Search
- Page Path
- HOME > Search
- Thyroid
- Recent Improvements in Genomic and Transcriptomic Understanding of Anaplastic and Poorly Differentiated Thyroid Cancers
- Seong-Keun Yoo, Young Shin Song, Young Joo Park, Jeong-Sun Seo
- Endocrinol Metab. 2020;35(1):44-54. Published online March 19, 2020
- DOI: https://doi.org/10.3803/EnM.2020.35.1.44
- 7,293 View
- 246 Download
- 16 Web of Science
- 18 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
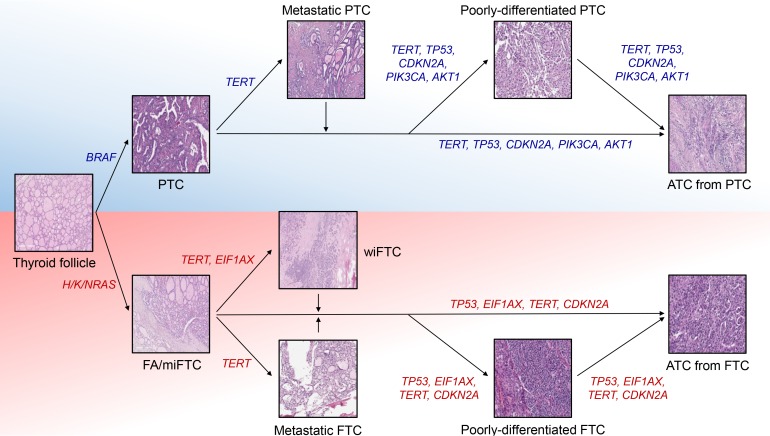
ePub Anaplastic thyroid cancer (ATC) is a lethal human cancer with a 5-year survival rate of less than 10%. Recently, its genomic and transcriptomic characteristics have been extensively elucidated over 5 years owing to advance in high throughput sequencing. These efforts have extended molecular understandings into the progression mechanisms and therapeutic vulnerabilities of aggressive thyroid cancers. In this review, we provide an overview of genomic and transcriptomic alterations in ATC and poorly-differentiated thyroid cancer, which are distinguished from differentiated thyroid cancers. Clinically relevant genomic alterations and deregulated signaling pathways will be able to shed light on more effective prevention and stratified therapeutic interventions for affected patients.
-
Citations
Citations to this article as recorded by- The genomic and evolutionary landscapes of anaplastic thyroid carcinoma
Peter Y.F. Zeng, Stephenie D. Prokopec, Stephen Y. Lai, Nicole Pinto, Michelle A. Chan-Seng-Yue, Roderick Clifton-Bligh, Michelle D. Williams, Christopher J. Howlett, Paul Plantinga, Matthew J. Cecchini, Alfred K. Lam, Iram Siddiqui, Jianxin Wang, Ren X.
Cell Reports.2024; 43(3): 113826. CrossRef - Comparative study between poorly differentiated thyroid cancer and anaplastic thyroid cancer: real-world pathological distribution, death attribution, and prognostic factor estimation
Kun Zhang, Xinyi Wang, Tao Wei, Zhihui Li, Jingqiang Zhu, Ya-Wen Chen
Frontiers in Endocrinology.2024;[Epub] CrossRef - Dabrafenib and Trametinib Therapy for Advanced Anaplastic Thyroid Cancer – Real-World Outcomes From UK Centres
C. Lorimer, L. Cheng, R. Chandler, K. Garcez, V. Gill, K. Graham, W. Grant, S. Sardo Infirri, J. Wadsley, L. Wall, N. Webber, K.H. Wong, K. Newbold
Clinical Oncology.2023; 35(1): e60. CrossRef - MicroRNA expression profiling of RAS-mutant thyroid tumors with follicular architecture: microRNA signatures to discriminate benign from malignant lesions
E. Macerola, A. M. Poma, P. Vignali, A. Proietti, L. Torregrossa, C. Ugolini, A. Basolo, A. Matrone, R. Elisei, F. Santini, F. Basolo
Journal of Endocrinological Investigation.2023; 46(8): 1651. CrossRef - RBX1 regulates PKM alternative splicing to facilitate anaplastic thyroid carcinoma metastasis and aerobic glycolysis by destroying the SMAR1/HDAC6 complex
Debin Xu, Jichun Yu, Yuting Yang, Yunyan Du, Hongcheng Lu, Shouhua Zhang, Qian Feng, Yi Yu, Liang Hao, Jun Shao, Leifeng Chen
Cell & Bioscience.2023;[Epub] CrossRef - Multi-Omics and Management of Follicular Carcinoma of the Thyroid
Thifhelimbilu Emmanuel Luvhengo, Ifongo Bombil, Arian Mokhtari, Maeyane Stephens Moeng, Demetra Demetriou, Claire Sanders, Zodwa Dlamini
Biomedicines.2023; 11(4): 1217. CrossRef - Genomic landscape of anaplastic thyroid cancer and implications on therapy
Leslie Cheng, Kate Newbold
Current Opinion in Endocrine and Metabolic Research.2023; 30: 100458. CrossRef - Modeling the tumor microenvironment of anaplastic thyroid cancer: an orthotopic tumor model in C57BL/6 mice
Zhen Xu, Hyo Shik Shin, Yoo Hyung Kim, Seong Yun Ha, Jae-Kyung Won, Su-jin Kim, Young Joo Park, Sareh Parangi, Sun Wook Cho, Kyu Eun Lee
Frontiers in Immunology.2023;[Epub] CrossRef - Characterization of the genomic alterations in poorly differentiated thyroid cancer
Yeeun Lee, SeongRyeol Moon, Jae Yeon Seok, Joon-Hyop Lee, Seungyoon Nam, Yoo Seung Chung
Scientific Reports.2023;[Epub] CrossRef - Genetica del carcinoma anaplastico della tiroide
Teresa Ramone, Chiara Mulè, Cristina Romei, Raffaele Ciampi, Eleonora Molinaro, Rossella Elisei
L'Endocrinologo.2022; 23(2): 208. CrossRef - Tissue and circulating PD-L2: moving from health and immune-mediated diseases to head and neck oncology
Elena Muraro, Rebecca Romanò, Giuseppe Fanetti, Emanuela Vaccher, Irene Turturici, Valentina Lupato, Fabio Biagio La Torre, Jerry Polesel, Elisabetta Fratta, Vittorio Giacomarra, Giovanni Franchin, Agostino Steffan, Michele Spina, Salvatore Alfieri
Critical Reviews in Oncology/Hematology.2022; 175: 103707. CrossRef - Recent Improvements in the Treatment of High-Risk Thyroid Cancer
Eun Kyung Lee
Korean Society for Head and Neck Oncology.2022; 38(1): 1. CrossRef - Inhibition of Glycogen Metabolism Induces Reactive Oxygen Species-Dependent Cytotoxicity in Anaplastic Thyroid Cancer in Female Mice
Cole D Davidson, Jennifer A Tomczak, Eyal Amiel, Frances E Carr
Endocrinology.2022;[Epub] CrossRef - Survival prognostic factors for differentiated thyroid cancer patients with pulmonary metastases: A systematic review and meta-analysis
Hao Zhao, Chun-Hao Liu, Yue Cao, Li-Yang Zhang, Ya Zhao, Yue-Wu Liu, Hong-Feng Liu, Yan-Song Lin, Xiao-Yi Li
Frontiers in Oncology.2022;[Epub] CrossRef - 2021 American Thyroid Association Guidelines for Management of Patients with Anaplastic Thyroid Cancer
Keith C. Bible, Electron Kebebew, James Brierley, Juan P. Brito, Maria E. Cabanillas, Thomas J. Clark, Antonio Di Cristofano, Robert Foote, Thomas Giordano, Jan Kasperbauer, Kate Newbold, Yuri E. Nikiforov, Gregory Randolph, M. Sara Rosenthal, Anna M. Saw
Thyroid.2021; 31(3): 337. CrossRef - Intratumoral Heterogeneity in Differentiated Thyroid Tumors: An Intriguing Reappraisal in the Era of Personalized Medicine
Antonio Ieni, Roberto Vita, Cristina Pizzimenti, Salvatore Benvenga, Giovanni Tuccari
Journal of Personalized Medicine.2021; 11(5): 333. CrossRef - The Role of Altered Mitochondrial Metabolism in Thyroid Cancer Development and Mitochondria-Targeted Thyroid Cancer Treatment
Siarhei A. Dabravolski, Nikita G. Nikiforov, Alexander D. Zhuravlev, Nikolay A. Orekhov, Liudmila M. Mikhaleva, Alexander N. Orekhov
International Journal of Molecular Sciences.2021; 23(1): 460. CrossRef - Mechanisms of TERT Reactivation and Its Interaction with BRAFV600E
Young Shin Song, Young Joo Park
Endocrinology and Metabolism.2020; 35(3): 515. CrossRef
- The genomic and evolutionary landscapes of anaplastic thyroid carcinoma

- Thyroid
- Combined Effects of Baicalein and Docetaxel on Apoptosis in 8505c Anaplastic Thyroid Cancer Cells via Downregulation of the ERK and Akt/mTOR Pathways
- Chan Ho Park, Se Eun Han, Il Seong Nam-Goong, Young Il Kim, Eun Sook Kim
- Endocrinol Metab. 2018;33(1):121-132. Published online March 21, 2018
- DOI: https://doi.org/10.3803/EnM.2018.33.1.121
- 5,071 View
- 64 Download
- 41 Web of Science
- 35 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub Background Anaplastic thyroid cancer (ATC) is one of the most lethal human malignancies. Docetaxel, a microtubule stabilizer, is a common chemotherapeutic agent used to treat various metastatic cancers. However, prolonged use results in various side effects and drug resistance. Flavonoids, such as baicalein, are accepted chemotherapeutic and dietary chemopreventive agents with many advantages, such as greater accessibility, affordability, and lower toxicity, compared with traditional chemotherapy agents. In this study, we evaluated whether baicalein enhances the effects of docetaxel on apoptosis and metastasis in 8505c ATC cells.
Methods The 8505c cells were treated with baicalein or docetaxel individually and in combination. Cell viability was measured by MTT (thiazolyl blue tetrazolium bromide) assay, and apoptosis was detected by fluorescence microscopy of Hoechst-stained cells. The expression of apoptotic (Bax and caspase-3), anti-apoptotic (Bcl-2), angiogenic (vascular endothelial growth factor [VEGF], transforming growth factor β [TGF-β], E-cadherin, and N-cadherin), and signaling (extracellular signal-regulated kinase [ERK] mitogen activated protein kinase [MAPK], Akt, and mammalian target of rapamycin [mTOR]) proteins was determined by Western blot analysis.
Results The combination of baicalein (50 or 100 µM) and docetaxel (10 nM) significantly inhibited proliferation and induced apoptosis compared with monotherapies. The combination treatment significantly inhibited the expression of Bax, caspase-3, VEGF, TGF-β1, E-cadherin, N-cadherin, and mTOR, but decreased the expression of Bcl-2 and significantly decreased the phosphorylation of ERK and Akt.
Conclusion The combination of baicalein and docetaxel effectively induced apoptosis and inhibited metastasis in 8505c cells through downregulation of apoptotic and angiogenic protein expression and blocking of the ERK and Akt/mTOR pathways in 8505c cells. These results suggest that baicalein enhances the anticancer effects of docetaxel in ATC.
-
Citations
Citations to this article as recorded by- Modulation of hypoxia-inducible factor-1 signaling pathways in cancer angiogenesis, invasion, and metastasis by natural compounds: a comprehensive and critical review
Sajad Fakhri, Seyed Zachariah Moradi, Farahnaz Faraji, Leila Kooshki, Kassidy Webber, Anupam Bishayee
Cancer and Metastasis Reviews.2024; 43(1): 501. CrossRef - Baicalein Alleviates Arsenic-induced Oxidative Stress through
Activation of the Keap1/Nrf2 Signalling Pathway in Normal Human
Liver Cells
Qi Wang, Aihua Zhang
Current Molecular Medicine.2024; 24(3): 355. CrossRef - Natural products reverse cancer multidrug resistance
Jia-Yu Zou, Qi-Lei Chen, Xiao-Ci Luo, Davaadagva Damdinjav, Usama Ramadan Abdelmohsen, Hong-Yan Li, Tungalag Battulga, Hu-Biao Chen, Yu-Qing Wang, Jian-Ye Zhang
Frontiers in Pharmacology.2024;[Epub] CrossRef - Identification of novel characteristic biomarkers and immune infiltration profile for the anaplastic thyroid cancer via machine learning algorithms
C. Li, X. Dong, Q. Yuan, G. Xu, Z. Di, Y. Yang, J. Hou, L. Zheng, W. Chen, G. Wu
Journal of Endocrinological Investigation.2023; 46(8): 1633. CrossRef - Promising Role of the Scutellaria baicalensis Root Hydroxyflavone–Baicalein in the Prevention and Treatment of Human Diseases
Marcelina Chmiel, Monika Stompor-Gorący
International Journal of Molecular Sciences.2023; 24(5): 4732. CrossRef - STEAP1 Knockdown Decreases the Sensitivity of Prostate Cancer Cells to Paclitaxel, Docetaxel and Cabazitaxel
Sandra M. Rocha, Daniel Nascimento, Rafaella S. Coelho, Ana Margarida Cardoso, Luís A. Passarinha, Sílvia Socorro, Cláudio J. Maia
International Journal of Molecular Sciences.2023; 24(7): 6643. CrossRef - Baicalein as Promising Anticancer Agent: A Comprehensive Analysis on Molecular Mechanisms and Therapeutic Perspectives
A K M Helal Morshed, Supti Paul, Arafat Hossain, Tuli Basak, Md. Sanower Hossain, Md. Mehedi Hasan, Md. Al Hasibuzzaman, Tanjim Ishraq Rahaman, Md. Abdur Rashid Mia, Pollob Shing, Md Sohel, Shabana Bibi, Dipta Dey, Partha Biswas, Md. Nazmul Hasan, Long Ch
Cancers.2023; 15(7): 2128. CrossRef - The role of flavonoids in the regulation of epithelial‐mesenchymal transition in cancer: A review on targeting signaling pathways and metastasis
Carina Proença, Marisa Freitas, Daniela Ribeiro, Ana T. Rufino, Eduarda Fernandes, José Miguel P. Ferreira de Oliveira
Medicinal Research Reviews.2023; 43(6): 1878. CrossRef - Developing a ternary conductive hydrogel of polyacrylamide, polyaniline, and carbon nanotube: A potential chemiresistive gas sensor
Mohsen Peykari, Saeed Pourmahdian
Journal of Composite Materials.2023; 57(27): 4291. CrossRef - Inhibiting MEK1 R189 citrullination enhances the chemosensitivity of docetaxel to multiple tumour cells
Teng Xue, Shujia Fei, Jian Gu, Nan Li, Pengxue Zhang, Xiaoqiu Liu, Paul R Thompson, Xuesen Zhang
Philosophical Transactions of the Royal Society B: Biological Sciences.2023;[Epub] CrossRef - Baicalein improves the chemoresistance of ovarian cancer through regulation of CirSLC7A6
Shuqing Li, Zhihui Yi, Mingqing Li, Zhiling Zhu
Journal of Ovarian Research.2023;[Epub] CrossRef - Primary cell cultures for the personalized therapy in aggressive thyroid cancer of follicular origin
Poupak Fallahi, Silvia Martina Ferrari, Giusy Elia, Francesca Ragusa, Armando Patrizio, Sabrina Rosaria Paparo, Gianni Marone, Maria Rosaria Galdiero, Giovanni Guglielmi, Rudy Foddis, Alfonso Cristaudo, Alessandro Antonelli
Seminars in Cancer Biology.2022; 79: 203. CrossRef - Synergistic effects of BAY606583 on docetaxel in esophageal cancer through modulation of ERK1/2
Zinab Mohammadi, Jahanbakhsh Asadi, Seyyed Mehdi Jafari
Cell Biochemistry and Function.2022; 40(6): 569. CrossRef - Regulation of Cell Signaling Pathways and Non-Coding RNAs by Baicalein in Different Cancers
Ammad Ahmad Farooqi, Gulnara Kapanova, Sundetgali Kalmakhanov, Gulnur Tanbayeva, Kairat S. Zhakipbekov, Venera S. Rakhmetova, Marat K. Syzdykbayev
International Journal of Molecular Sciences.2022; 23(15): 8377. CrossRef - Crosstalk between xanthine oxidase (XO) inhibiting and cancer chemotherapeutic properties of comestible flavonoids- a comprehensive update
Md Sohanur Rahaman, Md Afjalus Siraj, Md Arman Islam, Prayas Chakma Shanto, Ordha Islam, Md Amirul Islam, Jesus Simal-Gandara
The Journal of Nutritional Biochemistry.2022; 110: 109147. CrossRef - Anticancer effects of natural phytochemicals in anaplastic thyroid cancer (Review)
Yitian Li, Jing Zhang, Huihui Zhou, Zhen Du
Oncology Reports.2022;[Epub] CrossRef - The Multifaceted Role of Baicalein in Cancer Management through Modulation of Cell Signalling Pathways
Arshad Husain Rahmani, Ahmad Almatroudi, Amjad Ali Khan, Ali Yousif Babiker, Malak Alanezi, Khaled S. Allemailem
Molecules.2022; 27(22): 8023. CrossRef - Ouabain Effects on Human Anaplastic Thyroid Carcinoma 8505C Cells
Mariana Pires Teixeira, Natalia Ferreira Haddad, Eliza Freitas Passos, Marcelle Novaes Andrade, Maria Luisa Arantes Campos, Joyle Moreira Carvalho da Silva, Camila Saggioro de Figueiredo, Elizabeth Giestal-de-Araujo, Denise Pires de Carvalho, Leandro Mira
Cancers.2022; 14(24): 6168. CrossRef - Baicalein Represses Cervical Cancer Cell Growth, Cell Cycle Progression and Promotes Apoptosis via Blocking AKT/mTOR Pathway by the Regulation of circHIAT1/miR-19a-3p Axis
Jiaojiao Hu, Runkun Wang, Yi Liu, Jianbo Zhou, Ka Shen, Yun Dai
OncoTargets and Therapy.2021; Volume 14: 905. CrossRef - Phytochemicals in thyroid cancer: analysis of the preclinical studies
Stefania Bulotta, Francesca Capriglione, Marilena Celano, Valeria Pecce, Diego Russo, Valentina Maggisano
Endocrine.2021; 73(1): 8. CrossRef - Cancer-associated fibroblasts (CAFs) in thyroid papillary carcinoma: molecular networks and interactions
Jeehoon Ham, Bin Wang, Joseph William Po, Amandeep Singh, Navin Niles, Cheok Soon Lee
Journal of Clinical Pathology.2021; 74(12): 759. CrossRef - Clinical Study of Virtual Reality Augmented Technology Combined with Contrast-Enhanced Ultrasound in the Assessment of Thyroid Cancer
Qinghua Liu, Jian Cheng, Jingjing Li, Lei Liu, Hongbo Li, Zhihan Lv
Journal of Healthcare Engineering.2021; 2021: 1. CrossRef - Potential of baicalein in the prevention and treatment of cancer: A scientometric analyses based review
Elika Verma, Aviral Kumar, Uzini Devi Daimary, Dey Parama, Sosmitha Girisa, Gautam Sethi, Ajaikumar B. Kunnumakkara
Journal of Functional Foods.2021; 86: 104660. CrossRef - E-cadherin on epithelial–mesenchymal transition in thyroid cancer
Xiaoyu Zhu, Xiaoping Wang, Yifei Gong, Junlin Deng
Cancer Cell International.2021;[Epub] CrossRef - Combinatorial Therapies in Thyroid Cancer: An Overview of Preclinical and Clinical Progresses
Gheysen Laetitia, Saussez Sven, Journe Fabrice
Cells.2020; 9(4): 830. CrossRef - Preclinical and clinical combination therapies in the treatment of anaplastic thyroid cancer
Daniela Gentile, Paola Orlandi, Marta Banchi, Guido Bocci
Medical Oncology.2020;[Epub] CrossRef - Interleukin-22 enhances chemoresistance of lung adenocarcinoma cells to paclitaxel
Zhiliang Huang, Yu Gao, Dianchen Hou
Human Cell.2020; 33(3): 850. CrossRef - Plant natural products with anti-thyroid cancer activity
Javad Sharifi-Rad, Sadegh Rajabi, Miquel Martorell, Maria Dolores López, María Trinidad Toro, Susi Barollo, Decio Armanini, Patrick Valere Tsouh Fokou, Giuseppe Zagotto, Giovanni Ribaudo, Raffaele Pezzani
Fitoterapia.2020; 146: 104640. CrossRef - Baicalein: A metabolite with promising antineoplastic activity
Hardeep Singh Tuli, Vaishali Aggarwal, Jagjit Kaur, Diwakar Aggarwal, Gaurav Parashar, Nidarshana Chaturvedi Parashar, Muobarak Tuorkey, Ginpreet Kaur, Raj Savla, Katrin Sak, Manoj Kumar
Life Sciences.2020; 259: 118183. CrossRef - Impact of Antioxidant Natural Compounds on the Thyroid Gland and Implication of the Keap1/Nrf2 Signaling Pathway
Ana Paunkov, Dionysios V. Chartoumpekis, Panos G. Ziros, Niki Chondrogianni, Thomas W. Kensler, Gerasimos P. Sykiotis
Current Pharmaceutical Design.2019; 25(16): 1828. CrossRef - Reversal of Multidrug Resistance in Cancer by Multi-Functional Flavonoids
Qingmei Ye, Kai Liu, Qun Shen, Qingyue Li, Jinghui Hao, Fangxuan Han, Ren-Wang Jiang
Frontiers in Oncology.2019;[Epub] CrossRef - Inhibition of Cancer Stem-Like Phenotype by Curcumin and Deguelin in CAL-62 Anaplastic Thyroid Cancer Cells
Mehmet A. Kocdor, Hakan Cengiz, Halil Ates, Hilal Kocdor
Anti-Cancer Agents in Medicinal Chemistry.2019; 19(15): 1887. CrossRef - Induction of apoptotic but not autophagic cell death by Cinnamomum cassia extracts on human oral cancer cells
Ching‐Han Yu, Shu‐Chen Chu, Shun‐Fa Yang, Yih‐Shou Hsieh, Chih‐Yi Lee, Pei‐Ni Chen
Journal of Cellular Physiology.2019; 234(4): 5289. CrossRef - Licochalcone A induces apoptotic cell death via JNK/p38 activation in human nasopharyngeal carcinoma cells
Chun‐Yi Chuang, Cheng‐Ming Tang, Hsin‐Yu Ho, Chung‐Han Hsin, Chia‐Jui Weng, Shun‐Fa Yang, Pei‐Ni Chen, Chiao‐Wen Lin
Environmental Toxicology.2019; 34(7): 853. CrossRef - Rab23 contributes to the progression of colorectal cancer via protein kinase B and extracellular signal‑regulated kinase signaling pathways
Tongbi Zhao, Dong Han, Huan Meng
Oncology Letters.2019;[Epub] CrossRef
- Modulation of hypoxia-inducible factor-1 signaling pathways in cancer angiogenesis, invasion, and metastasis by natural compounds: a comprehensive and critical review

- Endocrine Research
- Macrophage Densities Correlated with CXC Chemokine Receptor 4 Expression and Related with Poor Survival in Anaplastic Thyroid Cancer
- Dae In Kim, Eunyoung Kim, Young A Kim, Sun Wook Cho, Jung Ah Lim, Young Joo Park
- Endocrinol Metab. 2016;31(3):469-475. Published online August 2, 2016
- DOI: https://doi.org/10.3803/EnM.2016.31.3.469
- 4,747 View
- 52 Download
- 22 Web of Science
- 16 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader Background Tumor associated macrophages (TAMs) and CXC chemokine receptor 4 (CXCR4) have emerged as potential biomarkers in various human cancers. The aims of this study were to investigate the clinical characteristics of anaplastic thyroid cancer (ATC) patients according to the TAM numbers in the tumor tissue, and to evaluate the associations between CXCR4 expressions and macrophage densities in ATC tumor microenvironment.
Methods Total 14 ATC samples from thyroid tissue microarray were used. Immunohistochemical staining was performed using anti-CD163 and anti-CXCR4 antibodies. According to the immunoreactivity of CD163, all subjects were divided into two groups: low-CD163 (
n =8) and high-CD163 (n =6) groups.Results The mean diagnostic age was 65±7 years and the median tumor size was 4.3 cm, ranging 2.5 to 15 cm. Clinicopathological characteristics were not significantly different between low-CD163 and high-CD163 groups, while age of diagnosis was younger in high-CD163 group than that of low-CD163 group with marginal significance (56.9±5.5 years vs. 67.5±6.8 years,
P =0.09). However, overall survival was significantly reduced in high-CD163 group (5.5 months [range, 1 to 10]) compared with low-CD163 groups (8.8 months [range, 6 to 121); log-rank test,P =0.0443). Moreover, high-CD163 group showed strong CXCR4 expressions in both cancer and stromal compartments, while low-CD163 group showed relatively weak, stromal-dominant CXCR4 expressions. Additionally, CD163 and CXCR4 expressions showed a strong positive correlation (γ2=0.432,P =0.013).Conclusion Increased number of TAMs showed poor overall survival in ATC, suggesting TAMs are potentially a prognostic biomarker for ATC. CXCR4 expression was significantly correlated with CD163-positive TAM densities, which suggest the possible role of CXCR4 in TAM recruitments.
-
Citations
Citations to this article as recorded by- IL2RA+VSIG4+ tumor-associated macrophage is a key subpopulation of the immunosuppressive microenvironment in anaplastic thyroid cancer
Zongfu Pan, Lisha Bao, Xixuan Lu, Xiaoping Hu, Lu Li, Jinming Chen, Tiefeng Jin, Yiwen Zhang, Zhuo Tan, Ping Huang, Minghua Ge
Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease.2023; 1869(1): 166591. CrossRef - Crosstalk between Thyroid Carcinoma and Tumor-Correlated Immune Cells in the Tumor Microenvironment
Mingyuan Song, Qi Liu, Wei Sun, Hao Zhang
Cancers.2023; 15(10): 2863. CrossRef - Modeling the tumor microenvironment of anaplastic thyroid cancer: an orthotopic tumor model in C57BL/6 mice
Zhen Xu, Hyo Shik Shin, Yoo Hyung Kim, Seong Yun Ha, Jae-Kyung Won, Su-jin Kim, Young Joo Park, Sareh Parangi, Sun Wook Cho, Kyu Eun Lee
Frontiers in Immunology.2023;[Epub] CrossRef - Tumor-associated macrophages as a potential therapeutic target in thyroid cancers
Liya Zhu, Xiu Juan Li, Prakash Gangadaran, Xiuli Jing, Byeong-Cheol Ahn
Cancer Immunology, Immunotherapy.2023; 72(12): 3895. CrossRef - Functional Phenotypes of Peritoneal Macrophages Upon AMD3100 Treatment During Colitis-Associated Tumorigenesis
Shuai Wu, Weiwei Luo, Xing Wu, Zhaohua Shen, Xiaoyan Wang
Frontiers in Medicine.2022;[Epub] CrossRef - Integrated analysis of novel macrophage related signature in anaplastic thyroid cancer
Yi Luo, Yi-Chen Yang, Ben Ma, Wei-Bo Xu, Tian Liao, Yu Wang
Endocrine.2022; 78(3): 517. CrossRef - Secreted Factors by Anaplastic Thyroid Cancer Cells Induce Tumor-Promoting M2-like Macrophage Polarization through a TIM3-Dependent Mechanism
Cinthia Carolina Stempin, Romina Celeste Geysels, Sunmi Park, Luz Maria Palacios, Ximena Volpini, Claudia Cristina Motran, Eva Virginia Acosta Rodríguez, Juan Pablo Nicola, Sheue-yann Cheng, Claudia Gabriela Pellizas, Laura Fozzatti
Cancers.2021; 13(19): 4821. CrossRef - Immunotherapy for anaplastic thyroid carcinoma: the present and future
Xixuan LU, Lisha BAO, Zongfu PAN, Minghua GE
Journal of Zhejiang University (Medical Sciences).2021; 50(6): 675. CrossRef - CXCL12/CXCR4 signal transduction in diseases and its molecular approaches in targeted-therapy
Ashraf Mousavi
Immunology Letters.2020; 217: 91. CrossRef - Nobiletin Alone or in Combination with Cisplatin Decreases the Viability of Anaplastic Thyroid Cancer Cell Lines
Diana P. Sousa, Marta Pojo, Ana T. Pinto, Valeriano Leite, Ana Teresa Serra, Branca M. Cavaco
Nutrition and Cancer.2020; 72(2): 352. CrossRef - The Thyroid Tumor Microenvironment: Potential Targets for Therapeutic Intervention and Prognostication
Laura MacDonald, Jonathan Jenkins, Grace Purvis, Joshua Lee, Aime T. Franco
Hormones and Cancer.2020; 11(5-6): 205. CrossRef - Mouse Models as a Tool for Understanding Progression in BrafV600E-Driven Thyroid Cancers
Iñigo Landa, Jeffrey A. Knauf
Endocrinology and Metabolism.2019; 34(1): 11. CrossRef - Low Lymphocyte-to-Monocyte Ratios Are Associated with Poor Overall Survival in Anaplastic Thyroid Carcinoma Patients
Jonghwa Ahn, Eyun Song, Hye-Seon Oh, Dong Eun Song, Won Gu Kim, Tae Yong Kim, Won Bae Kim, Young Kee Shong, Min Ji Jeon
Thyroid.2019; 29(6): 824. CrossRef - Targeting CD47 in Anaplastic Thyroid Carcinoma Enhances Tumor Phagocytosis by Macrophages and Is a Promising Therapeutic Strategy
Christian M. Schürch, Matthias A. Roelli, Stefan Forster, Marie-Hélène Wasmer, Frido Brühl, Renaud S. Maire, Sergio Di Pancrazio, Marc-David Ruepp, Roland Giger, Aurel Perren, Anja M. Schmitt, Philippe Krebs, Roch-Philippe Charles, Matthias S. Dettmer
Thyroid.2019; 29(7): 979. CrossRef - Recent advances and emerging therapies in anaplastic thyroid carcinoma
Maria E. Cabanillas, Mark Zafereo, Michelle D. Williams, Renata Ferrarotto, Ramona Dadu, Neil Gross, G. Brandon Gunn, Heath Skinner, Gilbert Cote, Horiana B. Grosu, Priyanka Iyer, Naifa L. Busaidy
F1000Research.2018; 7: 87. CrossRef - Pathological processes and therapeutic advances in radioiodide refractory thyroid cancer
Marika H Tesselaar, Johannes W Smit, James Nagarajah, Romana T Netea-Maier, Theo S Plantinga
Journal of Molecular Endocrinology.2017; 59(4): R141. CrossRef
- IL2RA+VSIG4+ tumor-associated macrophage is a key subpopulation of the immunosuppressive microenvironment in anaplastic thyroid cancer


 KES
KES

 First
First Prev
Prev



