Articles
- Page Path
- HOME > Endocrinol Metab > Volume 32(1); 2017 > Article
-
Review ArticleThe Implication of Coronary Artery Calcium Testing for Cardiovascular Disease Prevention and Diabetes
- Ron Blankstein1,2, Ankur Gupta1,2, Jamal S. Rana3,4, Khurram Nasir5,6
-
Endocrinology and Metabolism 2017;32(1):47-57.
DOI: https://doi.org/10.3803/EnM.2017.32.1.47
Published online: March 20, 2017
1Division of Cardiovascular Medicine, Department of Medicine, Brigham and Women's Hospital, Harvard Medical School, Boston, MA, USA.
2Department of Radiology, Brigham and Women's Hospital, Harvard Medical School, Boston, MA, USA.
3Division of Cardiology, Kaiser Permanente Northern California, Oakland, CA, USA.
4Division of Research, Kaiser Permanente Northern California, Oakland, CA, USA.
5Department of Medicine, Herbert Wertheim College of Medicine, Florida International University, Miami, FL, USA.
6Miami Cardiac & Vascular Institute, Baptist Health South Florida, Miami, FL, USA.
- Corresponding author: Ron Blankstein. Division of Cardiovascular Medicine, Department of Medicine and Department of Radiology, Brigham and Women's Hospital, 75 Francis St, Boston, MA 02115, USA. Tel: +1-857-307-1989, Fax: +1-857-307-1955, rblankstein@partners.org
Copyright © 2017 Korean Endocrine Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- ABSTRACT
- WHAT IS CORONARY ARTERY CALCIUM?
- PROGNOSTIC VALUE OF THE PRESENCE AND ABSENCE OF CORONARY ARTERY CALCIUM
- WHEN IS CAC TESTING MOST HELPFUL?
- USE OF CAC IN LIGHT OF RECENT GUIDELINES?
- WHEN IS CAC TESTING NOT HELPFUL?
- WHAT ARE THE LIMITATIONS OF CAC TESTING?
- WHAT SHOULD BE DONE WHEN CAC IS ELEVATED?
- WHAT PREDICTS THE DEVELOPMENT OF CAC LATER IN LIFE?
- DOES CAC TESTING IMPROVE OUTCOMES?
- WHAT ARE THE IMPLICATION OF CAC TESTING BEYOND CHD?
- THE ROLE OF CAC IN INDIVIDUALS WHO HAVE DIABETES
- FUTURE ROLE OF CAC BEYOND STATINS
- CONCLUSIONS
- Article information
- References
ABSTRACT
- Over the last two decades coronary artery calcium (CAC) scanning has emerged as a quick, safe, and inexpensive method to detect the presence of coronary atherosclerosis. Data from multiple studies has shown that compared to individuals who do not have any coronary calcifications, those with severe calcifications (i.e., CAC score >300) have a 10-fold increase in their risk of coronary heart disease events and cardiovascular disease. Conversely, those that have a CAC of 0 have a very low event rate (~0.1%/year), with data that now extends to 15 years in some studies. Thus, the most notable implication of identifying CAC in individuals who do not have known cardiovascular disease is that it allows targeting of more aggressive therapies to those who have the highest risk of having future events. Such identification of risk is especially important for individuals who are not on any therapies for coronary heart disease, or when intensification of treatment is being considered but has an uncertain role. This review will highlight some of the recent data on CAC testing, while focusing on the implications of those findings on patient management. The evolving role of CAC in patients with diabetes will also be highlighted.
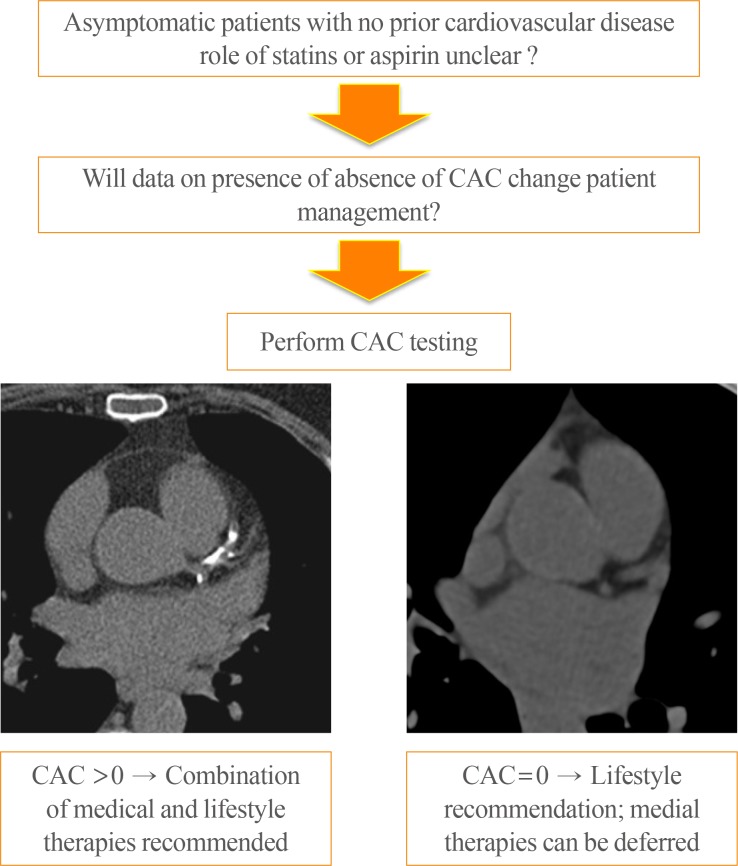
- As opposed to risk factors which tell us the risk someone might have of having disease, the detection of calcium in the coronary arteries shows us actual evidence of the disease that we are interested in treating or preventing. Thus, individuals who have coronary artery calcifications do, in fact, have coronary artery disease. However, such disease is often termed as “subclinical” as it is most often detected in asymptomatic individuals and is not associated with any clinical symptoms (Fig. 1).
WHAT IS CORONARY ARTERY CALCIUM?
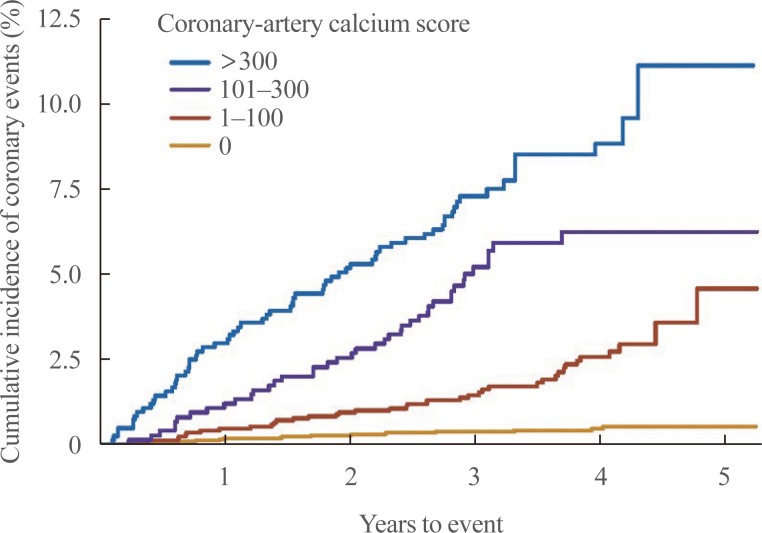
- There is substantial evidence to support the fact that among individuals who do not have known cardiovascular disease (CVD), the presence and severity of coronary artery calcium (CAC) provides the strongest measure of future cardiovascular risk. When compared to individuals who do not have any CAC, those with severe calcifications (i.e., CAC score >300) have a 10-fold increase in their risk of coronary heart disease (CHD) and CVD events (Fig. 2) [1].
- CAC scoring can independently predict cardiovascular events, and when compared to traditional risk factors, offers improved discrimination and reclassification [2345]. Indeed, several large and well-conducted observational studies have demonstrated that when added to standard risk prediction models, CAC has a significantly greater improvement in risk prediction compared with other novel biomarkers or a combined biomarker panel [678]. Specifically, among intermediate risk patients, the addition of CAC to the Framingham risk score has been found to have a high net reclassification improvement (66%), with most other novel risk markers—such as carotid intima-media thickness, brachial flow mediated dilation, ankle brachial index, and high-sensitivity C-reactive protein—having a net reclassification improvement index of <10%.
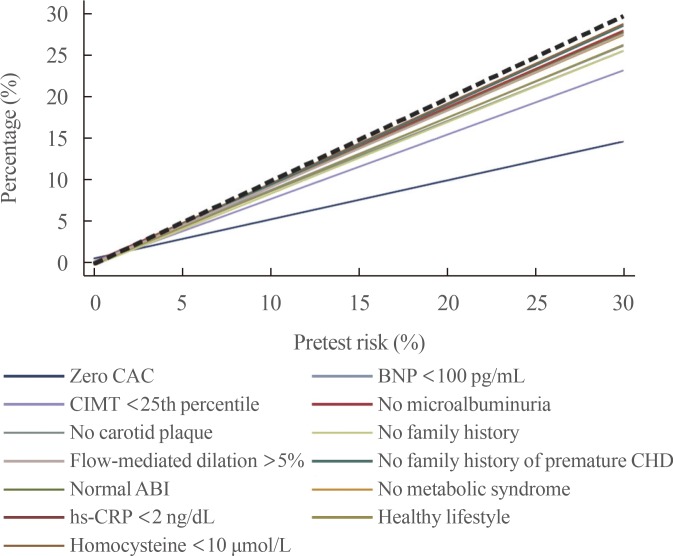
- Importantly, the superior predictive capabilities of CAC scoring are due to its ability to correctly reclassify patients to both high and low risk categories. In particular, individuals who do not have any coronary artery calcifications (CAC=0) have an extremely low risk of cardiovascular events of approximately 0.1% per year [9]. The ability of CAC to as a “negative risk marker” has been compared to multiple other risk factors among participants in the Multi-Ethnic Study of Atherosclerosis (MESA) [10]. In this study a CAC of 0 was found to result in the greatest downward shift in cardiovascular risk (Fig. 3).
- Much attention has been focused on the utility of CAC scoring to enhance risk prediction for individuals at intermediate risk. However, it is important to note that CAC has significant prognostic value across a wide spectrum of age and risk factor profiles [411]. For instance, a prior study by Blankstein et al. [3] examined individuals from MESA with low-density lipoprotein (LDL) cholesterol ≤130 mg/dL, and showed that the presence or absence of CAC was a strong predictor of incident CHD and CVD events, regardless of whether other risk factors were present or absent (Fig. 4). Among individuals traditionally classified as low risk, either based on risk factor burden or calculated risk score, a high CAC score (≥100) is associated with an estimated 10-year all CHD event rate of nearly 10%. In contrast, among individuals traditionally identified as high risk by risk factor burden or by the Framingham risk score, a CAC score of 0 is associated with a remarkably low 10-year all CHD event rate of roughly 3%. In fact, individuals with no risk factors and an elevated CAC score have nearly three times the event rate of those individuals with multiple risk factors and a CAC score of 0 [4].
PROGNOSTIC VALUE OF THE PRESENCE AND ABSENCE OF CORONARY ARTERY CALCIUM
- While many potential indications exist, in our experience the following groups of patients commonly benefit from statin therapy.
-
(1) Statin candidates averse to treatment: patients who are advised, by guidelines, to be on statin therapy (atherosclerotic cardiovascular disease [ASCVD] risk score >5% per the pooled cohort equation recommended by the American College of Cardiology/American Heart Association [ACC/AHA]) but who prefer to avoid such therapy.
(2) Statin intolerant patients: several studies have suggested that many individuals who previously were deemed as statin intolerant may be able to tolerate statins when re-challenged. While many such individuals prefer not to be on statins, the identification of coronary plaque may serve as a signal to reconsider a statin, recognizing the greater benefits of such therapies in patients who have higher risk.
(3) Patients with premature family history of CHD: such individuals may benefit from more personalized risk assessment since most traditional risk equations do not include family history of premature CHD. Importantly, even when a strong family history is present, the actual burden of CAC—and thus a person's risk—may be highly variable [12].
WHEN IS CAC TESTING MOST HELPFUL?
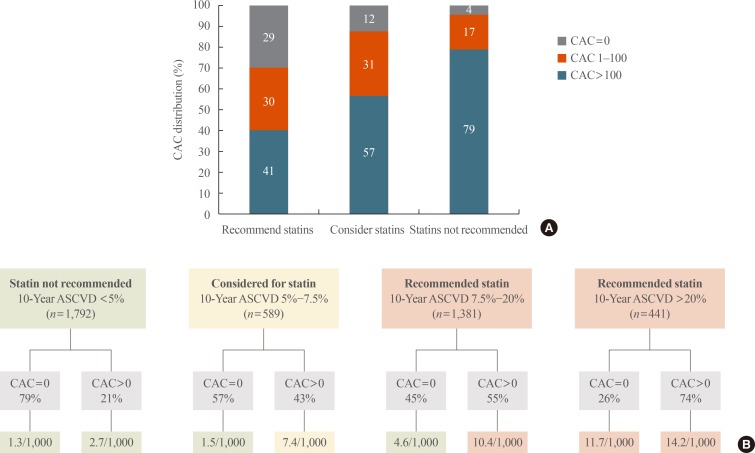
- Recent data has evaluated how CAC testing may complement the recent recommendations made by the European Society of Cardiology (ESC) and the AHA/ACC guidelines for use of statin therapy in primary prevention of CVD. Nasir et al. [13] evaluated how the absence of CAC can be used to downward reclassify individuals who are recommended for statin therapy by the AHA/ACC guidelines to a lower risk group who may defer such therapy. They applied the AHA/ACC guidelines to 4,758 participants of the MESA study, of which 50% were recommended by guidelines to be treated with moderate to high intensity statins. Among those individuals recommended for statin therapy, 41% had no CAC (Fig. 5A). Importantly, in the absence of CAC, those individuals who had a baseline 10-year ASCVD risk score between 7.5% to 20% had an observed 10-year risk of 4.6%, which is lower than the 5% threshold used by the guidelines for considering or recommending statins. On the other hand, among individuals who at baseline had an ASCVD risk of >20% and CAC=0, a high risk was observed (11.7%), while those who had a baseline ASCVD risk below <5% had a low event rate irrespective of whether CAC was present or absent (Fig. 5B). Based on these results, the role of CAC testing seems greatest among individuals who are considered or recommended statins, and have an ASCVD risk of 5% to 20%. In such scenarios, the absence of CAC can be used to identify a low risk group in which statin therapy may be deferred, particularly if such an option is preferred by patients and/or physicians based on evaluating the risk/benefit of treatment in the setting of low risk, while also incorporating patient values and preferences.
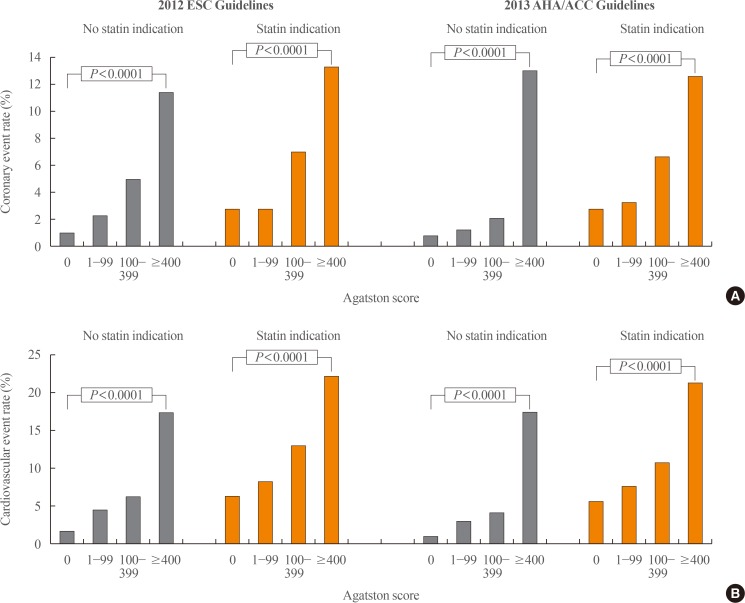
- A similar analysis was recently preformed by Mahabadi et al. [14], who applied both AHA/ACC guidelines and the ESC guidelines to participants of the Heinz Nixdorf Recall study. Similar to MESA, this study is a prospective cohort of individuals who did not have any known CVD at baseline. This study found that for both guidelines, and irrespective of statin indication, individuals with higher CAC scores had a higher CHD and CVD event rate (Fig. 6) [14]. Notably, CAC was absent in 43% of individuals who were met statin indications by the ESC guidelines and 53% of the individuals who met statin indication by the AHA/ACC guidelines.
USE OF CAC IN LIGHT OF RECENT GUIDELINES?
- Individuals who have known CHD, or who are already on aggressive medical therapy to prevent CVD, are unlikely to benefit from CAC testing. Such individuals are unlikely to have any meaningful changes in their management as a result of CAC testing. In addition, patients who are averse to treatment, and who are unlikely to initiate treatment even if CAC is identified, should not undergo CAC testing.
WHEN IS CAC TESTING NOT HELPFUL?
- CAC scanning is associated with a low level of radiation, estimated at 1 to 2 mSV, which is equivalent to the dose of a mammogram [15]. CAC scanning cannot be used to follow treatment response, as CAC does not regress, and may even progress with statin therapy. When CAC testing is performed, incidental findings (e.g., lung nodules) may be found, and these can result in additional downstream costs when further investigations are required.
WHAT ARE THE LIMITATIONS OF CAC TESTING?
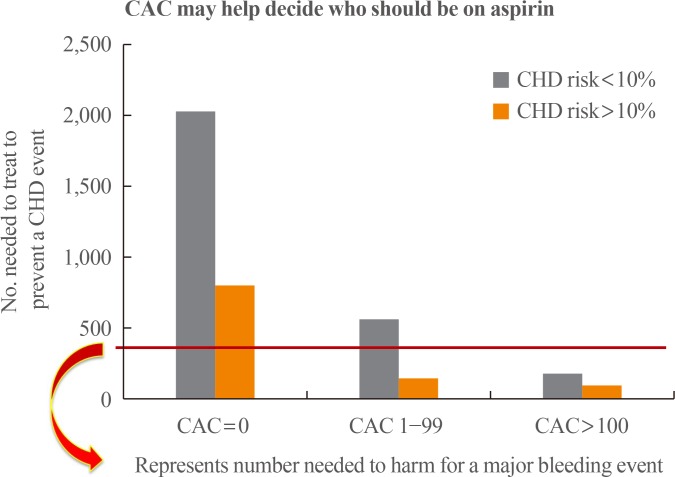
- Table 1 lists some of the recommended interventions to consider for individuals who are found to have elevated CAC. In addition to initiation of statin therapy, individuals with CAC >100 are also likely to benefit from aspirin therapy (Fig. 7) [16]. Information on CAC burden, when combined with other clinical risk factors, may also inform how aggressive to treat individuals with pre-hypertension or hypertension [17].
WHAT SHOULD BE DONE WHEN CAC IS ELEVATED?
- The Coronary Artery Risk Development in Young Adults (CARDIA) study examined the association of various risk factors in young adults with incident CAC 15 to 25 years later. This study found that the levels of modifiable risk factors in young adults predicted incident CAC in middle age. Similarly, longer duration of overall obesity (e.g., body mass index ≥30) or abdominal obesity was associated with greater CAC progression later in life [18]. While these findings support the fact that the development of atherosclerosis if often preceded by a constellation of unfavorable risk factors, there is no model which reliably predicts the development or absence of CAC and it has been established that even among individuals who do not have any traditional risk factors, the presence of CAC signified elevated risk of both CHD and CVD events [419].
- A recent important finding from the CARDIA study is that the presence of any CAC in early adult life (i.e., CAC >0), even after accounting for other risk factors, indicates a higher risk of having a future CHD or CVD event during the next decade [20]. Since the presence of CAC in young adults aged 32 to 46 was relatively rare (~10%), widescreen unselected CAC testing in those less than 45 is not recommended. However, future studies are needed to identify which young individuals are most likely to benefit from CAC screening. However, the fact that CAC prevalence significantly increases in the third to fifth decade of life suggests that this is a particularly important time period to implement preventive measures [21].
WHAT PREDICTS THE DEVELOPMENT OF CAC LATER IN LIFE?
- Some have proposed a large prospective randomized controlled trial (RCT) showing the impact of CAC testing on patient outcomes. Such a study is unlikely to occur in the current era where statins are increasingly being used for prevention. The challenges with a CAC RCT trial are also inherent in the fact that it would be unethical to randomized patients that have severe amount of CAC into an arm of no therapy. It is also noteworthy, that such trial data is also not available for any risk scores, which have been proposed for evaluating patients with suspected CAD.
- Nevertheless, there are a few insights that that were highlighted by prior studies. In the St. Francis Heart study, 1,005 asymptomatic, apparently healthy men and women age 50 to 70 years with CAC scores ≥80th percentile for age and gender were randomized to atorvastatin 20 mg daily in addition to vitamins C and E in a double-blind placebo-controlled trial [22]. After a mean follow-up of 4.3 years, treatment with atorvastatin reduced LDL cholesterol levels by nearly 40% and showed a trend towards reduction in atherosclerotic CVD events (6.9% vs. 9.9% in atorvastatin vs. placebo arms respectively, P=0.08). The effect of atorvastatin on CVD event reduction was stronger in the subgroup of patients with CAC >400 (8.7% vs. 15%, P=0.046). This study, while small when compared to contemporary trials, and clearly underpowered, provides an important signal that patients with increased CAC are most likely to benefit from cholesterol lowering therapies.
- When evaluating the impact of CAC testing on patient outcomes it is important to consider the various mechanisms by which CAC testing may improve outcomes, including both pharmacological and non-pharmacological interventions (Table 2). A recent meta-analysis by our group assessed the odds of initiation or continuation of pharmacological and lifestyle cardiovascular preventive therapies in patients with non-zero versus zero CAC score detected on cardiac computed tomography [23]. In this meta-analysis of six studies [242526272829] involving 11,256 participants in a mean follow-up of 1.6 to 6 years, we found that identifying calcified coronary plaque, significantly increased the likelihood of initiation of aspirin and blood pressure lowering medications, initiation and continuation of lipid lowering medications, as well as intensification of exercise and dietary changes. These findings remained significant after adjustment for baseline patient characteristics and cardiovascular risk factors in individual studies.
- To date, several RCTs have investigated the effect of CAC scan versus no scan on preventive pharmacotherapies in asymptomatic individuals [24303132]. O'Malley et al. [30] did not find the impact of CAC scan on change in 10-year Framingham risk score over 1 year among group of patients to whom CAC scan information was provided versus withheld. However, participants in this trial were asymptomatic active duty United States Army personnel, with very low cardiovascular risk at baseline (0 CAC score in 85.3% of patients), in whom an additional risk-reduction would be hard to achieve. Similarly, a small RCT of 56 postmenopausal women without known CAD did not show an independent benefit of CAC scanning on cardiovascular risk factor control that included systolic blood pressure and lipid profile (CAC <10 in 73.1% of 26 women who underwent CAC scan) [32]. It must be emphasized that the low risk populations in these studies are not fully representative of the population in which CAC scan is intended. Indeed, consistent evidence supports the concept that individuals who are at intermediate risk by traditional risk scores have the greatest potential for risk reclassification and modification of primary prevention therapies. By comparison, Rozanski et al. [24], in Early Identification of Subclinical Atherosclerosis by Noninvasive Imaging Research (EISNER) trial, performed a large, well-designed RCT of 2,137 middle-aged subjects with cardiovascular risk factors in whom a large proportion of the patients with CAC testing had a non-zero CAC score (n=680/1,311, 52%). In this study CAC scanning was associated with superior CAD risk factor control compared to usual care alone.
- Whelton et al. [33] conducted an updated meta-analysis of the four available RCTs and found a non-significant trend towards reduction in blood pressure, lipid levels, and smoking cessation among individuals who had a CAC scan compared to those who were managed by standard care. However, the EISNER trial [24] showed that within the CAC scan group, there was significant increase in aspirin, statin and blood pressure lowering medications in individuals with non-zero CAC. Therefore, abnormal CAC score, and not just merely the CAC scan itself, likely accounts for behavioral changes following CAC scan. A RCT by Mols et al. [34] further supports this view. They found that in stable chest pain patients with hyperlipidemia and no obstructive CAD who have CAC >70 on calcium scan, visualization of CAC, and brief recommendations about risk modification led to a favorable influence on plasma total cholesterol concentration and adherence to statin therapy.
DOES CAC TESTING IMPROVE OUTCOMES?
- Recent studies have shown that increased CAC is associated with higher rate of cerebrovascular accidents [3536], heart failure [3738], and atrial fibrillation [39]. In addition, compared to individuals who have no CAC, those with increased CAC are more likely to have other non-cardiac conditions such as cancer, chronic kidney disease, chronic obstructive pulmonary disease, and hip fractures [40].
WHAT ARE THE IMPLICATION OF CAC TESTING BEYOND CHD?
- For more than a decade, the presence of diabetes has been considered a CHD risk equivalent. However, several recent studies have shown that the CVD risk of patients with diabetes is markedly variable. A recent large, contemporary cohort showed that diabetes alone did not confer the same level of CHD risk as individuals with prior CHD. Compared to individuals without diabetes or CHD, the risk doubled among those with diabetes alone, but tripled among those with prior CHD alone. Importantly CHD event rates were very low among individuals with diabetes who were less than 40 years of age [41]. While these results suggest a potential role for further risk stratification among those with diabetes [42], prior trials using nuclear myocardial perfusion imaging [43] or coronary CT angiography [44] have not shown any improvement in patient outcomes associated with screening. In part these negative results were due to the fact that event rates in these studies were lower than predicted, and medical therapy was highly prevalent, regardless of whether screening was performed or not. Accordingly, while there may remain an unproven role for screening very high risk individuals with diabetes using functional testing, in younger and lower risk individuals the role of screening may ultimately be to identify which individuals—despite their diabetes—have a sufficiently low long-term risk that they can defer treatment with statins.
- Supporting the potential role of CAC screening in diabetes or metabolic syndrome, Malik et al. [45] evaluated the association of CAC with CHD events in 6,603 people aged 45 to 84 years without clinical CVD in the MESA and risk factor-adjusted hazard ratios for CHD for CAC 1 to 99 to ≥400 versus 0 in subjects with neither metabolic syndrome nor diabetes ranged from 2.6 to 9.5; in those with metabolic syndrome ranged from 3.9 to 11.9; and in those with diabetes ranged from 2.9 to 6.2 (all P<0.05). Patients with metabolic syndrome or diabetes and a CAC score of 0 had a projected annual CHD event rate of 0.4% and 0.8%, respectively, whereas those with CAC score of >400 had annual CHD event rate of 4.6% and 5.1%, respectively. Diabetes mellitus has traditionally been considered as a CHD risk equivalent. However, 45% of patients with metabolic syndrome and 38% patients with diabetes in this study had a CAC score of 0 with annual CHD event rate similar to those without these conditions (Fig. 8).
- These findings imply that CAC testing in these patients could help with risk-reclassification of a significant proportion of patients with diabetes or metabolic syndrome. Nevertheless, the use of routine CAC screening in individuals with diabetes remains uncertain, and a trial assessing the impact of CAC testing in this population has not been conducted. Such a trial would have to be based on the fact that the greatest utility of CAC testing is to identify those individuals who are being considered for statin therapy, in whom there is a strong patient preference to avoid statins (if the CAC score is 0), but a willingness to initiate therapy if coronary atherosclerosis is detected.
THE ROLE OF CAC IN INDIVIDUALS WHO HAVE DIABETES
- In the future, additional preventive therapies beyond statins may be considered for selected individuals who do not have known CVD. Realizing such therapies are likely to be more expensive and may be associated with more adverse effects, it is plausible that the allocation of newer therapies may be improved by using CAC testing in order to identify those with the highest risk, who therefore stand to have the greatest reduction in events. Realizing this potential role, future drug development trials may benefit from using CAC testing as a way to better enrich the population tested.
FUTURE ROLE OF CAC BEYOND STATINS
- CAC testing is now well recognized as a simple, reproducible, and inexpensive test to assess for the presence or absence of coronary atherosclerosis. Most individuals who have a CAC=0 have a very low risk of future cardiovascular events over the next 10 to 15 years and can elect to defer statin therapy and instead focus on lifestyle intervention. On the other hand, for those who are being considered for statin therapy, the presence of CAC—irrespective of whether other risk factors are present or absent, and regardless of age—can be used to provide a more precise risk assessment, and consequently provide a more compelling case for using pharmacotherapy.
CONCLUSIONS
-
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.
Article information
- 1. Detrano R, Guerci AD, Carr JJ, Bild DE, Burke G, Folsom AR, et al. Coronary calcium as a predictor of coronary events in four racial or ethnic groups. N Engl J Med 2008;358:1336–1345. ArticlePubMed
- 2. Blaha MJ, Budoff MJ, DeFilippis AP, Blankstein R, Rivera JJ, Agatston A, et al. Associations between C-reactive protein, coronary artery calcium, and cardiovascular events: implications for the JUPITER population from MESA, a population-based cohort study. Lancet 2011;378:684–692. ArticlePubMedPMC
- 3. Blankstein R, Budoff MJ, Shaw LJ, Goff DC Jr, Polak JF, Lima J, et al. Predictors of coronary heart disease events among asymptomatic persons with low low-density lipoprotein cholesterol MESA (Multi-Ethnic Study of Atherosclerosis). J Am Coll Cardiol 2011;58:364–374. ArticlePubMed
- 4. Silverman MG, Blaha MJ, Krumholz HM, Budoff MJ, Blankstein R, Sibley CT, et al. Impact of coronary artery calcium on coronary heart disease events in individuals at the extremes of traditional risk factor burden: the Multi-Ethnic Study of Atherosclerosis. Eur Heart J 2014;35:2232–2241. ArticlePubMedPDF
- 5. Polonsky TS, McClelland RL, Jorgensen NW, Bild DE, Burke GL, Guerci AD, et al. Coronary artery calcium score and risk classification for coronary heart disease prediction. JAMA 2010;303:1610–1616. ArticlePubMedPMC
- 6. Yeboah J, McClelland RL, Polonsky TS, Burke GL, Sibley CT, O'Leary D, et al. Comparison of novel risk markers for improvement in cardiovascular risk assessment in intermediate-risk individuals. JAMA 2012;308:788–795. ArticlePubMedPMC
- 7. Kavousi M, Elias-Smale S, Rutten JH, Leening MJ, Vliegenthart R, Verwoert GC, et al. Evaluation of newer risk markers for coronary heart disease risk classification: a cohort study. Ann Intern Med 2012;156:438–444. ArticlePubMed
- 8. Rana JS, Gransar H, Wong ND, Shaw L, Pencina M, Nasir K, et al. Comparative value of coronary artery calcium and multiple blood biomarkers for prognostication of cardiovascular events. Am J Cardiol 2012;109:1449–1453. ArticlePubMed
- 9. Sarwar A, Shaw LJ, Shapiro MD, Blankstein R, Hoffmann U, Cury RC, et al. Diagnostic and prognostic value of absence of coronary artery calcification. JACC Cardiovasc Imaging 2009;2:675–688. ArticlePubMed
- 10. Blaha MJ, Cainzos-Achirica M, Greenland P, McEvoy JW, Blankstein R, Budoff MJ, et al. Role of coronary artery calcium score of zero and other negative risk markers for cardiovascular disease: the Multi-Ethnic Study of Atherosclerosis (MESA). Circulation 2016;133:849–858. ArticlePubMedPMC
- 11. Tota-Maharaj R, Blaha MJ, McEvoy JW, Blumenthal RS, Muse ED, Budoff MJ, et al. Coronary artery calcium for the prediction of mortality in young adults <45 years old and elderly adults >75 years old. Eur Heart J 2012;33:2955–2962. ArticlePubMedPDF
- 12. Blankstein R, Foody JM. Screening for coronary artery disease in patients with family history... how, when, and in whom? Circ Cardiovasc Imaging 2014;7:417–419. ArticlePubMed
- 13. Nasir K, Bittencourt MS, Blaha MJ, Blankstein R, Agatson AS, Rivera JJ, et al. Implications of coronary artery calcium testing among statin candidates according to American College of Cardiology/American Heart Association Cholesterol Management Guidelines: MESA (Multi-Ethnic Study of Atherosclerosis). J Am Coll Cardiol 2015;66:1657–1668. ArticlePubMed
- 14. Mahabadi AA, Mohlenkamp S, Lehmann N, Kalsch H, Dykun I, Pundt N, et al. CAC score improves coronary and CV risk assessment above statin indication by ESC and AHA/ACC primary prevention guidelines. JACC Cardiovasc Imaging 2017;10:143–153. ArticlePubMed
- 15. Messenger B, Li D, Nasir K, Carr JJ, Blankstein R, Budoff MJ. Coronary calcium scans and radiation exposure in the multi-ethnic study of atherosclerosis. Int J Cardiovasc Imaging 2016;32:525–529. ArticlePubMedPDF
- 16. Miedema MD, Duprez DA, Misialek JR, Blaha MJ, Nasir K, Silverman MG, et al. Use of coronary artery calcium testing to guide aspirin utilization for primary prevention: estimates from the Multi-Ethnic Study of Atherosclerosis. Circ Cardiovasc Qual Outcomes 2014;7:453–460. ArticlePubMedPMC
- 17. McEvoy JW, Martin SS, Dardari ZA, Miedema MD, Sandfort V, Yeboah J, et al. Coronary artery calcium to guide a personalized risk-based approach to initiation and intensification of antihypertensive therapy. Circulation 2017;135:153–165. ArticlePubMed
- 18. Reis JP, Loria CM, Lewis CE, Powell-Wiley TM, Wei GS, Carr JJ, et al. Association between duration of overall and abdominal obesity beginning in young adulthood and coronary artery calcification in middle age. JAMA 2013;310:280–288. ArticlePubMedPMC
- 19. Nasir K, Rubin J, Blaha MJ, Shaw LJ, Blankstein R, Rivera JJ, et al. Interplay of coronary artery calcification and traditional risk factors for the prediction of all-cause mortality in asymptomatic individuals. Circ Cardiovasc Imaging 2012;5:467–473. ArticlePubMed
- 20. Carr JJ, Jacobs DR Jr, Terry JG, Shay CM, Sidney CM, Liu K, et al. Association of coronary artery calcium in adults aged 32 to 46 years with incident coronary heart disease and death. JAMA Cardiol 2017 2 08 [Epub]. Article
- 21. Blankstein R, Greenland P. Screening for coronary artery disease at an earlier age. JAMA Cardiol 2017 2 08 [Epub]. Article
- 22. Arad Y, Spadaro LA, Roth M, Newstein D, Guerci AD. Treatment of asymptomatic adults with elevated coronary calcium scores with atorvastatin, vitamin C, and vitamin E: the St. Francis Heart Study randomized clinical trial. J Am Coll Cardiol 2005;46:166–172. ArticlePubMed
- 23. Gupta A, Varshney R, Lau E, Hulten E, Bittencourt MS, Blaha MJ, et al. The identification of coronary atherosclerosis is associated with initiation of pharmacologic and lifestyle preventive therapies: a systematic review and metaanalysis In: 2016 American College of Cardiology (ACC) 65th Annual Scientific Session and Expo; 2016 Apr 2-4; Chicago, IL.
- 24. Rozanski A, Gransar H, Shaw LJ, Kim J, Miranda-Peats L, Wong ND, et al. Impact of coronary artery calcium scanning on coronary risk factors and downstream testing the EISNER (Early Identification of Subclinical Atherosclerosis by Noninvasive Imaging Research) prospective randomized trial. J Am Coll Cardiol 2011;57:1622–1632. ArticlePubMedPMC
- 25. Orakzai RH, Nasir K, Orakzai SH, Kalia N, Gopal A, Musunuru K, et al. Effect of patient visualization of coronary calcium by electron beam computed tomography on changes in beneficial lifestyle behaviors. Am J Cardiol 2008;101:999–1002. ArticlePubMed
- 26. Kalia NK, Miller LG, Nasir K, Blumenthal RS, Agrawal N, Budoff MJ. Visualizing coronary calcium is associated with improvements in adherence to statin therapy. Atherosclerosis 2006;185:394–399. ArticlePubMed
- 27. Taylor AJ, Bindeman J, Feuerstein I, Le T, Bauer K, Byrd C, et al. Community-based provision of statin and aspirin after the detection of coronary artery calcium within a community-based screening cohort. J Am Coll Cardiol 2008;51:1337–1341. ArticlePubMed
- 28. Nasir K, McClelland RL, Blumenthal RS, Goff DC Jr, Hoffmann U, Psaty BM, et al. Coronary artery calcium in relation to initiation and continuation of cardiovascular preventive medications: the Multi-Ethnic Study of Atherosclerosis (MESA). Circ Cardiovasc Qual Outcomes 2010;3:228–235. ArticlePubMedPMC
- 29. Schwartz J, Allison M, Wright CM. Health behavior modification after electron beam computed tomography and physician consultation. J Behav Med 2011;34:148–155. ArticlePubMed
- 30. O'Malley PG, Feuerstein IM, Taylor AJ. Impact of electron beam tomography, with or without case management, on motivation, behavioral change, and cardiovascular risk profile: a randomized controlled trial. JAMA 2003;289:2215–2223. ArticlePubMed
- 31. Obuchowski NA, Holden D, Modic MT, Cheah G, Fu AZ, Brant-Zawadzki M, et al. Total-body screening: preliminary results of a pilot randomized controlled trial. J Am Coll Radiol 2007;4:604–611. ArticlePubMed
- 32. Lederman J, Ballard J, Njike VY, Margolies L, Katz DL. Information given to postmenopausal women on coronary computed tomography may influence cardiac risk reduction efforts. J Clin Epidemiol 2007;60:389–396. ArticlePubMed
- 33. Whelton SP, Nasir K, Blaha MJ, Gransar H, Metkus TS, Coresh J, et al. Coronary artery calcium and primary prevention risk assessment: what is the evidence? An updated meta-analysis on patient and physician behavior. Circ Cardiovasc Qual Outcomes 2012;5:601–607. ArticlePubMedPMC
- 34. Mols RE, Jensen JM, Sand NP, Fuglesang C, Bagdat D, Vedsted P, et al. Visualization of coronary artery calcification: influence on risk modification. Am J Med 2015;128:1023.e23–1023.e31.ArticlePubMed
- 35. Gibson AO, Blaha MJ, Arnan MK, Sacco RL, Szklo M, Herrington DM, et al. Coronary artery calcium and incident cerebrovascular events in an asymptomatic cohort. The MESA study. JACC Cardiovasc Imaging 2014;7:1108–1115. ArticlePubMedPMC
- 36. Hermann DM, Gronewold J, Lehmann N, Moebus S, Jockel KH, Bauer M, et al. Coronary artery calcification is an independent stroke predictor in the general population. Stroke 2013;44:1008–1013. ArticlePubMed
- 37. Bittencourt MS, Blankstein R, Mao S, Rivera JJ, Bertoni AG, Shaw LJ, et al. Left ventricular area on non-contrast cardiac computed tomography as a predictor of incident heart failure: the Multi-Ethnic Study of Atherosclerosis. J Cardiovasc Comput Tomogr 2016;10:500–506. ArticlePubMedPMC
- 38. Kalsch H, Lehmann N, Mohlenkamp S, Neumann T, Slomiany U, Schmermund A, et al. Association of coronary artery calcium and congestive heart failure in the general population: results of the Heinz Nixdorf recall study. Clin Res Cardiol 2010;99:175–182. ArticlePubMedPDF
- 39. O'Neal WT, Efird JT, Dawood FZ, Yeboah J, Alonso A, Heckbert SR, et al. Coronary artery calcium and risk of atrial fibrillation (from the Multi-Ethnic Study of Atherosclerosis). Am J Cardiol 2014;114:1707–1712. ArticlePubMedPMC
- 40. Handy CE, Desai CS, Dardari ZA, Al-Mallah MH, Miedema MD, Ouyang P, et al. The association of coronary artery calcium with noncardiovascular disease: the Multi-Ethnic Study of Atherosclerosis. JACC Cardiovasc Imaging 2016;9:568–576. ArticlePubMedPMC
- 41. Rana JS, Liu JY, Moffet HH, Jaffe M, Karter AJ. Diabetes and prior coronary heart disease are not necessarily risk equivalent for future coronary heart disease events. J Gen Intern Med 2016;31:387–393. ArticlePubMedPDF
- 42. Rana JS, Blankstein R. Are all individuals with diabetes equal, or some more equal than others? JACC Cardiovasc Imaging 2016;9:1289–1291. ArticlePubMed
- 43. Young LH, Wackers FJ, Chyun DA, Davey JA, Barrett EJ, Taillefer R, et al. Cardiac outcomes after screening for asymptomatic coronary artery disease in patients with type 2 diabetes: the DIAD study: a randomized controlled trial. JAMA 2009;301:1547–1555. ArticlePubMedPMC
- 44. Muhlestein JB, Lappe DL, Lima JA, Rosen BD, May HT, Knight S, et al. Effect of screening for coronary artery disease using CT angiography on mortality and cardiac events in high-risk patients with diabetes: the FACTOR-64 randomized clinical trial. JAMA 2014;312:2234–2243. ArticlePubMed
- 45. Malik S, Budoff MJ, Katz R, Blumenthal RS, Bertoni AG, Nasir K, et al. Impact of subclinical atherosclerosis on cardiovascular disease events in individuals with metabolic syndrome and diabetes: the Multi-Ethnic Study of Atherosclerosis. Diabetes Care 2011;34:2285–2290. ArticlePubMedPMC
References
Conceptual frameworks of how the presence or absence of coronary artery calcium (CAC) may influence patient management.
Unadjusted Kaplan-Meier cumulative-event curves for coronary events among participants with coronary artery calcium scores of 0, 1 to 100, 101 to 300, and more than 300. The figure shows the rates for any coronary event. The differences among all curves are statistically significant (P<0.001). Adapted from Detrano et al. [1], with permission from Massachusetts Medical Society.
Relationship between pretest and posttest cardiovascular disease (CVD) risk after the knowledge of the negative result of each risk marker. The regression lines display the relationship between the pretest predicted 10-year atherosclerotic CVD risk (x axis) and the posttest risk (y axis) after the knowledge of the negative result of each risk marker. A broken back line is displayed as reference (risk shift with no additional testing). Results were obtained by plotting the pretest and posttest risk on the basis of the diagnostic likelihood ratio of each Multi-Ethnic Study of Atherosclerosis (MESA) participant and then applying a linear fit. Adapted from Blaha et al. [10], with permission from Wolters Kluwer Health, Inc. CAC, coronary artery calcium; BNP, brain natriuretic peptide; CIMT, carotid intima-media thickness; CHD, coronary heart disease; ABI, ankle brachial index; hs-CRP, high-sensitivity C-reactive protein.
Cumulative incidence of coronary heart disease events among Multi-Ethnic Study of Atherosclerosis (MESA) participants with low low-density lipoprotein cholesterol (<130 mg/dL) stratified by the presence or absence of diabetes, hypertension as well as the presence or absence of coronary artery calcium (CAC). (A) No diabetes, (B) diabetes, (C) no hypertension, and (D) hypertension. Adapted from Blankstein et al. [3].

(A) Coronary artery calcium (CAC) distribution across statin eligibility groups according to the American College of Cardiology/American Heart Association (ACC/AHA) cholesterol management guidelines. (B) Impact of the absence of CAC in reclassifying risk below the threshold for statin consideration suggested by ACC/AHA cholesterol management guidelines across the spectrum of estimated 10-year atherosclerotic cardiovascular disease (ASCVD) risk score (nondiabetic patients with low-density lipoprotein cholesterol of 70 to 189 mg/dL). Adapted from Nasir et al. [13].
(A, B) Coronary and cardiovascular event rate for subjects with and without statin indication according to European Society of Cardiology (ESC) and American Heart Association/American College of Cardiology (AHA/ACC) guidelines, stratified by coronary artery calcification (CAC) group, showing a distinct increase in event rates for both coronary and cardiovascular events with increasing CAC score, irrespective of statin indication according to ESC and AHA/ACC guidelines. Adapted from Mahabadi et al. [14], with permission from Elsevier.
Estimated risk/benefit of aspirin in primary prevention by coronary artery calcium (CAC) score in Multi-Ethnic Study of Atherosclerosis (MESA) participants. Coronary heart disease (CHD) risk was calculated using the Framingham Risk Score. The red line represents the estimated 5-year number needed to harm based on a 0.23% increase in major bleeding over 5 years. The 5-year number needed to treat estimations is based on an 18% relative reduction in coronary heart disease events. Adapted from Miedema et al. [16], with permission from Wolters Kluwer Health, Inc.
Annualized unadjusted cardiovascular disease (CVD) event rates in Multi-Ethnic Study of Atherosclerosis (MESA) stratified by coronary artery calcification (CAC) and the presence of diabetes mellitus (DM), metabolic syndrome (MetS), or neither. Adapted from Malik et al. [45], with permission from American Diabetes Association.

Recommended Interventions to Prevent Cardiovascular Disease Based on CAC Score
Mechanisms by Which Coronary Artery Calcium Testing May Improve Cardiovascular Outcomes
Figure & Data
References
Citations

- Artificial Intelligence in Coronary Artery Calcium Scoring Detection and Quantification
Khaled Abdelrahman, Arthur Shiyovich, Daniel Huck, Adam Berman, Brittany Weber, Sumit Gupta, Rhanderson Cardoso, Ron Blankstein
Diagnostics.2024; 14(2): 125. CrossRef - Association of Strict Versus Lenient Cholesterol Lowering with Cardiac Outcomes, Diabetes Progression and Complications, and Mortality in Patients with Diabetes Treated with Statins: Is Less More?
Victoria Odeleye, Omar Masarweh, Jorge Restrepo, Carlos A Alvarez, Ishak A. Mansi
Drug Safety.2023; 46(11): 1105. CrossRef - Impact of coronary artery calcium testing on patient management
Wanda Y. Wu, David W. Biery, Adam N. Berman, Grace Hsieh, Sanjay Divakaran, Sumit Gupta, Michael L. Steigner, Ayaz Aghayev, Hicham Skali, Donna M. Polk, Jorge Plutzky, Christopher P. Cannon, Marcelo F. Di Carli, Ron Blankstein
Journal of Cardiovascular Computed Tomography.2022; 16(4): 303. CrossRef - Application of ensemble machine learning algorithms on lifestyle factors and wearables for cardiovascular risk prediction
Weiting Huang, Tan Wei Ying, Woon Loong Calvin Chin, Lohendran Baskaran, Ong Eng Hock Marcus, Khung Keong Yeo, Ng See Kiong
Scientific Reports.2022;[Epub] CrossRef - The Influence of Obesity and Metabolic Health on Vascular Health
Eun-Jung Rhee
Endocrinology and Metabolism.2022; 37(1): 1. CrossRef - The role of cardiac computed tomography in predicting adverse coronary events
Maria Emfietzoglou, Michail C. Mavrogiannis, Athanasios Samaras, Georgios P. Rampidis, George Giannakoulas, Polydoros N. Kampaktsis
Frontiers in Cardiovascular Medicine.2022;[Epub] CrossRef - Subclinical Hypertension-Mediated Organ Damage (HMOD) in Hypertension: Atherosclerotic Cardiovascular Disease (ASCVD) and Calcium Score
Damiano Rizzoni, Claudia Agabiti-Rosei, Carolina De Ciuceis, Gianluca Edoardo Mario Boari
High Blood Pressure & Cardiovascular Prevention.2022; 30(1): 17. CrossRef - Coronary Artery Calcium Score: Current Efficacy of Cardiac CT in Patients at Hail Region, Saudi Arabia
Qurain Turki Alshammari, Ohood Alrashidi, Wed Almutairi, Eman Alshammari, Meshari T. Alshammari, Shashi Kumar CG, Mohammed Salih, Abdelmoneim Sulieman, Moawia Gameraddin, Bushra Abdul Malik, Amjad R. Alyahyawi
International Journal of Pharmaceutical Research And Allied Sciences.2022; 11(3): 132. CrossRef - The co-existence of diabetes and subclinical atherosclerosis in rural central Appalachia: Do residential characteristics matter?
Hadii M. Mamudu, Antwan Jones, Timir K. Paul, Fenose Osedeme, David Stewart, Arsham Alamian, Liang Wang, Sylvester Orimaye, James Bledsoe, Amy Poole, Gerald Blackwell, Mathew Budoff
Journal of Diabetes and its Complications.2021; 35(4): 107851. CrossRef - Usefulness of skin advanced glycation end products to predict coronary artery calcium score in patients with type 2 diabetes
Alejandra Planas, Olga Simó-Servat, Jordi Bañeras, Mónica Sánchez, Esther García, Ángel M. Ortiz, Marisol Ruiz-Meana, Cristina Hernández, Ignacio Ferreira-González, Rafael Simó
Acta Diabetologica.2021; 58(10): 1403. CrossRef - Value of addition of coronary artery calcium to risk scores in the prediction of major cardiovascular events in patients with type 2 diabetes
Barak Zafrir, Walid Saliba, Rachel Shay Li Widder, Razi Khoury, Elad Shemesh, David A. Halon
BMC Cardiovascular Disorders.2021;[Epub] CrossRef - Coronary Artery Calcium Score - A Reliable Indicator of Coronary Artery Disease?
Devarashetty Shreya, Diana I Zamora, Gautami S Patel, Idan Grossmann, Kevin Rodriguez, Mridul Soni, Pranay K Joshi, Saawan C Patel, Ibrahim Sange
Cureus.2021;[Epub] CrossRef - Metformin, resveratrol, and exendin-4 inhibit high phosphate-induced vascular calcification via AMPK-RANKL signaling
Jinmi Lee, Seok-Woo Hong, Min-Jeong Kim, Hyemi Kwon, Se Eun Park, Eun-Jung Rhee, Won-Young Lee
Biochemical and Biophysical Research Communications.2020; 530(2): 374. CrossRef - Coronary Computed Tomography Angiography From Clinical Uses to Emerging Technologies
Khaled M. Abdelrahman, Marcus Y. Chen, Amit K. Dey, Renu Virmani, Aloke V. Finn, Ramzi Y. Khamis, Andrew D. Choi, James K. Min, Michelle C. Williams, Andrew J. Buckler, Charles A. Taylor, Campbell Rogers, Habib Samady, Charalambos Antoniades, Leslee J. Sh
Journal of the American College of Cardiology.2020; 76(10): 1226. CrossRef - Optimizing coronary artery calcium scanning to meet the challenges of population screening
Andrew J. Einstein
Journal of Cardiovascular Computed Tomography.2019; 13(6): 303. CrossRef - Usefulness of the Coronary Artery Calcium Score in Predicting Subsequent Coronary Interventions—A Ten-Year Single-Center Perspective
Agnieszka Mlynarska, Rafal Mlynarski, Maciej Sosnowski
International Journal of Environmental Research and Public Health.2019; 16(12): 2132. CrossRef - Cardiovascular Risk Heterogeneity in Adults with Diabetes: Selective Use of Coronary Artery Calcium in Statin Use Decision-making
Sudipa Sarkar, Olusola A. Orimoloye, Caitlin M. Nass, Roger S. Blumenthal, Seth S. Martin
Journal of General Internal Medicine.2019; 34(11): 2643. CrossRef - CT coronary imaging–a fast evolving world
P Rajiah, S Abbara
QJM: An International Journal of Medicine.2018; 111(9): 595. CrossRef - Being Metabolically Healthy, the Most Responsible Factor for Vascular Health
Eun-Jung Rhee
Diabetes & Metabolism Journal.2018; 42(1): 19. CrossRef - Diabetes, subclinical atherosclerosis and multiple cardiovascular risk factors in hard-to-reach asymptomatic patients
Hadii M Mamudu, Arsham Alamian, Timir Paul, Pooja Subedi, Liang Wang, Antwan Jones, Ali E Alamin, David Stewart, Gerald Blackwell, Matthew Budoff
Diabetes and Vascular Disease Research.2018; 15(6): 519. CrossRef - Calcium detection, its quantification, and grayscale morphology-based risk stratification using machine learning in multimodality big data coronary and carotid scans: A review
Sumit K. Banchhor, Narendra D. Londhe, Tadashi Araki, Luca Saba, Petia Radeva, Narendra N. Khanna, Jasjit S. Suri
Computers in Biology and Medicine.2018; 101: 184. CrossRef - Association Between Coronary Artery Calcification and the Hemoglobin Glycation Index: The Kangbuk Samsung Health Study
Eun-Jung Rhee, Jung-Hwan Cho, Hyemi Kwon, Se Eun Park, Cheol-Young Park, Ki-Won Oh, Sung-Woo Park, Won-Young Lee
The Journal of Clinical Endocrinology & Metabolism.2017; 102(12): 4634. CrossRef - Increased risk for development of coronary artery calcification in subjects with non-alcoholic fatty liver disease and systemic inflammation
Jihyun Kim, Da Young Lee, Se Eun Park, Cheol-Young Park, Won-Young Lee, Ki-Won Oh, Sung-Woo Park, Eun-Jung Rhee, Susanne Kaser
PLOS ONE.2017; 12(7): e0180118. CrossRef

 KES
KES
 PubReader
PubReader Cite
Cite









