Articles
- Page Path
- HOME > Endocrinol Metab > Volume 32(4); 2017 > Article
-
Original ArticleClinical Study Effects of Single Vitamin D3 Injection (200,000 Units) on Serum Fibroblast Growth Factor 23 and Sclerostin Levels in Subjects with Vitamin D Deficiency
-
Dongdong Zhang1,2
 , Da Hea Seo3
, Da Hea Seo3 , Han Seok Choi4
, Han Seok Choi4 , Hye-Sun Park3
, Hye-Sun Park3 , Yoon-Sok Chung5
, Yoon-Sok Chung5 , Sung-Kil Lim1,3
, Sung-Kil Lim1,3
-
Endocrinology and Metabolism 2017;32(4):451-459.
DOI: https://doi.org/10.3803/EnM.2017.32.4.451
Published online: December 14, 2017
1Brain Korea 21 PLUS Project for Medical Science, Yonsei University, Seoul, Korea.
2Division of Endocrinology and Metabolism, Department of Internal Medicine, Yantai Affiliated Hospital of Binzhou Medical University, Yantai, China.
3Division of Endocrinology and Endocrine Research Institute, Department of Internal Medicine, Yonsei University College of Medicine, Seoul, Korea.
4Division of Endocrinology and Metabolism, Department of Internal Medicine, Dongguk University Ilsan Hospital, Dongguk University College of Medicine, Goyang, Korea.
5Division of Endocrinology and Metabolism, Department of Internal Medicine, Ajou University School of Medicine, Suwon, Korea.
- Corresponding author: Sung-Kil Lim. Division of Endocrinology and Endocrine Research Institute, Department of Internal Medicine, Yonsei University College of Medicine, 50-1 Yonsei-ro, Seodaemun-gu, Seoul 03722, Korea. Tel: +82-2-2228-0878, Fax: +82-2-393-6884, lsk@yuhs.ac
Copyright © 2017 Korean Endocrine Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
-
Background
- Vitamin D deficiency remains common in all age groups and affects skeletal and non-skeletal health. Fibroblast growth factor 23 is a bone-derived hormone that regulates phosphate and 1,25-dihydroxyvitamin D homeostasis as a counter regulatory factor. 1,25-Dihydroxyvitamin D stimulates fibroblast growth factor 23 synthesis in bone, while fibroblast growth factor 23 suppresses 1,25-dihydroxyvitamin D production in the kidney. The aim of this study was to evaluate the effects of vitamin D3 intramuscular injection therapy on serum fibroblast growth factor 23 concentrations, and several other parameters associated with bone metabolism such as sclerostin, dickkopf-1, and parathyroid hormone.
-
Methods
- A total of 34 subjects with vitamin D deficiency (defined by serum 25-hydroxyvitamin D levels below 20 ng/mL) were randomly assigned to either the vitamin D injection group (200,000 units) or placebo treatment group. Serum calcium, phosphate, urine calcium/creatinine, serum 25-hydroxyvitamin D, fibroblast growth factor 23, sclerostin, parathyroid hormone, and dickkopf-1 levels were serially measured after treatment.
-
Results
- Comparing the vitamin D injection group with the placebo group, no significant changes were observed in serum fibroblast growth factor 23, parathyroid hormone, or dickkopf-1 levels. Serum sclerostin concentrations transiently increased at week 4 in the vitamin D group. However, these elevated levels declined later and there were no statistically significant differences as compared with baseline levels.
-
Conclusion
- Serum fibroblast factor 23, sclerostin, parathyroid hormone, and dickkopf-1 levels were not affected significantly by single intramuscular injection of vitamin D3.
- Vitamin D plays an essential role in bone and mineral metabolism [1]. It is also important in non-skeletal tissues, and its deficiency is closely associated with increased risk of cancers, infections, autoimmune diseases, cardiovascular diseases, and diabetes mellitus [2345]. Despite growing public awareness of the multiple health benefits of vitamin D, epidemiological studies have revealed a very high prevalence of vitamin D deficiency worldwide, especially in Asian countries [67].
- Vitamin D is mainly produced in the skin when directly exposed to sunlight, or obtained from the diet. Active vitamin D maintains calcium and phosphate homeostasis by promoting intestinal absorption for the bone mineralization process [8]. Aside from its role in the endocrine pathway, active vitamin D is known to affect the differentiation and function of bone cells by targeting key genes involved in bone formation and resorption [9]. Oral supplementation with vitamin D and calcium is common practice in the treatment of vitamin D deficiency. In addition to oral supplementation, vitamin D can also be administered by intramuscular injection. Heikinheimo et al. [10] reported that annual intramuscular injection of ergocalciferol vitamin D2 (150,000 to 300,000 IU) resulted in a significant reduction in the incidence of fractures.
- To maintain calcium and phosphate homeostasis, active vitamin D works in combination with two other hormones: fibroblast growth factor 23 (FGF23) and parathyroid hormone (PTH) [11]. 1,25-Dihydroxyvitamin D (1,25(OH)2D) stimulates FGF23 synthesis in bone, while FGF23 suppresses the production of 1,25(OH)2D; thus, acting as a counter-regulatory factor [12]. FGF23 levels are reportedly significantly elevated in patients with chronic kidney disease (CKD), who are at increased risk of mortality mainly from cardiovascular disease [13]. CKD-mineral and bone disorder (CKD-MBD) occurs from the early stages of CKD, and there is a strong association between FGF23 and cardiovascular risks, left ventricular hypertrophy, and vascular calcification [131415]. A study conducted in patients with vitamin D deficiency who were given a high dose vitamin D2 injection (300,000 IU) combined with usual daily oral supplementations of calcium (1.2 g) and vitamin D3 (800 IU) reported that high dose vitamin D increased 1,25(OH)2D and FGF23 concentrations [16]. However, there is a paucity of data regarding the effects of single intramuscular injections of 200,000 IU of vitamin D3.
- Sclerostin and dickkopf-1 (DKK1) are two important endogenous Wnt signaling antagonists, mainly produced in bone [1718]. Sankaralingam et al. [19] reported that bolus intramuscular injection of 300,000 IU of vitamin D combined with oral supplementation of vitamin D and calcium increased sclerostin and DKK1 concentrations. In contrast, another study demonstrated that in patients with vitamin D deficiency who were given a monthly intramuscular injection of 300,000 IU of vitamin D, sclerostin levels decreased considerably after treatment [20].
- The aim of our study was to determine the effects of single intramuscular injection of vitamin D3 at 200,000 IU on circulating concentrations of 25-hydroxyvitamin D (25(OH)D), FGF23, sclerostin, and DKK1 in patients with vitamin D deficiency
INTRODUCTION
- Study subjects and design
- Thirty-four subjects (five males and 29 females) aged 33.2±7.7 years, diagnosed with vitamin D deficiency at Severance Hospital, Yonsei University College of Medicine and Dongguk University Ilsan Hospital in Korea were recruited in this study. Vitamin D deficiency was diagnosed if serum 25(OH)D concentrations were below 20 ng/mL. Patients who were taking medications that may affect vitamin D metabolism were excluded from the study.
- This study was designed as a randomized, double-blinded, and placebo-controlled clinical trial. Subjects were randomly assigned to the experimental group or the placebo group at a 2:1 ratio. All subjects were given either a single intramuscular injection of vitamin D3 (200,000 IU), or a placebo at the beginning of the study. During this intervention period, consumption of any additional vitamin D supplements and exposure to direct sunlight for more than 10 hours were prohibited. Usual diets were maintained, except diets containing abundant vitamin D.
- Laboratory measurements
- Fasting blood samples were collected at baseline and weeks 4, 8, 12, and 14 during follow-up visits after vitamin D or placebo injections. Routine biochemical parameters including basic chemistry tests (measuring glucose, calcium, phosphorus, blood urea nitrogen, and creatinine), hematology tests (hemoglobin, hematocrit, and hemoglobin A1c), and urinalyses were conducted via standard methods at each institution, and additional serum samples were stored at −70 for subsequent analyses. Serum 25(OH)D and intact PTH concentrations were measured by electrochemiluminescence immunoassay (ECLIA) (Roche Diagnostics, Mannheim, Germany). The intra- and inter-assay coefficients of variation (CV) for 25(OH)D measurements were 2.2% to 6.8% and 3.4% to 13.1%, respectively. The intra- and inter-assay CV of intact PTH measurements were 1.1% to 2.0% and 2.8% to 3.4%, respectively. Serum FGF23 levels were measured using an intact FGF23 (iFGF23) enzyme-linked immunosorbent assay (ELISA) kit (Millipore Corporation, Billerica, MA, USA). The intra- and inter-assay CV of iFGF23 measurements were 7.8% to 11.2% and 2.4% to 11.31%, respectively. Sclerostin and DKK1 concentrations were also measured using human sclerostin and DKK-1 ELISA kits (R&D Systems, Minneapolis, MN, USA) according to the manufacturer's instructions. The intra- and inter-assay CV of sclerostin measurements were 1.8% to 2.1% and 8.2% to 10.8%, respectively. The intra- and inter-assay CV of DKK1 measurements were 3.3% to 4.2% and 4.6% to 7.6%, respectively.
- Ethics statement
- This study protocol was reviewed and approved by the Institutional Review Broad (IRB: 4-2014-0377) at both institutions. Informed consent was submitted by all subjects when they were enrolled.
- Statistical analysis
- All statistical analyses were performed using GraphPad Prism version 5.01 (GraphPad Software, CA, USA). Results are presented as mean±standard error (SE). Student paired t test was used to compare the data from different time points with baseline values when the values were normally distributed, and Wilcoxon signed rank test was used when the data were nonparametric. An analysis of variance (ANOVA) was used to compare different time points between groups. Dunnett's test and Bonferroni correction were performed for the repeated measures test of one-way ANOVA and two-way ANOVA, respectively. A P value of <0.05 (95% confidence interval) was considered statistically significant.
METHODS
- Demographic characteristics and changes in routine biochemical parameters following single vitamin D injection
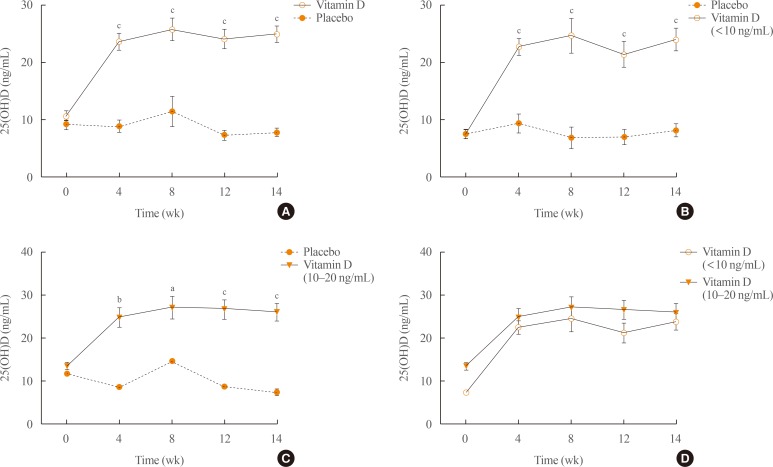
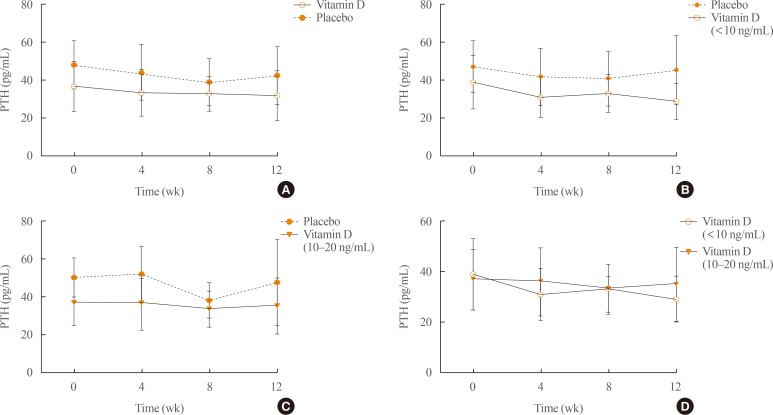
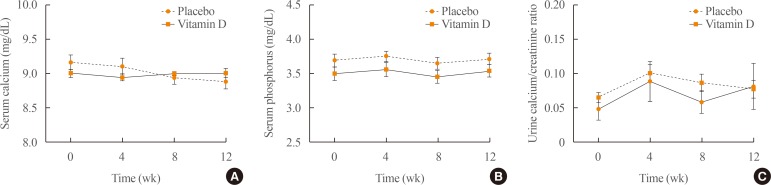
- Demographic information and other baseline characteristics of the subjects are shown in Table 1. There were no statistically significant differences between the vitamin D and placebo groups (P>0.05). Compared with the placebo group, no statistically significant differences were observed in serum calcium, phosphorus, and the ratio of urine calcium to creatinine at weeks 4, 8, and 12 after vitamin D3 injection (Fig. 1). Serum 25(OH)D concentrations increased significantly throughout the study (P<0.001) after vitamin D3 injection (Fig. 2A–C). Moreover, there were no significant differences between the two subgroups receiving vitamin D3 injections which had previously been grouped based on baseline levels of 25(OH)D (Fig. 2D). On the other hand, serum PTH concentrations at weeks 4, 8, and 12 after vitamin D3 injection in the vitamin D group did not change significantly (P>0.05) (Fig. 3A, D). No statistically significant differences were observed in the vitamin D group compared with the placebo group (P>0.05) (Fig. 3).
- Effects of vitamin D on serum FGF23 concentrations
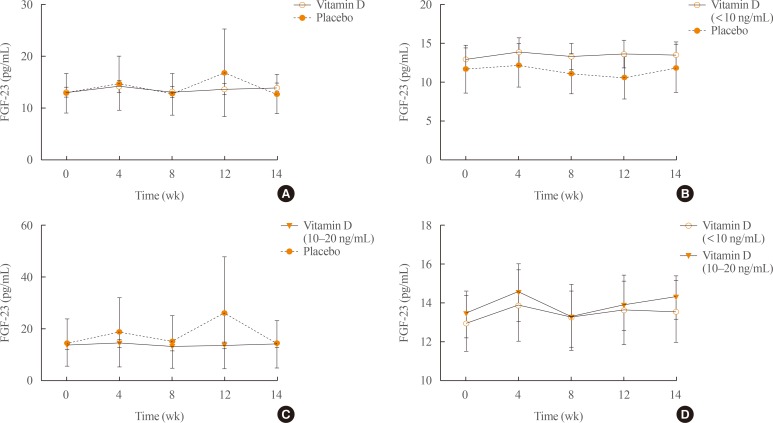
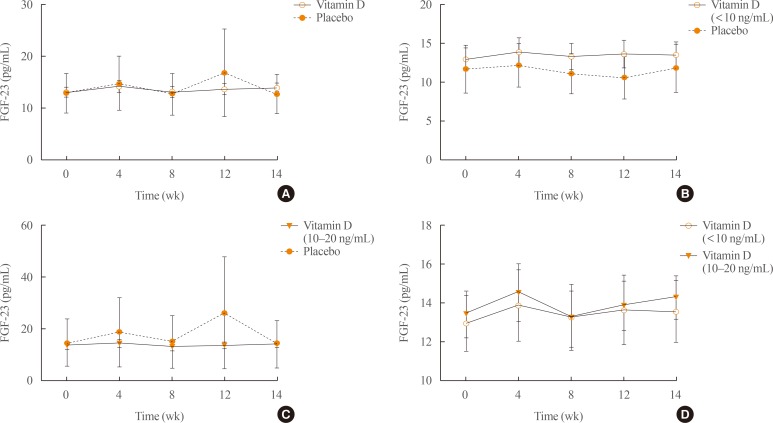
- To determine whether a single injection of vitamin D3 (200,000 IU) increases circulating FGF23 levels, we measured serum FGF23 concentrations at weeks 4, 8, 12, and 14 after vitamin D3 treatment. The results showed that no significant difference were observed between vitamin D3 treatment group and placebo group (Fig. 4A). Similar results were found in the two subgroups (Fig. 4B–D). However, subject 35 (placebo group) showed particularly elevated FGF23 levels throughout the study period including at baseline, even though this was in the placebo group without vitamin D3 injection.
- Changes in sclerostin and DKK1 levels
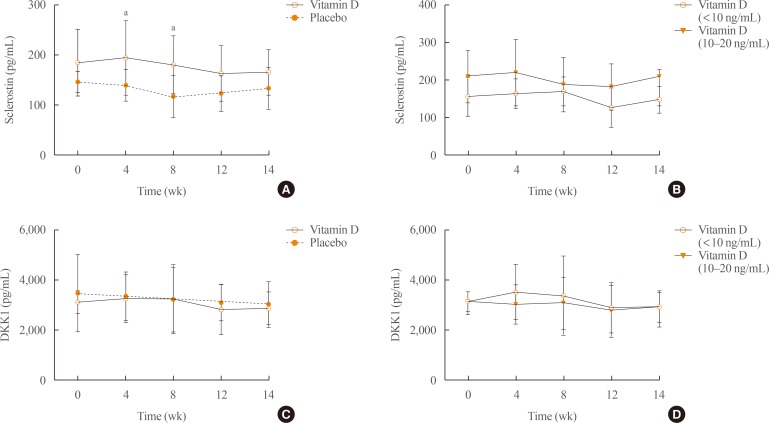
- To evaluate whether vitamin D3 injection stimulates the expression of Wnt inhibitors, we measured serum sclerostin and DKK1 concentrations after vitamin D3 injection. In the vitamin D group, serum sclerostin levels were transiently increased at week 4 after vitamin D3 injection. Comparing the vitamin D group with the placebo group, there were significant differences at weeks 4 and 8 (P<0.05) (Fig. 5A). However, the slightly increased levels declined thereafter, and were not significantly different when compared to baseline (Fig. 5A). Moreover, there were no significant differences in sclerostin levels between the two subgroups with vitamin D3 injection (Fig. 5B). DKK1 levels were not increased in the vitamin D group and there were no significant differences between this and the placebo group (Fig. 5C, D).
RESULTS
- In the present study, we have shown that serum 25(OH)D levels significantly increased following a single intramuscular injection of vitamin D3 with 200,000 IU, which is consistent with the report [21]. However, the levels of serum PTH, FGF23, and DKK1 did not change during the 14-week follow-up period after vitamin D3 injection. Serum sclerostin levels were slightly increased 4 weeks post-treatment but declined thereafter.
- Calcium alone, or combined with oral vitamin D, has been suggested as an inexpensive therapeutic method to prevent osteoporotic bone loss and fractures. However, this treatment is less effective if the patients' compliance is poor. Furthermore, therapeutic levels are only reached after a long period. Intramuscular injection of vitamin D alone or in combination with oral supplementation can maintain increased levels of serum 25(OH)D for at least 6 months [22]. Turner et al. [16] reported that intramuscular injection of vitamin D together with oral calcium and vitamin D supplementation, increased 1,25(OH)2D concentrations significantly. Similar findings were also demonstrated recently where 25(OH)D levels were elevated in vitamin D deficient patients who were given a monthly intramuscular injection of 300,000 IU of vitamin D for 3 consecutive months [20]. Our results are consistent with these findings over the 14 weeks following a single vitamin D3 injection at the dose of 200,000 IU.
- Active vitamin D stimulates the production of FGF23 in osteocytes and osteoblasts [23]. Furthermore, increased FGF23 levels reduce expression of 1α-hydroxylase but increase expression of 24-hydroxylase, which converts 1,25(OH)2D to the les biologically active 24,25(OH)2D, resulting in decreased 1,25(OH)2D production [12]. FGF23 concentrations were also increased after intramuscular injection of vitamin D (300,000 IU) administered in conjunction with usual daily supplementation of vitamin D and calcium [18]. In contrast, Uzum et al. [24] reported that FGF23 concentrations further declined during vitamin D replacement therapy in vitamin D deficient patients who were treated daily with an oral combination of vitamin D and calcium for 6 weeks. In our study, serum FGF23 levels were not significantly elevated after intramuscular injection of vitamin D3 (200,000 IU). In contrast with the previous reports, our study did not provide any evidence that single intramuscular injection of vitamin D3 (200,000 IU) could increase or decrease serum FGF23 levels at all. Participants, medication dosage, and treatment methods were all different in the various studies cited. Therefore, serum FGF23 levels may be affected by these factors. In this study, one subject in the placebo group showed high FGF23 concentrations at baseline and after-treatment, and was diagnosed with anemia without other abnormal test parameters. It is known that iron deficiency could elevate C-terminal FGF23 (cFGF23) levels, but not iFGF23 levels [25]. Following intravenous iron repletion, cFGF23 levels fell within 24 hours, whereas iFGF23 did not change significantly. Interestingly, culprit iron formulations could uncouple FGF23 production and cleavage, by decreasing cleavage to a greater extent than production, and thereby increase the serum concentration [26]. We used an iFGF23 detection ELISA kit which only measures iFGF23 and no information was available regarding iron supplementation in the subject. Therefore, the reasons behind the high iFGF23 levels in this subject are still unknown.
- Previous reports have shown that the loading dose in vitamin D supplementation influences PTH levels [27]. The sustained increase in serum PTH may affect bone metabolism negatively by increasing bone turnover, and it is expected that decreased PTH levels after vitamin D3 injection could be beneficial for skeletal bone health. In this study, we did not observe significant changes in serum PTH, calcium, and phosphate levels. The following may explain our findings: firstly, patients did not have secondary hyperparathyroidism at baseline; secondly, short term intramuscular injection of vitamin D3 at a dose of 200,000 IU may not reveal the PTH suppression effect.
- 1,25(OH)2D was reported to increase the expression of low density lipoprotein receptor-related protein 5 (LRP5), a Wnt co-receptor that plays a key role in Wnt signaling and bone formation [9]. Hence, we hypothesized that canonical Wnt signaling could be also regulated after vitamin D injection through up or down regulating Wnt inhibitors, such as DKK1 and sclerostin which bind to LRP5/6 and inhibit bone formation. In the previous study, bisphosphonate and denosumab, both used in the treatment of osteoporosis, have been shown to increase sclerostin levels, and either decrease or have no effect on DKK1 levels [2829]. The DKK1 response was lower than that of sclerostin, which occurred at 12 months following treatment with denosumab [28]. In our study, a slight increase in sclerostin levels was observed at week 4, and there was no significant change in DKK1 levels after vitamin D3 injection. These findings are consistent with the previous study where a significant increase in sclerostin was observed after 3 months, but no change was seen in DKK1 after a loading dose of vitamin D injection [19].
- The limitations of this study include the fact that only the effects of intramuscular injection of vitamin D3 (200,000 IU) in subjects with vitamin D deficiency were assessed, while serum 1,25(OH)D and bioavailable 25(OH)D levels were not measured. In addition, FGF23 levels were mostly below the limit of detection of the assay used, and a different assay with better sensitivity should be performed in future. The effects of vitamin D3 injection on FGF23 should be assessed further in patients with CKD, in whom 1,25(OH)2D replacement thereby for suppression of PTH increases serum FGF23 concentrations significantly.
- In conclusion, a single vitamin D3 injection (200,000 IU) significantly increased serum 25(OH)D concentrations, without affecting serum FGF23, PTH, and DKK1 levels during short term follow up of 14 weeks, and caused only a slight increase in serum sclerostin levels.
DISCUSSION
-
Acknowledgements
- This research was supported by the Brain Korea 21 PLUS Project for Medical Science of Yonsei University, and supported by Kwangdong pharmaceutical company.
ACKNOWLEDGMENTS
-
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.
Article information
- 1. Hofbauer LC, Dunstan CR, Spelsberg TC, Riggs BL, Khosla S. Osteoprotegerin production by human osteoblast lineage cells is stimulated by vitamin D, bone morphogenetic protein-2, and cytokines. Biochem Biophys Res Commun 1998;250:776–781. ArticlePubMed
- 2. Lin J, Manson JE, Lee IM, Cook NR, Buring JE, Zhang SM. Intakes of calcium and vitamin D and breast cancer risk in women. Arch Intern Med 2007;167:1050–1059. ArticlePubMed
- 3. Kamen D, Aranow C. Vitamin D in systemic lupus erythematosus. Curr Opin Rheumatol 2008;20:532–537. ArticlePubMed
- 4. Judd SE, Tangpricha V. Vitamin D deficiency and risk for cardiovascular disease. Am J Med Sci 2009;338:40–44. ArticlePubMedPMC
- 5. Choi HS, Kim KA, Lim CY, Rhee SY, Hwang YC, Kim KM, et al. Low serum vitamin D is associated with high risk of diabetes in Korean adults. J Nutr 2011;141:1524–1528. ArticlePubMedPDF
- 6. Yu S, Fang H, Han J, Cheng X, Xia L, Li S, et al. The high prevalence of hypovitaminosis D in China: a multicenter vitamin D status survey. Medicine (Baltimore) 2015;94:e585. ArticlePubMedPMC
- 7. Rizzoli R, Eisman JA, Norquist J, Ljunggren O, Krishnarajah G, Lim SK, et al. Risk factors for vitamin D inadequacy among women with osteoporosis: an international epidemiological study. Int J Clin Pract 2006;60:1013–1019. ArticlePubMed
- 8. Anderson PH, May BK, Morris HA. Vitamin D metabolism: new concepts and clinical implications. Clin Biochem Rev 2003;24:13–26. PubMedPMC
- 9. Jurutka PW, Bartik L, Whitfield GK, Mathern DR, Barthel TK, Gurevich M, et al. Vitamin D receptor: key roles in bone mineral pathophysiology, molecular mechanism of action, and novel nutritional ligands. J Bone Miner Res 2007;22(Suppl 2):V2–V10. ArticlePubMed
- 10. Heikinheimo RJ, Inkovaara JA, Harju EJ, Haavisto MV, Kaarela RH, Kataja JM, et al. Annual injection of vitamin D and fractures of aged bones. Calcif Tissue Int 1992;51:105–110. ArticlePubMedPDF
- 11. Shimada T, Hasegawa H, Yamazaki Y, Muto T, Hino R, Takeuchi Y, et al. FGF-23 is a potent regulator of vitamin D metabolism and phosphate homeostasis. J Bone Miner Res 2004;19:429–435. ArticlePubMed
- 12. Liu S, Tang W, Zhou J, Stubbs JR, Luo Q, Pi M, et al. Fibroblast growth factor 23 is a counter-regulatory phosphaturic hormone for vitamin D. J Am Soc Nephrol 2006;17:1305–1315. ArticlePubMed
- 13. Seiler S, Reichart B, Roth D, Seibert E, Fliser D, Heine GH. FGF-23 and future cardiovascular events in patients with chronic kidney disease before initiation of dialysis treatment. Nephrol Dial Transplant 2010;25:3983–3989. ArticlePubMedPDF
- 14. Faul C, Amaral AP, Oskouei B, Hu MC, Sloan A, Isakova T, et al. FGF23 induces left ventricular hypertrophy. J Clin Invest 2011;121:4393–4408. ArticlePubMedPMC
- 15. Nasrallah MM, El-Shehaby AR, Salem MM, Osman NA, El Sheikh E, Sharaf El Din UA. Fibroblast growth factor-23 (FGF-23) is independently correlated to aortic calcification in haemodialysis patients. Nephrol Dial Transplant 2010;25:2679–2685. ArticlePubMedPDF
- 16. Turner C, Dalton N, Inaoui R, Fogelman I, Fraser WD, Hampson G. Effect of a 300,000-IU loading dose of ergocalciferol (vitamin D2) on circulating 1,25(OH)2-vitamin D and fibroblast growth factor-23 (FGF-23) in vitamin D insufficiency. J Clin Endocrinol Metab 2013;98:550–556. ArticlePubMed
- 17. Atkins GJ, Rowe PS, Lim HP, Welldon KJ, Ormsby R, Wijenayaka AR, et al. Sclerostin is a locally acting regulator of late-osteoblast/preosteocyte differentiation and regulates mineralization through a MEPE-ASARM-dependent mechanism. J Bone Miner Res 2011;26:1425–1436. ArticlePubMed
- 18. Morvan F, Boulukos K, Clement-Lacroix P, Roman Roman S, Suc-Royer I, Vayssiere B, et al. Deletion of a single allele of the Dkk1 gene leads to an increase in bone formation and bone mass. J Bone Miner Res 2006;21:934–945. ArticlePubMed
- 19. Sankaralingam A, Roplekar R, Turner C, Dalton RN, Hampson G. Changes in dickkopf-1 (DKK1) and sclerostin following a loading dose of vitamin D 2 (300,000 IU). J Osteoporos 2014;2014:682763ArticlePubMedPMCPDF
- 20. Acibucu F, Dokmetas HS, Acibucu DO, Kilicli F, Aydemir M, Cakmak E. Effect of vitamin D treatment on serum sclerostin level. Exp Clin Endocrinol Diabetes 2017;125:634–637. ArticlePubMedPDF
- 21. Chung YS, Chung DJ, Kang MI, Kim IJ, Koh JM, Min YK, et al. Vitamin D repletion in Korean postmenopausal women with osteoporosis. Yonsei Med J 2016;57:923–927. ArticlePubMedPMC
- 22. Burns J, Paterson CR. Single dose vitamin D treatment for osteomalacia in the elderly. Br Med J (Clin Res Ed) 1985;290:281–282.Article
- 23. Saito H, Maeda A, Ohtomo S, Hirata M, Kusano K, Kato S, et al. Circulating FGF-23 is regulated by 1alpha,25-dihydroxyvitamin D3 and phosphorus in vivo. J Biol Chem 2005;280:2543–2549. ArticlePubMed
- 24. Uzum AK, Salman S, Telci A, Boztepe H, Tanakol R, Alagol F, et al. Effects of vitamin D replacement therapy on serum FGF23 concentrations in vitamin D-deficient women in short term. Eur J Endocrinol 2010;163:825–831. ArticlePubMed
- 25. Wolf M, Koch TA, Bregman DB. Effects of iron deficiency anemia and its treatment on fibroblast growth factor 23 and phosphate homeostasis in women. J Bone Miner Res 2013;28:1793–1803. ArticlePubMed
- 26. Wolf M, White KE. Coupling fibroblast growth factor 23 production and cleavage: iron deficiency, rickets, and kidney disease. Curr Opin Nephrol Hypertens 2014;23:411–419. ArticlePubMedPMC
- 27. Romagnoli E, Mascia ML, Cipriani C, Fassino V, Mazzei F, D'Erasmo E, et al. Short and long-term variations in serum calciotropic hormones after a single very large dose of ergocalciferol (vitamin D2) or cholecalciferol (vitamin D3) in the elderly. J Clin Endocrinol Metab 2008;93:3015–3020. ArticlePubMedPDF
- 28. Gatti D, Viapiana O, Fracassi E, Idolazzi L, Dartizio C, Povino MR, et al. Sclerostin and DKK1 in postmenopausal osteoporosis treated with denosumab. J Bone Miner Res 2012;27:2259–2263. ArticlePubMed
- 29. Gatti D, Viapiana O, Adami S, Idolazzi L, Fracassi E, Rossini M. Bisphosphonate treatment of postmenopausal osteoporosis is associated with a dose dependent increase in serum sclerostin. Bone 2012;50:739–742. ArticlePubMed
References
(A) Serum calcium, (B) phosphorus concentrations, and (C) ratio of urine calcium to creatinine in subjects were measured before and after vitamin D3 or placebo treatment.
Serum 25-hydroxyvitamin D (25(OH)D) concentrations were increased at different time points after treatment. (A) All subjects, (B) subjects with baseline 25(OH)D below 10 ng/mL, (C) subjects with baseline levels of 10 to 20 ng/mL, and (D) the vitamin D group. aP<0.05; bP<0.01; cP<0.001.
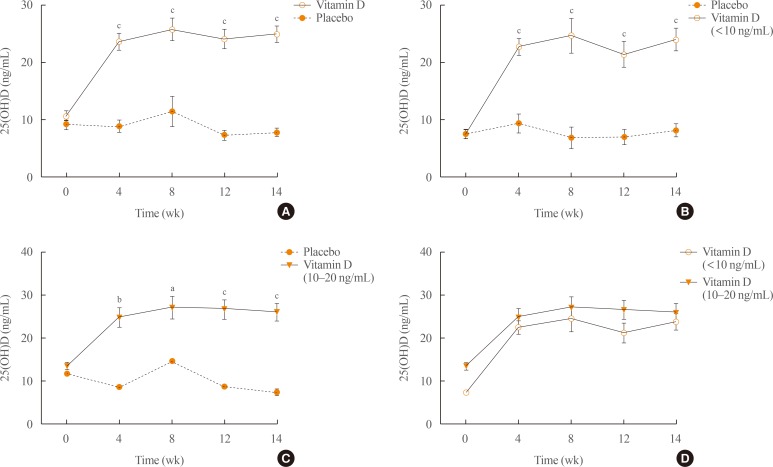
Serum parathyroid hormone (PTH) levels were detected at the different time points after treatment. The comparison in (A) all subjects, (B) the subjects with less 10 ng/mL of baseline 25-hydroxyvitamin D (25(OH)D), (C) the subjects with 10 to 20 ng/mL of baseline 25(OH)D, and (D) the vitamin D group.

Serum fibroblast growth factor 23 (FGF23) levels were measured at the indicated times. (A) All subjects, (B) subjects with baseline 25(OH)D below 10 ng/mL, (C) subjects with baseline levels of 10 to 20 ng/mL, and (D) the vitamin D group.
(A, B) Serum sclerostin and (C, D) dickkopf-1 (DKK1) concentrations were measured at different time points after treatment. Mean±SEM are shown. aP<0.05 compared to placebo.
Demographic and Other Baseline Characteristics of the Study Subjects
Figure & Data
References
Citations

- Effect of vitamin D supplementation on circulating fibroblast growth factor-23 concentration in adults with prediabetes
Lisa Ceglia, Anastassios G. Pittas, Bess Dawson-Hughes
Aging Clinical and Experimental Research.2023; 35(3): 525. CrossRef - Fibroblast Growth Factor 23 in COVID-19: An Observational Study
Athena Myrou, Theodoros Aslanidis, Keli Makedou, Athanasios Mitsianis, Aikaterini Thisiadou, Paraskevi Karalazou, Georgios Chatzopoulos, Anastasios Papadopoulos, Antonios Kalis, Dimitrios Giagkoulis, Fotios Lezgidis, Christos Savopoulos
Cureus.2023;[Epub] CrossRef - The effect of vitamin D supplementation on serum levels of fibroblast growth factor- 23: A systematic review and meta-analysis of randomized controlled trials
Fatemeh Meshkini, Sepideh Soltani, Cain C.T. Clark, Vivian Tam, David Meyre, Omid Toupchian, Sahar Saraf-Bank, Shima Abdollahi
The Journal of Steroid Biochemistry and Molecular Biology.2022; 215: 106012. CrossRef - Serum sclerostin levels in osteoporotic fracture patients
Erwin A. Gorter, Casper R. Reinders, Pieta Krijnen, Natasha M. Appelman-Dijkstra, Inger B. Schipper
European Journal of Trauma and Emergency Surgery.2022; 48(6): 4857. CrossRef - Clinical Utility of Preoperative Vitamin D3 Injection for Preventing Transient Hypocalcemia after Total Thyroidectomy
Kwangsoon Kim, Cho Rok Lee, Sang-Wook Kang, Jandee Lee, Jong Ju Jeong, Kee-Hyun Nam, Woong Youn Chung, Claudio Casella
International Journal of Endocrinology.2021; 2021: 1. CrossRef - The effect of vitamin D supplementation on fibroblast growth factor‐23 in patients with chronic kidney disease: A systematic review and meta‐analysis
Elmira Karimi, Sama Bitarafan, Seyed Mohammad Mousavi, Nikan Zargarzadeh, Pari Mokhtari, Jessie Hawkins, Alipasha Meysamie, Fariba Koohdani
Phytotherapy Research.2021; 35(10): 5339. CrossRef - Pharmacodynamics of Oral Cholecalciferol in Healthy Individuals with Vitamin D Deficiency: A Randomized Open-Label Study
Angelo Fassio, Davide Gatti, Maurizio Rossini, Camilla Benini, Elena Fracassi, Eugenia Bertoldo, Ombretta Viapiana, Stefano Milleri, Matteo Gatti, Giovanni Adami
Nutrients.2021; 13(7): 2293. CrossRef - Vitamin D Deficiency at Mid-Pregnancy Is Associated with a Higher Risk of Postpartum Glucose Intolerance in Women with Gestational Diabetes Mellitus
Kyung-Soo Kim, Seok Won Park, Yong-Wook Cho, Soo-Kyung Kim
Endocrinology and Metabolism.2020; 35(1): 97. CrossRef - Effects of vitamin D supplementation on bone turnover markers and other bone-related substances in subjects with vitamin D deficiency
Rolf Jorde, Astrid Kamilla Stunes, Julia Kubiak, Ragnar Joakimsen, Guri Grimnes, Per Medbøe Thorsby, Unni Syversen
Bone.2019; 124: 7. CrossRef - Vitamin D Enhances the Efficacy of Topical Artificial Tears in Patients With Dry Eye Disease
Jin Sun Hwang, Yoon Pyo Lee, Young Joo Shin
Cornea.2019; 38(3): 304. CrossRef

 KES
KES
 PubReader
PubReader Cite
Cite