Articles
- Page Path
- HOME > Endocrinol Metab > Volume 30(4); 2015 > Article
-
Original ArticleClinical Study Association between Bsm1 Polymorphism in Vitamin D Receptor Gene and Diabetic Retinopathy of Type 2 Diabetes in Korean Population
- Yong Joo Hong, Eun Seok Kang, Myoung Jin Ji, Hyung Jin Choi, Taekeun Oh, Sung-Soo Koong, Hyun Jeong Jeon
-
Endocrinology and Metabolism 2015;30(4):469-474.
DOI: https://doi.org/10.3803/EnM.2015.30.4.469
Published online: December 31, 2015
Department of Internal Medicine, Chungbuk National University College of Medicine, Cheongju, Korea.
- Corresponding author: Hyun Jeong Jeon. Division of Endocrinology and Metabolism, Department of Internal Medicine, Chungbuk National University College of Medicine, 776 1sunhwan-ro, Seowon-gu, Cheongju 28644, Korea. Tel: +82-43-269-6352, Fax: +82-43-273-3252, endoann@naver.com
Copyright © 2015 Korean Endocrine Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
-
Background
- Type 2 diabetes is one of the most common diseases with devastating complications. However, genetic susceptibility of diabetic complications has not been clarified. The vitamin D endocrine system is related with calcification and lipolysis, insulin secretion, and may be associated with many complicated disease including diabetes and cardiovascular disease. Recent studies reported that single nucleotide polymorphisms of vitamin D receptor (VDR) gene were associated with diabetic complications.
-
Methods
- In present study, we evaluated the association of BsmI polymorphism of VDR with diabetic complications in Korean diabetes patients. Total of 537 type 2 diabetic subjects from the Endocrinology Clinic of Chungbuk National University Hospital were investigated. Polymerase chain reaction-restriction fragment length polymorphism was used to test the genotype and allele frequency of BsmI (rs1544410; BB, Bb, bb) polymorphisms.
-
Results
- Mean age was 62.44±10.64 years and mean disease duration was 13.65±7.39 years. Patients with B allele (BB or Bb) was significantly associated with lower risk of diabetic retinopathy (severe non-proliferative diabetic retinopathy or proliferative retinopathy; 7.4%, 5/68) compared with patients without B allele (bb; 17.3%, 81/469; P=0.035). This association was also significant after adjusting for hemoglobin A1c level, body mass index, age, sex, and diabetes mellitus duration, concurrent dyslipidemia and hypertension (odds ratio, 2.99; 95% confidence interval, 1.08 to 8.29; P=0.035) in logistic regression analysis.
-
Conclusion
- Our findings suggest that B allele of Bsm1 polymorphism in VDR gene is associated with lower risk of diabetic retinopathy in type 2 diabetic patients. Bsm1 genotype could be used as a susceptibility marker to predict the risk of diabetes complication.
- Despite recent advances, diabetes is a complex condition to treat in clinical practice, and mortality remains high. Diabetic complications may play an important role in increasing mortality of diabetic patients. The mechanisms underlying the role of hyperglycemia in diabetic complications remain incompletely explained, which means potential involvement of genetic factors in diabetic complications. The vitamin D endocrine system is associated with various disease including diabetes, cancer, cardiovascular disorder, metabolic syndrome, autoimmune disorders, and tuber-culosis [1234]. Active vitamin D mediates its biological effects by binding to the vitamin D receptor (VDR), which is located in the nuclei of target cells. VDR is associated with insulin secretion and sensitivity, also it is expressed in the pancreas. These results show that VDR is a possible role in the development of diabetes.
- To date, more than 25 different polymorphisms have been mapped to the VDR locus. The human VDR gene is located on chromosome 12q13.1. It has at least 5 promoter regions, 8 protein-coding exons and 6 untranslated exons, which are alternatively spliced into FokI (in exon 2), BsmI and ApaI (both in intron 8), and TaqI (in exon 9). Recently, there are several reports these VDR polymorphisms are associated with type 2 diabetes, insulin secretion [5678910]. Also, VDR polymorphisms are related with metabolic syndrome, metabolic changes related with obesity and diabetic retinopathy. These studies suggested that VDR polymorphism may play a possible role in the development of diabetic complications. Although, there is insufficient studies of association between VDR polymorphism and Korean type 2 diabetes patients.
- In this study, we evaluate the association of VDR polymorphism with diabetic complications in Korean type 2 diabetes patients.
INTRODUCTION
- The study was designed as a case-control study, which enrolled 537 of patients with type 2 diabetes in Chungbuk National University Hospital. The study was approved by the International Review Board of Chungbuk National University Hospital and all patients gave informed consent prior to being included in the study.
- Diagnosis of type 2 diabetes was based on the World Health Organization criteria for diabetes. Diabetic retinopathy was assessed through dilated pupils by trained ophthalmologists and classified as no apparent diabetic retinopathy (mild to moderate, severe), non-proliferative diabetic retinopathy, or proliferative diabetic retinopathy, which were based on the International Clinical Retinopathy Severity Scale. Diabetic nephropathy was defined by the presence of microalbuminuria or overt albuminuria and elevated creatinine level. The macrovascular complications including coronary heart disease or stroke were assessed by the medical records. All subjects underwent biochemical tests, medical history and family history of diabetes. Patients were divided into two groups according to presence of B allele (BB or Bb genotype vs. bb genotype). We compared the clinical characteristics and prevalence of diabetic complications according to presence of B allele.
- Genotyping
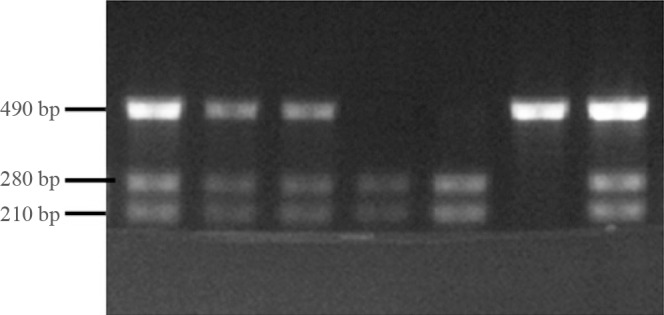
- The genomic DNA was isolated from peripheral leukocytes obtained from ethylenediaminetetraacetic acid-treated whole blood. Polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP) was used to determine the Bsm1 VDR gene polymorphism. The primers for VDR gene was designed based on previous literature. The following Bsm1 primers were used for amplification: forward 5'-CAACCAAGACTACAAGTACCGCGTCAGTGA-3´ and reverse 5'-AACCAGCGGAAGAGGTCAAGGG-3'. The PCR was conducted in a 25 µL volume, with an initial denaturation of 3 minutes at 94℃, followed by 30 seconds denaturation at 94℃, 30 seconds annealing at 62℃, and 1 minute extension at 72℃ for 30 cycles, and a final extension of 5 minutes at 72℃. The PCR products were digested overnight at 37℃ by Fermentas restriction enzymes, and resolved in 1.5% agarose gel ethidium bromide by electrophoresis for genotype analysis. Digestion with Bsm1 yields 3 genotypes: BB (825 bp), Bb (825, 650, 175 bp), and bb (650, 175 bp) (Fig. 1).
- Statistical analysis
- Hardy-Weinberg equilibrium was tested to evaluate the relationship between gene frequencies and genotype frequencies. The data was expressed as mean±standard deviation.
- The clinical characteristics of the patients were compared using the chi-square test for categorical variables and Student t test for continuous variables (Table 1). Multivariate analysis was using the logistic regression to adjust interaction effect of metabolic risk factors to diabetic complication outcomes (Table 2). P values of <0.05 were considered statistically significant. All statistical analyses were performed using SPSS version 15.0 (SPSS Inc., Chicago, IL, USA).
METHODS
- Clinical characteristics
- The clinical and biochemical characteristics of the patients are summarized in Table 1. The mean age of the patient population was 62.44±10.64 years, body mass index (BMI) was 25.02±3.64 kg/m2, duration of diabetes was 13.65±7.39 years. There was no difference in sex, duration of diabetes, family history of diabetes and concurrent hypertension, dyslipidemia in between two groups, but results showed significant difference in age (P=0.007), BMI (P=0.002) between two groups (Table 1).
- Bsm1 polymorphism and diabetic retinopathy
- The genotypic distribution of Bsm1 met Hardy-Weinberg equilibrium. The frequency of Bsm1 genotype in patients was as follows: BB, 1.86% (n=10); Bb, 10.80% (n=58); bb, 87.34% (n=469) (Table 3). Patients with BB or Bb genotype was significantly associated with lower risk of diabetic retinopathy compared with patients with bb genotype (BB or Bb genotype 7.4% vs. bb genotype 82.7%, P=0.035) (Table 4).
- In logistic regression analysis, B allele was associated with the lower risk of diabetic retinopathy in this study (odds ratio, 2.63; P=0.037). And this association continued after adjusted with other clinical factors (Table 4). Multivariate logistic regression analysis with an enter method was used to evaluate the effect of other clinical factors. In baseline characteristics, there was significant difference with age and BMI between two groups, but after adjustment with these factors, the association with diabetic retinopathy and Bsm1 type still continued (Table 4).
- The retinopathy divided to two groups, one is no apparent diabetic retinopathy and mild to moderate non-proliferative retinopathy and the other is severe non-proliferative retinopathy and proliferative retinopathy. Because of some confused description of severities of non-proliferative retinopathy (mild, moderate), but severe retinopathy and proliferative retinopathy may lead to significant consequence (laser treatment or surgery), so this two groups' separation was clinically significant.
- Bsm1 polymorphism and diabetic microvascular and macrovascular complications
- We also evaluate the association of Bsm1 polymorphism and diabetic nephropathy, neuropathy, diabetic foot, coronary artery disease, stroke, and peripheral artery disease. There was no association between the Bsm1 polymorphism and diabetic macrovascular complication, nephropathy, neuropathy, and diabetic foot (Table 2).
RESULTS
- We evaluated the association of Bsm1 polymorphism with diabetic complications in Korean type 2 diabetic patients. Patients with B allele (BB or Bb) were significantly associated with lower risk of diabetic retinopathy compared with patients without B allele (bb). It showed that B allele in VDR may be a protective factor in the development of diabetic retinopathy. There are a few data about the association with Bsm1 polymorphism and diabetic retinopathy. Bucan et al. [11] showed that bb genotype in VDR has a higher risk of developing diabetic retinopathy. But they didn't show that the association of b allele and diabetic retinopathy. Although Capoluongo et al. [12] did not find Bsm1 association with diabetic complications in the Italian population. Cyganek et al. [5] also reported the same results that no association was observed between the Bsm1 polymorphism and diabetic retinopathy. The relationship of Bsm1 polymorphism with diabetic retinopathy is still controversial. The discrepancies of these results were likely due to the difference in ethnicity or enrolled characteristics of subject among studies.
- Diabetic microvascular complications result from several mechanisms including hyperglycemia, hypertension, seem to be also affected by genetic factors. There are several genes about the development of diabetic microvascular complications including endothelial physiology, vasoconstriction, coagulation, oxidative processes, and growth factors. Vitamin D is well known for maintaining normal homeostasis of calcium and phosphorus also has anti-proliferative, anti-angiogenic effect and apoptosis process. The active form of this vitamin D through a specific VDR, VDR is widely expressed in human tissues and organs, including retina. As mentioned earlier, vitamin D has anti-proliferative action in vitro; vitamin D inhibits vascular smooth muscle cell growth and effects on endothelial physiology. Therefore, it is postulated that VDR polymorphism may influence on the development of diabetic retinopathy.
- In this study, we demonstrated of possible association with B allele and lower risk of diabetic retinopathy. And this result was about Korean population, there was no previous study about association with diabetic complication and VDR polymorphisms.
- We try to evaluate another association in diabetic complication, such as nephropathy, macrovascular complications. There was no association between Bsm1 polymorphism and diabetic nephropathy (end-stage renal disease, P=0.580; normo-/micro-/macroalbuminuria, P=0.302). Zhang et al. [8] reported that B allele in Bsm1 is correlated with large amount albuminuria in the Han Chinese population with type 2 diabetes, and is probably a risk factor for early-onset diabetic nephropathy. Contrary to our result, this study showed that B allele could be responsible for the pathogenesis of early-onset diabetic nephropathy. Bucan et al. [11] showed that no association was observed between Bsm1 genotype and diabetic nephropathy, in common with our data. Marco et al. [13] reported that the greater prevalence of ischemic heart disease and death from cardiovascular causes in BB and Bb genotypes. They suggested that the Bsm1 VDR polymorphism might affect cardiovascular morbidity or mortality through calcium metabolism or VDR mediated influence of 1, 25-dihydroxyvitmain D3 on vascular smooth muscle cells and myocytes. Gyorffy et al. [14] showed that patient with the BB genotype in VDR have a higher risk of development in coronary artery disease. In our study, there was no association between the Bsm1 polymorphism and diabetic macrovascular complications especially myocardiac infarction (Table 2).
- These two contrary results (lower risk of retinopathy in B allele in our study, higher risk of coronary artery disease in B allele in other studies) may explained by biochemical features of Bsm1 polymorphism. Polymorphism for Bsm1 was known functional effects because it is located within intron 8 which will be removed during mRNA posttranscriptional. Although, VDR itself may not be the disease affecting locus, but rather a marker locus in linkage disequilibrium with the residual locus, and the discrepant findings may reflect variable strengths of linkage disequilibrium in different populations [15]. Another possible mechanism is that alternation of Bsm1 in intronic sequence may influence protein expression [14].
- There are several hypotheses for development of metabolic disorder in alternation of vitamin D system. First, vitamin D is fat soluble and can be stored in adipose tissue. Second, large cohort studies have been reported that high BMI is strongly and inversely correlated with serum vitamin D level. Third, vitamin D regulates calcium trafficking in pancreatic β-cells in vitro. Also, vitamin D effects intracellular ionized calcium signaling in the adipocyte, which in turn induces increased lipogenesis and decreased lipolysis via uncoupling protein-2. These mechanisms postulated that alternation of the VDR potentially contributes to the development of metabolic syndrome and cardiovascular disease. Unfortunately, our patients have only few data with serum vitamin D level, so we couldn't evaluate the association with vitamin D level and other results.
- In present study, we suggested that Bsm1 polymorphism in VDR gene is associated with lower risk of diabetic retinopathy in type 2 diabetic patients. Bsm1 polymorphism in VDR gene could be used as a susceptibility marker to predict the risk of diabetic complication. But further large study will be needed.
DISCUSSION
-
Acknowledgements
- This work was supported by a research fund of Daejeon and Chungcheong Branch of Korean Endocrine Society. The biospecimens for this study were provided by the Chungbuk National University Hospital, a member of the National Biobank of Korea, which is supported by the Ministry of Health, Welfare and Family Affairs. All samples derived from the National Biobank of Korea were obtained with informed consent under Institutional Review Board-approved protocols.
ACKNOWLEDGMENTS
-
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.
Article information
- 1. Tamilselvan B, Seshadri KG, Venkatraman G. Role of vitamin D on the expression of glucose transporters in L6 myotubes. Indian J Endocrinol Metab 2013;17(Suppl 1):S326–S328. PubMedPMC
- 2. Kahn BB. Facilitative glucose transporters: regulatory mechanisms and dysregulation in diabetes. J Clin Invest 1992;89:1367–1374. ArticlePubMedPMC
- 3. Vaidya A, Williams JS. The relationship between vitamin D and the renin-angiotensin system in the pathophysiology of hypertension, kidney disease, and diabetes. Metabolism 2012;61:450–458. ArticlePubMed
- 4. Riek AE, Oh J, Sprague JE, Timpson A, de las Fuentes L, Bernal-Mizrachi L, et al. Vitamin D suppression of endoplasmic reticulum stress promotes an antiatherogenic monocyte/macrophage phenotype in type 2 diabetic patients. J Biol Chem 2012;287:38482–38494. ArticlePubMedPMC
- 5. Cyganek K, Mirkiewicz-Sieradzka B, Malecki MT, Wolkow P, Skupien J, Bobrek J, et al. Clinical risk factors and the role of VDR gene polymorphisms in diabetic retinopathy in Polish type 2 diabetes patients. Acta Diabetol 2006;43:114–119. ArticlePubMedPDF
- 6. Nosratabadi R, Arababadi MK, Salehabad VA, Shamsizadeh A, Mahmoodi M, Sayadi AR, et al. Polymorphisms within exon 9 but not intron 8 of the vitamin D receptor are associated with the nephropathic complication of type-2 diabetes. Int J Immunogenet 2010;37:493–497. ArticlePubMed
- 7. Velayoudom-Cephise FL, Larifla L, Donnet JP, Maimaitiming S, Deloumeaux J, Blanchet A, et al. Vitamin D deficiency, vitamin D receptor gene polymorphisms and cardiovascular risk factors in Caribbean patients with type 2 diabetes. Diabetes Metab 2011;37:540–545. ArticlePubMed
- 8. Zhang H, Wang J, Yi B, Zhao Y, Liu Y, Zhang K, et al. BsmI polymorphisms in vitamin D receptor gene are associated with diabetic nephropathy in type 2 diabetes in the Han Chinese population. Gene 2012;495:183–188. ArticlePubMed
- 9. Ferrarezi DA, Bellili-Munoz N, Dubois-Laforgue D, Cheurfa N, Lamri A, Reis AF, et al. Allelic variations of the vitamin D receptor (VDR) gene are associated with increased risk of coronary artery disease in type 2 diabetics: the DIABHYCAR prospective study. Diabetes Metab 2013;39:263–270. ArticlePubMed
- 10. Yokoyama K, Nakashima A, Urashima M, Suga H, Mimura T, Kimura Y, et al. Interactions between serum vitamin D levels and vitamin D receptor gene FokI polymorphisms for renal function in patients with type 2 diabetes. PLoS One 2012;7:e51171ArticlePubMedPMC
- 11. Bucan K, Ivanisevic M, Zemunik T, Boraska V, Skrabic V, Vatavuk Z, et al. Retinopathy and nephropathy in type 1 diabetic patients: association with polymorphysms of vitamin D-receptor, TNF, neuro-D and IL-1 receptor 1 genes. Coll Antropol 2009;33(Suppl 2):99–105.
- 12. Capoluongo E, Pitocco D, Concolino P, Santonocito C, Di Stasio E, d'Onofrio G, et al. Slight association between type 1 diabetes and "ff" VDR FokI genotype in patients from the Italian Lazio Region. Lack of association with diabetes complications. Clin Biochem 2006;39:888–892. ArticlePubMed
- 13. Marco MP, Craver L, Betriu A, Fibla J, Fernandez E. Influence of vitamin D receptor gene polymorphisms on mortality risk in hemodialysis patients. Am J Kidney Dis 2001;38:965–974. ArticlePubMed
- 14. Gyorffy B, Vasarhelyi B, Krikovszky D, Madacsy L, Tordai A, Tulassay T, et al. Gender-specific association of vitamin D receptor polymorphism combinations with type 1 diabetes mellitus. Eur J Endocrinol 2002;147:803–808. ArticlePubMed
- 15. Tawfeek MA, Habib FA, Mouhamed Saultan EE. Vitamin D receptor Bsm1 gene polymorphisms and gestational diabetes mellitus: a Saudi study. Br J Med Med Res 2011;1:459–468.ArticlePDF
References
Clinical Characteristics of Patients of Type 2 Diabetes according to Bsm1 Genotype
Values are expressed as mean±SD or number (%).
BMI, body mass index; DM, diabetes mellitus; PP2, post-prandial 2 hours blood glucose; HbA1c, hemoglobin A1c; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; BUN, blood urea nitrogen; eGFR, estimated glomerular filtration rate; uACR, urinary microalbumin-creatinine ratio.
Prevalence of Other Diabetic Microvascular/Macrovascular Complications according to Bsm1 Genotype
| Variable | BB+Bb | bb | P value |
|---|---|---|---|
| ESRDa | 0 | 4 (9) | 0.580 |
| Polyneuropathyb | 33 (48.5) | 233 (49.7) | 0.979 |
| Strokec | 5 (7.4) | 50 (10.7) | 0.273 |
| MI | 4 (5.9) | 14 (3.0) | 0.183 |
| PADd | 0 | 6 (1.3) | 0.442 |
| DM foot | 2 (2.9) | 18 (3.8) | 0.524 |
Values are expressed as number (%).
ESRD, end-stage renal disease; MI, myocardiac infarction; PAD, peripheral artery disease; DM, diabetes mellitus. aPatient who have kidney replacement therapy; bPatient who have anti-neuralgia medicine; cCerebral ischemic event including transient ischemic attack; dAbove 50% arterial stenosis confirmed by angiography.
Distribution of Bsm1 Genotypes in Type 2 Diabetic Patients
| No. (%) | |
|---|---|
| Total | 537 (100) |
| BB | 10 (1.86) |
| Bb | 58 (10.80) |
| bb | 469 (87.34) |
Logistic Analysis of Multiple Risk Factors for the Association with Diabetic Retinopathy according to Bsm1 Genotype
Figure & Data
References
Citations

- The Vitamin D Receptor Bsm1 Variant is not Associated With Temporomandibular Disorder With or Without Bruxism
Serkan YILDIZ, Serbülent YİĞİT, Ayşe Feyda NURSAL, Nevin KARAKUŞ, Mehmet Kemal TÜMER
ADO Klinik Bilimler Dergisi.2024; 13(1): 100. CrossRef - Association analysis between the VDR gene variants and type 2 diabetes
Shabnam Salehizadeh, Sara Ramezani, Mojgan Asadi, Mahdi Afshari, Seyed Hamid Jamaldini, Farhad Adhami Moghadam, Mandana Hasanzad
Journal of Diabetes & Metabolic Disorders.2023;[Epub] CrossRef - Metabolic impact of the VDR rs1544410 in diabetic retinopathy
Caroline Severo de Assis, Tainá Gomes Diniz, João Otávio Scarano Alcântara, Vanessa Polyana Alves de Sousa Brito, Rayner Anderson Ferreira do Nascimento, Mayara Karla dos Santos Nunes, Alexandre Sérgio Silva, Isabella Wanderley de Queiroga Evangelista, Ma
PLOS ONE.2022; 17(2): e0263346. CrossRef - Vitamin D, the Vitamin D Receptor, Calcitriol Analogues and Their Link with Ocular Diseases
Miłosz Caban, Urszula Lewandowska
Nutrients.2022; 14(11): 2353. CrossRef - Vitamin D-Related Single Nucleotide Polymorphisms as Risk Biomarker of Cardiovascular Disease
Paula González Rojo, Cristina Pérez Ramírez, José María Gálvez Navas, Laura Elena Pineda Lancheros, Susana Rojo Tolosa, María del Carmen Ramírez Tortosa, Alberto Jiménez Morales
International Journal of Molecular Sciences.2022; 23(15): 8686. CrossRef - Effects of Vitamin D Receptor Genotype on Lipid Profiles and Retinopathy Risk in Type 2 Diabetes Patients: A Pilot Study
Hussam Alhawari, Yazun Jarrar, Dina Abulebdah, Sara J. Abaalkhail, Marah Alkhalili, Sura Alkhalili, Hussein Alhawari, Munther Momani, Mohammed N. Obeidat, Rand K. Fram, Mohammad A. Salahat, Su-Jun Lee
Journal of Personalized Medicine.2022; 12(9): 1488. CrossRef - Relation of vitamin D and BsmI variant with temporomandibular diseases in the Turkish population
S. Yildiz, M.K. Tumer, S. Yigit, A.F. Nursal, A. Rustemoglu, Y. Balel
British Journal of Oral and Maxillofacial Surgery.2021; 59(5): 555. CrossRef - The relationship between vitamin D receptor (VDR) rs2228570 and rs7975232 genetic variants and the risk of recurrent pregnancy loss
Zohreh Salari, Nasrollah Saleh-Gohari, Monire Rezapour, Ahamad Khosravi, Hadi Tavakkoli, Ehsan Salarkia, Fatemeh Karami-Robati
Meta Gene.2021; 27: 100833. CrossRef - Association of Bone Metabolism Indices and Bone Mineral Density with Diabetic Retinopathy in Elderly Patients with Type 2 Diabetes Mellitus: A Cross-Sectional Inpatient Study in China
Xin Zhao, Lili Huo, Xiaofeng Yu, Xiaomei Zhang, Fabio Vescini
Journal of Diabetes Research.2021; 2021: 1. CrossRef - The Associations Between Vitamin D Receptor BsmI and ApaI Polymorphisms and Obesity in Korean Patients with Type 2 Diabetes Mellitus
Sang Won Nam, Jinwoo Choi, Hyun Jeong Jeon, Tae Keun Oh, Dong-Hwa Lee
Diabetes, Metabolic Syndrome and Obesity: Targets and Therapy.2021; Volume 14: 557. CrossRef - Vitamin D Deficiency at Mid-Pregnancy Is Associated with a Higher Risk of Postpartum Glucose Intolerance in Women with Gestational Diabetes Mellitus
Kyung-Soo Kim, Seok Won Park, Yong-Wook Cho, Soo-Kyung Kim
Endocrinology and Metabolism.2020; 35(1): 97. CrossRef - The role of vitamin D in the pathogenesis and treatment of diabetes mellitus: a narrative review
Maria Grammatiki, Spiros Karras, Kalliopi Kotsa
Hormones.2019; 18(1): 37. CrossRef - Vitamin D-binding protein and vitamin D receptor genotypes and 25-hydroxyvitamin D levels are associated with development of aortic and mitral valve calcification and coronary artery diseases
Amir Kiani, Ehsan Mohamadi-Nori, Asad Vaisi-Raygani, Maryam Tanhapour, Said Elahi-Rad, Fariborz Bahrehmand, Zohreh Rahimi, Tayebeh Pourmotabbed
Molecular Biology Reports.2019; 46(5): 5225. CrossRef - Is Hypovitaminosis D Related to Incidence of Type 2 Diabetes and High Fasting Glucose Level in Healthy Subjects: A Systematic Review and Meta-Analysis of Observational Studies
Shamaila Rafiq, Per Jeppesen
Nutrients.2018; 10(1): 59. CrossRef - Association between the vitamin D receptor gene polymorphisms and diabetic nephropathy risk: A meta‐analysis
Fang Yin, Jing Liu, Ming‐Xiu Fan, Xiao‐Li Zhou, Xiao‐Ling Zhang
Nephrology.2018; 23(2): 107. CrossRef - BSMI single nucleotide polymorphism in vitamin D receptor gene is associated with decreased circulatory levels of serum 25-hydroxyvitamin D among micro and macrovascular complications of type 2 diabetes mellitus
Krishnamoorthy Ezhilarasi, Umapathy Dhamodharan, Viswanathan Vijay
International Journal of Biological Macromolecules.2018; 116: 346. CrossRef - Diabetic macular oedema: under‐represented in the genetic analysis of diabetic retinopathy
Suzanne Broadgate, Christine Kiire, Stephanie Halford, Victor Chong
Acta Ophthalmologica.2018; 96(A111): 1. CrossRef - Expression of JAZF1, ABCC8, KCNJ11and Notch2 genes and vitamin D receptor polymorphisms in type 2 diabetes, and their association with microvascular complications
Maha A. Rasheed, Nagwa Kantoush, Nagwa Abd El-Ghaffar, Hebatallah Farouk, Solaf Kamel, Alshaymaa Ahmed Ibrahim, Aliaa Shalaby, Eman Mahmoud, Hala M. Raslan, Omneya M. Saleh
Therapeutic Advances in Endocrinology and Metabolism.2017; 8(6): 97. CrossRef - Vitamin D and diabetes mellitus: Causal or casual association?
M. Grammatiki, E. Rapti, S. Karras, R. A. Ajjan, Kalliopi Kotsa
Reviews in Endocrine and Metabolic Disorders.2017; 18(2): 227. CrossRef - The Association between Vitamin D Deficiency and Diabetic Retinopathy in Type 2 Diabetes: A Meta-Analysis of Observational Studies
Bang-An Luo, Fan Gao, Lu-Lu Qin
Nutrients.2017; 9(3): 307. CrossRef - The Association between VDR Gene Polymorphisms and Diabetic Retinopathy Susceptibility: A Systematic Review and Meta-Analysis
Yun Zhang, Wei Xia, Ping Lu, Huijuan Yuan
BioMed Research International.2016; 2016: 1. CrossRef - Candidate gene studies of diabetic retinopathy in human
Petra Priščáková, Gabriel Minárik, Vanda Repiská
Molecular Biology Reports.2016; 43(12): 1327. CrossRef

 KES
KES

 PubReader
PubReader Cite
Cite