as numbers and percentages. To compare the baseline characteristics of participants with and without sarcopenia, continuous variables were analyzed using the Student’s t test and categorical variables were assessed using the chi-square test. Analysis of covariance (ANCOVA) was applied to compare the adjusted means of plasma SDF-1 levels with respect to sarcopenia status and related parameters, while adjusting for sex, age, and body mass index (BMI). Pearson and partial correlation analyses were performed to evaluate the associations between plasma SDF-1 levels and specific muscle parameters relevant to sarcopenia before and after controlling for sex, age, and BMI, respectively. Logistic regression analysis was conducted to calculate the odds ratios (ORs) for the risk of sarcopenia and adverse muscle outcomes according to SD increase in plasma SDF-1 level. ANCOVA was employed again to compare the adjusted means of the sarcopenia parameters among plasma SDF-1 tertiles. All statistical analyses were carried out using R software version 3.6.3 (R Foundation for Statistical Computing, Vienna, Austria), and statistical significance was determined at a two-sided P value of <0.05.
METHODS
- The baseline characteristics of the study population are outlined in Table 1. Among the 97 participants, fourteen individuals fulfilled the criteria for sarcopenia. The mean age of participants with sarcopenia was 72.5±5.1 years, which was slightly higher than the mean age of those without sarcopenia (69.0±6.3 years), though this difference did not reach statistical significance. Participants with sarcopenia tended to be female (P=0.084) and exhibited lower measurements for weight, height, BMI, ASM, and SMI (all P<0.001). Moreover, they displayed weaker grip strength, slower gait speed, and lower SPPB scores in comparison to controls without sarcopenia (P=0.004 to 0.048). While participants with sarcopenia had longer chair standing times than those without sarcopenia, this variance was not statistically meaningful. The normal distribution of plasma SDF-1 levels is depicted in Supplemental Fig. S1 as a histogram, and this pattern was validated by the Kolmogorov-Smirnov test (P=0.939). The median plasma SDF-1 level for the entire participant group was 1,622.2 pg/mL (interquartile range, 1,321.2 to 1,849.9), and the minimum and maximum values were 555.0 and 2,942.7 pg/mL, respectively.
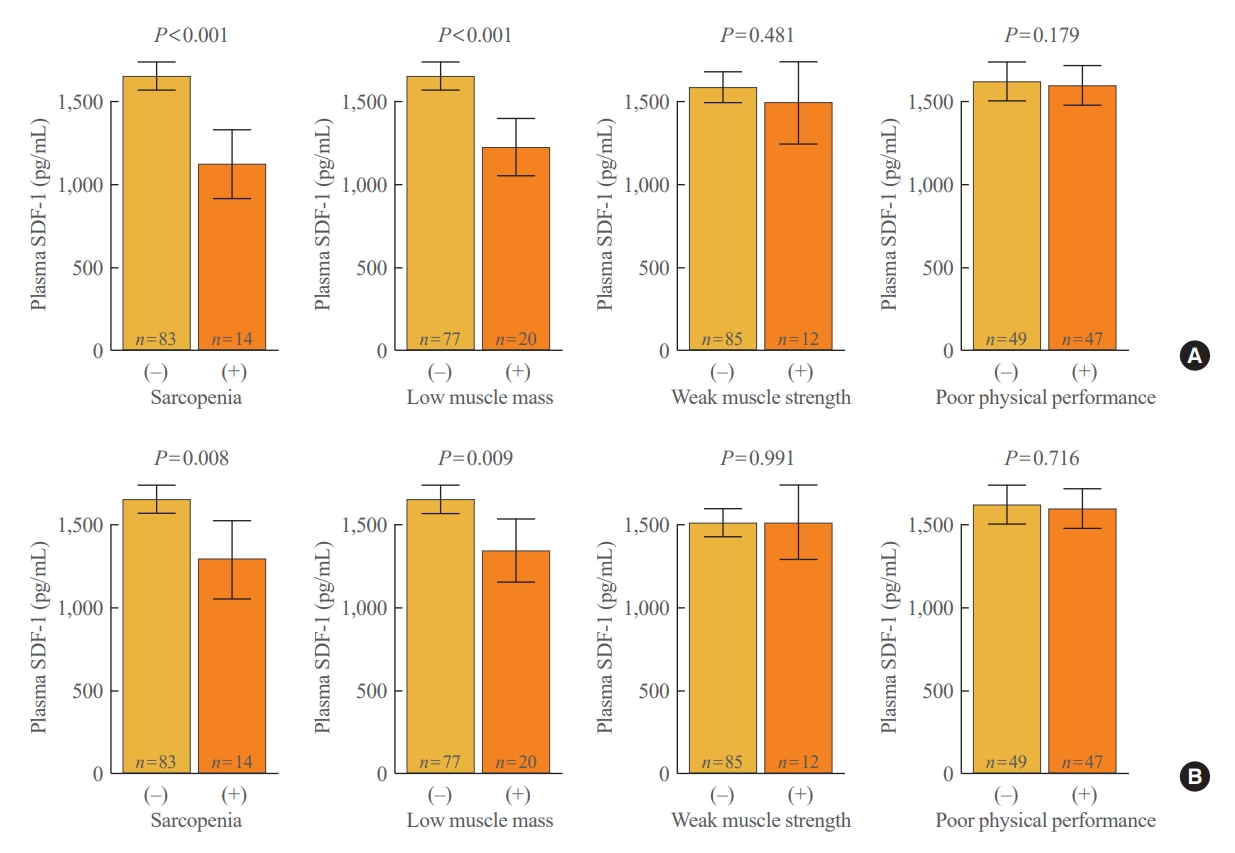
- A total of 97 participants were categorized based on the presence of sarcopenia, low muscle mass, weak muscle strength, poor physical performance, and their circulating SDF-1 concentrations were compared with those of each respective control group by ANCOVA (Fig. 1). In the crude analyses, participants with sarcopenia and low muscle mass exhibited plasma SDF-1 levels that were 32.1% and 26.0% lower than those without these conditions, respectively (both P<0.001). Moreover, these differences remained statistically significant after adjustment for sex, age, and BMI (P=0.008 and P=0.009, respectively). However, there were no significant differences in plasma SDF-1 levels observed between participants with or without weak muscle strength and poor physical performance, regardless of adjustment models.
- The Pearson and partial correlation analyses were conducted to explore the relationship of plasma SDF-1 levels with specific muscle parameters relevant to sarcopenia (Table 2). Higher plasma SDF-1 levels exhibited a significant correlation with higher SMI and gait speed, both before and after accounting for sex, age, and BMI (P<0.001 to 0.043). However, the positive association between circulating SDF-1 concentrations and grip strength was only evident in the univariate model (P=0.012), with statistical significance diminishing upon adjustment for potential confounding factors. Furthermore, no significant correlation was found between plasma SDF-1 levels and chair standing times across any of the adjustment models.
- Logistic regression analyses were performed to investigate the risk of sarcopenia and adverse muscle outcomes based on plasma SDF-1 levels (Table 3). Prior to accounting for confounding variables, the ORs for sarcopenia and low muscle mass per SD increase in plasma SDF-1 level were 0.23 and 0.31, respectively (both P<0.001). In addition, the reduced risk of these conditions with elevated plasma SDF-1 levels was still significant even after adjustment for sex, age, and BMI (P=0.045 and P=0.030, respectively). Conversely, the ORs for plasma SDF-1 levels on weak muscle strength and poor physical performance were insignificant in either unadjusted or adjusted analyses.
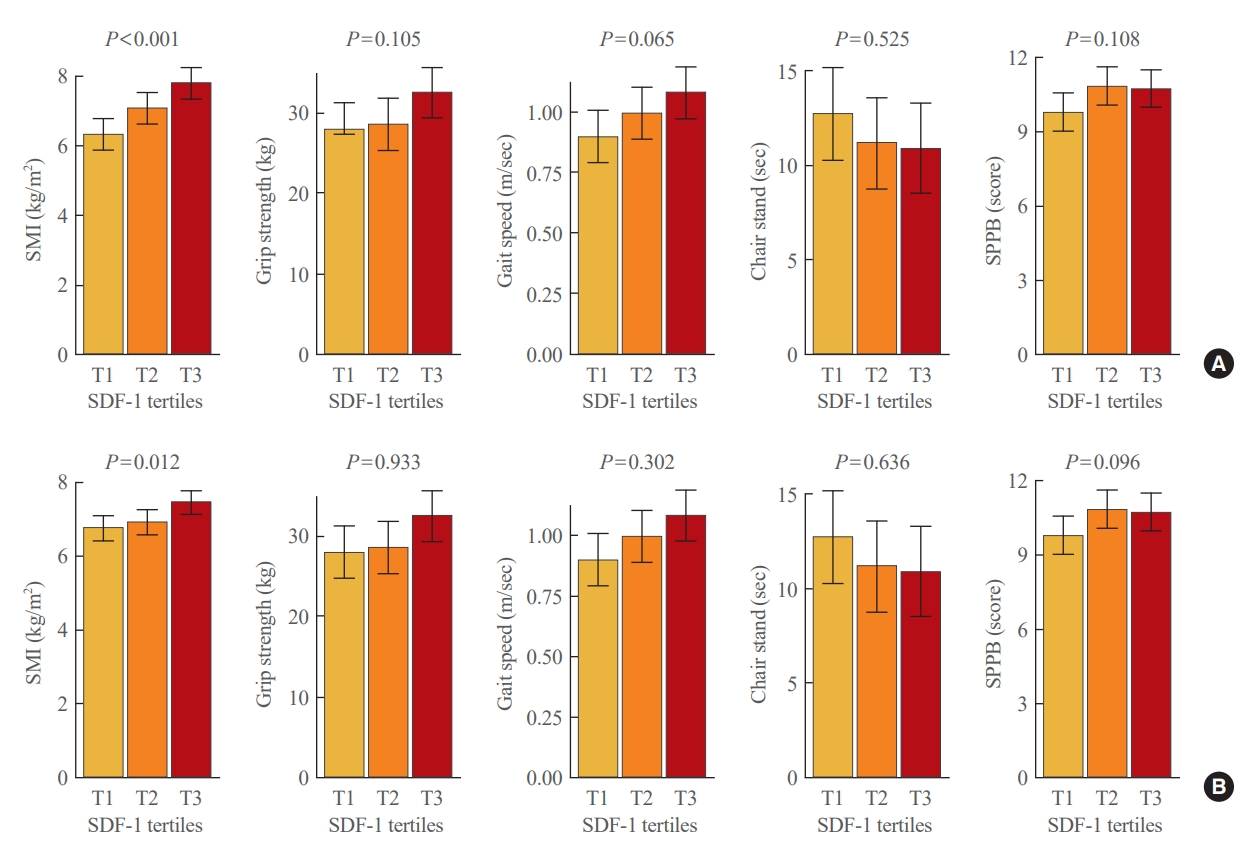
- In order to ascertain whether there is a threshold effect rather than a gradual effect in the link between circulating SDF-1 concentrations and muscle phenotypes, the participants were divided into three groups according to their plasma SDF-1 levels (Fig. 2). Participants in the highest SDF-1 tertile (T3) exhibited 23.2% and 10.2% higher SMI compared to those in the lowest tertile (T1), both before and after adjustment for sex, age, and BMI, respectively (P<0.001 and P=0.012, respectively). On the other hand, no significant differences in grip strength, gait speed, chair standing times, or SPPB scores were observed among the tertile groups in any of the adjustment models.
- When testing whether any association between circulating SDF-1 levels and muscle phenotypes could be influenced by sex, we found no evidence of interactions between plasma SDF-1 levels (expressed as a continuous variable) and sex (coded as 0 and 1 for men and women, respectively, and treated as categorical variables) in relation to their impact on muscle mass, strength, and physical performance (P for tests of interaction=0.876 to 0.939). As a result, we did not separate men and women, and refrained from conducting further analyses based on sex.
RESULTS
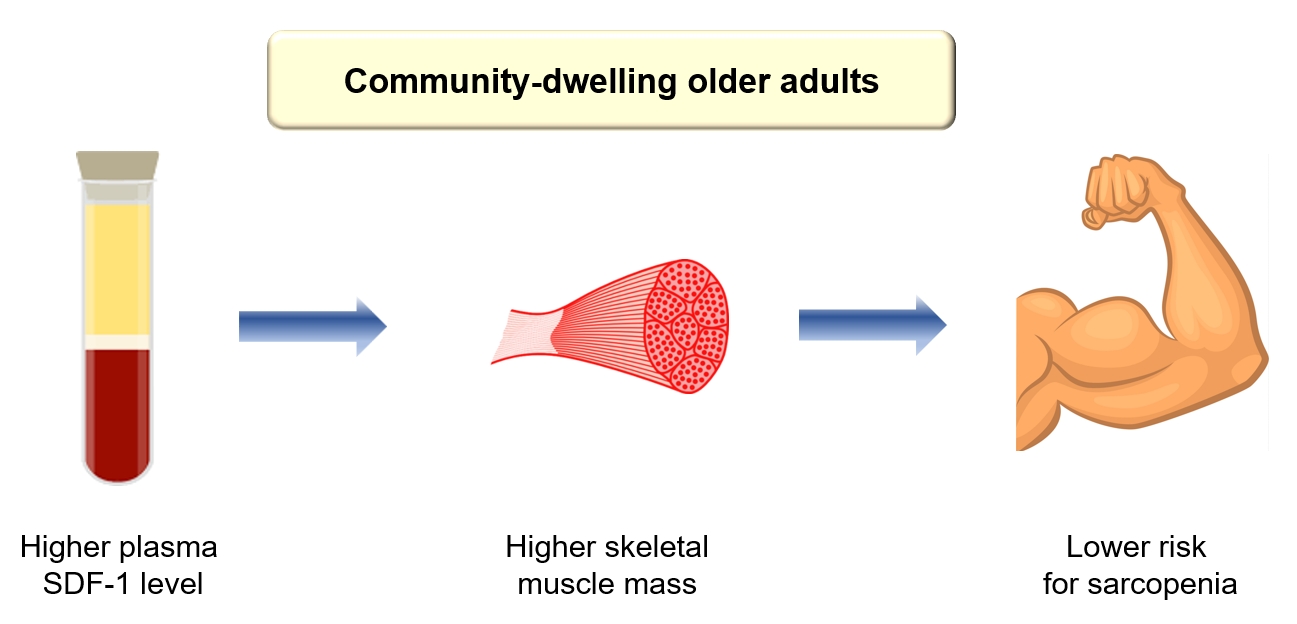
- In this cross-sectional study involving older adults, participants with sarcopenia exhibited 21.8% lower levels of circulating SDF-1 compared to those without sarcopenia, and the risk of sarcopenia decreased by 58% per SD increase in plasma SDF-1 levels after adjusting for sex, age, and BMI. Among the various muscle phenotypes examined, plasma SDF-1 concentrations showed a significant association with muscle mass rather than muscle strength and physical performance. This study represents the first investigation in humans to determine a difference in plasma SDF-1 levels based on sarcopenia status, and is clinically meaningful in that it supports the potential role of circulating SDF-1 as a biomarker for human muscle health.
- SDF-1 is a chemokine protein that plays various roles in angiogenesis, hematopoiesis, neurogenesis, inflammation, cancer metastasis, and human immunodeficiency virus infection [10-13]. Many lines of evidence from in vitro and animal experiments indicate that SDF-1 could be implicated in muscle homeostasis as well. Upregulation of SDF-1 within muscles experiencing cachexia led to a 20% augmentation in fiber area, affording protection against deterioration [24]. Likewise, myotubes undergoing atrophy and treated with SDF-1 exhibited higher overall protein content, stemming from diminished degradation of long-lived proteins [24]. This particular chemokine also had a beneficial effect on muscle regeneration by stimulating the migration of myoblasts through mechanisms reliant on CXCR4 [17]. Moreover, Bobadilla et al. [19] demonstrated that treatment with recombinant SDF-1 enhanced the recovery of injured muscles, while the inhibition of SDF-1 or CXCR4 hindered muscle regeneration. Taken together, these findings provide compelling evidence of the advantageous contribution of SDF-1 to postnatal myogenesis. The current study, which demonstrates reduced risks of sarcopenia in older adults with elevated plasma SDF-1 levels, holds significant clinical implications. It not only corroborates earlier experimental discoveries highlighting the muscle anabolic effects of SDF-1 but also establishes a foundation that supports the potential of SDF-1 as a viable target molecule for the diagnosis and treatment of sarcopenia.
- A particularly interesting finding in our study is that plasma SDF-1 level was mainly related to muscle mass and has little correlation with muscle strength and physical performance among the diagnostic criteria for sarcopenia. In fact, muscle mass, muscle strength, and physical performance each provide distinct insights into different facets of muscle health. Muscle mass signifies the quantity of muscular tissue present, reflecting structural composition [25]. Muscle strength represents the force-generating capacity of muscles, indicating their functional prowess [26]. Meanwhile, physical performance encompasses a range of abilities such as coordination, endurance, and power, reflecting the integrated efficiency of muscle groups during activities [27]. The diverse nature of these parameters highlights that the effects on muscle mass might not inherently extend to influencing muscle strength or physical performance. Therefore, it is plausible that SDF-1 might predominantly impact a specific aspect of muscle health while maintaining no discernible connection to others, and this observation underscores the complexity and multifaceted nature of muscular physiology.
- While this study possesses several strengths, such as the integration of various indicators of muscle health, it is crucial to acknowledge its limitations. Most importantly, as a cross-sectional study, it cannot establish causal relationships. Secondly, the inclusion of participants from a single medical center may constrain the generalizability of our findings. Hence, future investigations should encompass multiple centers. Thirdly, the utilization of bioelectrical impedance analysis represents an indirect approach to assess muscle mass, potentially affecting measurement accuracy [28]. Therefore, it is imperative to employ alternative methods, such as the D3-creatine dilution technique, which offers enhanced precision in measuring muscle mass. Lastly, while we made efforts to account for several confounding factors in our analyses, we cannot dismiss the possibility that the observed association could have arisen due to uncontrolled factors influencing SDF-1 and/or muscle variables, such as cancers, cardiovascular diseases, and a history of fractures.
- In conclusion, elevated plasma SDF-1levels exhibited a significant correlation with a reduced risk of sarcopenia and lower muscle mass among older adults, even after adjustment for confounding variables. These findings imply that the measurement of plasma SDF-1 levels could offer supplementary insights for evaluating muscle health in the elderly population. To ascertain the precise role of circulating SDF-1 levels as a blood-based biomarker for sarcopenia, further meticulously designed large-scale longitudinal studies are imperative.
DISCUSSION
Supplementary Material
Supplemental Fig. S1.
-
CONFLICTS OF INTEREST
No potential conflict of interest relevant to this article was reported.
-
AUTHOR CONTRIBUTIONS
Conception or design: I.Y.J., E.L., J.Y.B., B.J.K. Acquisition, analysis, or interpretation of data: S.J., K.K., S.J.P., J.Y.L., H. W.J., H.J.Y., I.Y.J., B.J.K. Drafting the work or revising: S.J., K.K., I.Y.J., E.L., J.Y.B., B.J.K. Final approval of the manuscript: I.Y.J., E.L., J.Y.B., B.J.K.
Article information
-
Acknowledgements
- This study was supported by grants from the Korean Endocrine Society for Convergence Research Award 2021 and the Asan Institute for Life Science, Asan Medical Center, Seoul, South Korea (grant number: 2017IL0659).
| Variable | Sarcopenia (n=14) | No sarcopenia (n=83) | P value |
|---|---|---|---|
| Sex | 0.084 | ||
| Female | 10 (71.4) | 37 (44.6) | |
| Male | 4 (28.6) | 46 (55.4) | |
| Age, yr | 72.5±5.1 | 69.0±6.3 | 0.051 |
| Weight, kg | 56.1±6.7a | 69.7±9.8a | <0.001a |
| Height, cm | 155.5±2.8a | 161.1±9.4a | <0.001a |
| BMI, kg/m2 | 23.2±2.9a | 26.8±2.8a | <0.001a |
| ASM, kg | 13.2±1.4a | 19.4±5.1a | <0.001a |
| SMI, kg/m2 | 5.44±0.52a | 7.35±1.33a | <0.001a |
| Grip strength, kg | 23.5±6.5a | 30.8±9.4a | 0.006a |
| Gait speed, m/sec | 0.78±0.29a | 1.03±0.30a | 0.004a |
| Chair stand, sec | 16.1±13.9 | 10.8±4.5 | 0.176 |
| SPPB score (range, 0–12) | 9.1±3.0a | 10.8±1.6a | 0.048a |
Values are expressed as number (%) or mean±standard deviation. The Student’s t test was employed to compare continuous variables between the two groups, while the chi-square test was used for categorical variables.
BMI, body mass index; ASM, appendicular skeletal muscle mass; SMI, skeletal muscle mass index; SPPB, short physical performance battery.
a Statistically significant values.
| Variable |
Unadjusted |
Sex, age, and BMI adjusted |
||
|---|---|---|---|---|
| γa | P value | γb | P value | |
| SMI | 0.446c | <0.001c | 0.210c | 0.043c |
| Grip strength | 0.253c | 0.012c | 0.028 | 0.787 |
| Gait speed | 0.301c | 0.003c | 0.210c | 0.043c |
| Chair stand | –0.124 | 0.227 | –0.039 | 0.713 |
The analysis did not include the short physical performance battery score due to its non-normal distribution.
SDF-1, stromal cell-derived factor 1; BMI, body mass index; SMI, skeletal muscle mass index.
a Pearson’s correlation coefficient;
b Partial correlation coefficient;
c Statistically significant values.
| Variable |
Unadjusted |
Sex, age, and BMI adjusted |
||
|---|---|---|---|---|
| OR (95% CI) per SD increment in plasma SDF-1 | P value | OR (95% CI) per SD increment in plasma SDF-1 | P value | |
| Sarcopenia | 0.23 (0.11–0.49)a | <0.001a | 0.42 (0.18–0.98)a | 0.045a |
| Low muscle mass | 0.31 (0.17–0.57)a | <0.001a | 0.45 (0.22–0.93)a | 0.030a |
| Weak muscle strength | 0.80 (0.44–1.47) | 0.477 | 1.01 (0.40–2.04) | 0.981 |
| Poor physical performance | 0.75 (0.50–1.14) | 0.180 | 0.91 (0.57–1.47) | 0.706 |
- 1. Baek KW, Jung YK, Park JS, Kim JS, Hah YS, Kim SJ, et al. Two types of mouse models for sarcopenia research: senescence acceleration and genetic modification models. J Bone Metab 2021;28:179–91.ArticlePubMedPMCPDF
- 2. Baek JY, Jung HW, Kim KM, Kim M, Park CY, Lee KP, et al. Korean Working Group on Sarcopenia guideline: expert consensus on sarcopenia screening and diagnosis by the Korean Society of Sarcopenia, the Korean Society for Bone and Mineral Research, and the Korean Geriatrics Society. Ann Geriatr Med Res 2023;27:9–21.ArticlePubMedPMCPDF
- 3. Lee JY, Kim DA, Choi E, Lee YS, Park SJ, Kim BJ. Aldosterone inhibits in vitro myogenesis by increasing intracellular oxidative stress via mineralocorticoid receptor. Endocrinol Metab (Seoul) 2021;36:865–74.ArticlePubMedPMCPDF
- 4. Cruz-Jentoft AJ, Sayer AA. Sarcopenia. Lancet 2019;393:2636–46.ArticlePubMed
- 5. Dao T, Green AE, Kim YA, Bae SJ, Ha KT, Gariani K, et al. Sarcopenia and muscle aging: a brief overview. Endocrinol Metab (Seoul) 2020;35:716–32.ArticlePubMedPMCPDF
- 6. Cruz-Jentoft AJ, Bahat G, Bauer J, Boirie Y, Bruyere O, Cederholm T, et al. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing 2019;48:16–31.ArticlePubMedPMCPDF
- 7. Visser M, Schaap LA. Consequences of sarcopenia. Clin Geriatr Med 2011;27:387–99.ArticlePubMed
- 8. Sadri F, Rezaei Z, Fereidouni M. The significance of the SDF-1/CXCR4 signaling pathway in the normal development. Mol Biol Rep 2022;49:3307–20.ArticlePubMedPDF
- 9. Schrader AJ, Lechner O, Templin M, Dittmar KE, Machtens S, Mengel M, et al. CXCR4/CXCL12 expression and signalling in kidney cancer. Br J Cancer 2002;86:1250–6.ArticlePubMedPMCPDF
- 10. Bleul CC, Fuhlbrigge RC, Casasnovas JM, Aiuti A, Springer TA. A highly efficacious lymphocyte chemoattractant, stromal cell-derived factor 1 (SDF-1). J Exp Med 1996;184:1101–9.ArticlePubMedPMCPDF
- 11. Lau TT, Wang DA. Stromal cell-derived factor-1 (SDF-1): homing factor for engineered regenerative medicine. Expert Opin Biol Ther 2011;11:189–97.ArticlePubMed
- 12. Shiba Y, Takahashi M, Yoshioka T, Yajima N, Morimoto H, Izawa A, et al. M-CSF accelerates neointimal formation in the early phase after vascular injury in mice: the critical role of the SDF-1-CXCR4 system. Arterioscler Thromb Vasc Biol 2007;27:283–9.PubMed
- 13. Psenak O. Stromal cell-derived factor 1 (SDF-1): its structure and function. Cas Lek Cesk 2001;140:355–63.PubMed
- 14. Vasyutina E, Stebler J, Brand-Saberi B, Schulz S, Raz E, Birchmeier C. CXCR4 and Gab1 cooperate to control the development of migrating muscle progenitor cells. Genes Dev 2005;19:2187–98.ArticlePubMedPMC
- 15. Hunger C, Odemis V, Engele J. Expression and function of the SDF-1 chemokine receptors CXCR4 and CXCR7 during mouse limb muscle development and regeneration. Exp Cell Res 2012;318:2178–90.ArticlePubMed
- 16. Melchionna R, Di Carlo A, De Mori R, Cappuzzello C, Barberi L, Musaro A, et al. Induction of myogenic differentiation by SDF-1 via CXCR4 and CXCR7 receptors. Muscle Nerve 2010;41:828–35.ArticlePubMedPDF
- 17. Brzoska E, Kowalewska M, Markowska-Zagrajek A, Kowalski K, Archacka K, Zimowska M, et al. Sdf-1 (CXCL12) improves skeletal muscle regeneration via the mobilisation of Cxcr4 and CD34 expressing cells. Biol Cell 2012;104:722–37.ArticlePubMed
- 18. Brzoska E, Kowalski K, Markowska-Zagrajek A, Kowalewska M, Archacki R, Plaskota I, et al. Sdf-1 (CXCL12) induces CD9 expression in stem cells engaged in muscle regeneration. Stem Cell Res Ther 2015;6:46.ArticlePubMedPMCPDF
- 19. Bobadilla M, Sainz N, Abizanda G, Orbe J, Rodriguez JA, Paramo JA, et al. The CXCR4/SDF1 axis improves muscle regeneration through MMP-10 activity. Stem Cells Dev 2014;23:1417–27.ArticlePubMedPMC
- 20. Roberts HC, Denison HJ, Martin HJ, Patel HP, Syddall H, Cooper C, et al. A review of the measurement of grip strength in clinical and epidemiological studies: towards a standardised approach. Age Ageing 2011;40:423–9.ArticlePubMed
- 21. Peel NM, Kuys SS, Klein K. Gait speed as a measure in geriatric assessment in clinical settings: a systematic review. J Gerontol A Biol Sci Med Sci 2013;68:39–46.ArticlePubMed
- 22. Jung HW, Roh H, Cho Y, Jeong J, Shin YS, Lim JY, et al. Validation of a multi-sensor-based kiosk for short physical performance battery. J Am Geriatr Soc 2019;67:2605–9.ArticlePubMedPDF
- 23. Chen LK, Woo J, Assantachai P, Auyeung TW, Chou MY, Iijima K, et al. Asian Working Group for Sarcopenia: 2019 consensus update on sarcopenia diagnosis and treatment. J Am Med Dir Assoc 2020;21:300–7.PubMed
- 24. Martinelli GB, Olivari D, Re Cecconi AD, Talamini L, Ottoboni L, Lecker SH, et al. Activation of the SDF1/CXCR4 pathway retards muscle atrophy during cancer cachexia. Oncogene 2016;35:6212–22.ArticlePubMedPDF
- 25. Tosato M, Marzetti E, Cesari M, Savera G, Miller RR, Bernabei R, et al. Measurement of muscle mass in sarcopenia: from imaging to biochemical markers. Aging Clin Exp Res 2017;29:19–27.ArticlePubMedPDF
- 26. Bhasin S, Travison TG, Manini TM, Patel S, Pencina KM, Fielding RA, et al. Sarcopenia definition: the position statements of the sarcopenia definition and outcomes consortium. J Am Geriatr Soc 2020;68:1410–8.ArticlePubMedPDF
- 27. Meza-Valderrama D, Marco E, Davalos-Yerovi V, Muns MD, Tejero-Sanchez M, Duarte E, et al. Sarcopenia, malnutrition, and cachexia: adapting definitions and terminology of nutritional disorders in older people with cancer. Nutrients 2021;13:761.ArticlePubMedPMC
- 28. Aleixo GF, Shachar SS, Nyrop KA, Muss HB, Battaglini CL, Williams GR. Bioelectrical impedance analysis for the assessment of sarcopenia in patients with cancer: a systematic review. Oncologist 2020;25:170–82.ArticlePubMedPMCPDF
References
Figure & Data
References
Citations


 KES
KES
 PubReader
PubReader ePub Link
ePub Link Cite
Cite