Articles
- Page Path
- HOME > Endocrinol Metab > Volume 32(1); 2017 > Article
-
Review ArticleUpdate on Familial Hypercholesterolemia: Diagnosis, Cardiovascular Risk, and Novel Therapeutics
-
Sang-Hak Lee1,2

-
Endocrinology and Metabolism 2017;32(1):36-40.
DOI: https://doi.org/10.3803/EnM.2017.32.1.36
Published online: January 19, 2017
1Division of Cardiology, Department of Internal Medicine, Severance Hospital, Yonsei University College of Medicine, Seoul, Korea.
2Cardiovascular Research Institute, Yonsei University College of Medicine, Seoul, Korea.
- Corresponding author: Sang-Hak Lee. Division of Cardiology, Department of Internal Medicine, Severance Hospital, Yonsei University College of Medicine, 50-1 Yonsei-ro, Seodaemun-gu, Seoul 03722, Korea. Tel: +82-2-2228-8460, Fax: +82-2-2227-7732, shl1106@yuhs.ac
• Received: November 18, 2016 • Revised: December 8, 2016 • Accepted: December 20, 2016
Copyright © 2017 Korean Endocrine Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- ABSTRACT
- INTRODUCTION
- PREVALENCE OF FAMILIAL HYPERCHOLESTEROLEMIA
- CLINICAL AND GENETIC DIAGNOSIS
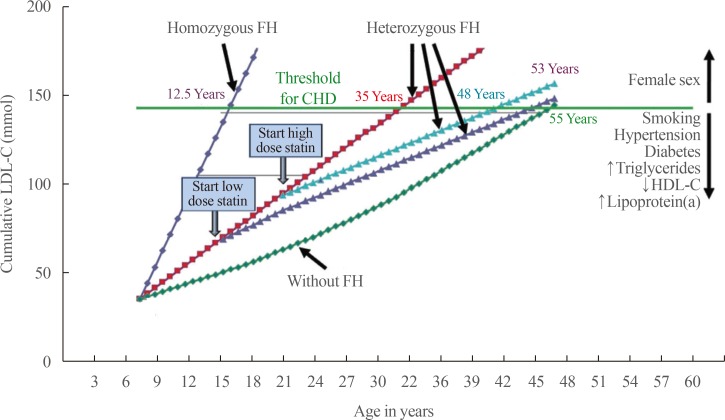
- CARDIOVASCULAR RISK IN FAMILIAL HYPERCHOLESTEROLEMIA
- THE GOALS AND RESULTS OF LIPID-LOWERING TREATMENT AND NOVEL THERAPEUTICS
- FAMILIAL HYPERCHOLESTEROLEMIA IN ASIA AND KOREA
- CONCLUSIONS
- ACKNOWLEDGMENTS
- Article information
- References
Figure & Data
References
Citations
Citations to this article as recorded by 

- Effectiveness and Safety of a Fixed-Dose Combination of Valsartan and Rosuvastatin (Rovatitan® Tablet) in Patients with Concomitant Hypertension and Hyperlipidemia: An Observational Study
Kwang Je Lee, Jae-Kean Ryu, Yun-Hyeong Cho, Won Yong Shin, Jeong Su Kim, Young Won Yoon, Ji Yong Jang, Won Ho Kim, Jong Wook Beom, Seok-Min Kang
Drug Design, Development and Therapy.2023; Volume 17: 1047. CrossRef - Role of PCSK9 Inhibitors in Patients with Familial Hypercholesterolemia
Brian Tomlinson, Nivritti Gajanan Patil, Manson Fok, Christopher Wai Kei Lam
Endocrinology and Metabolism.2021; 36(2): 279. CrossRef - Identification and Functional Characterization of a Low-Density Lipoprotein Receptor Gene Pathogenic Variant in Familial Hypercholesterolemia
Hong-Yan Shu, Wei Zhang, Cong-Cong Zheng, Man-Yun Gao, Yong-Cun Li, Yan-Gang Wang
Frontiers in Genetics.2021;[Epub] CrossRef - Gut Microbiota and Complications of Type-2 Diabetes
Camelia Oana Iatcu, Aimee Steen, Mihai Covasa
Nutrients.2021; 14(1): 166. CrossRef - LDLR Gene Mutation p.Asp360His and Familial Hypercholesterolemia in a Mexican Community
Teresita De Jesús Hernández Flores, Juan Ramón González García, Yoaly Josefina Sánchez López, Norma Alejandra Vázquez Cárdenas, Ana Gabriela Colima Fausto, Sergio Yair Rodríguez Preciado, María Teresa Magaña Torres
Archives of Medical Research.2020; 51(2): 153. CrossRef - A Rare Double Heterozygous Mutation in Low-Density Lipoprotein Receptor and Apolipoprotein B-100 Genes in a Severely Affected Familial Hypercholesterolaemia Patient
Lilla Juhász, István Balogh, László Madar, Beáta Kovács, Mariann Harangi
Cureus.2020;[Epub] CrossRef - Efficacy and Safety of a Fixed-Dose Combination of Candesartan and Rosuvastatin on Blood Pressure and Cholesterol in Patients With Hypertension and Hypercholesterolemia: A Multicenter, Randomized, Double-Blind, Parallel Phase III Clinical Study
Kyoung Im Cho, Bo Hyun Kim, Yong Hyun Park, Jeong-Cheon Ahn, Sang Hyun Kim, Wook Jin Chung, Weon Kim, Il Suk Sohn, Jin Ho Shin, Yong Jin Kim, Kiyuk Chang, Cheol Woong Yu, Soe Hee Ahn, Seok Yeon Kim, Jae Kean Ryu, Jong Young Lee, Bum Kee Hong, Taek Jong Ho
Clinical Therapeutics.2019; 41(8): 1508. CrossRef - Diagnosis and Treatment of Heterozygous Familial Hypercholesterolemia
Mary P. McGowan, Seyed Hamed Hosseini Dehkordi, Patrick M. Moriarty, P. Barton Duell
Journal of the American Heart Association.2019;[Epub] CrossRef - Autosomal recessive hypercholesterolemia: Case report
Zaneta Petrulioniene, Urte Gargalskaite, Violeta Mikstiene, Rimvydas Norvilas, Egle Skiauteryte, Algirdas Utkus
Journal of Clinical Lipidology.2019; 13(6): 887. CrossRef -
Q192R polymorphism in the
PON1
gene and familial hypercholesterolemia in a Saudi population
Khalid Khalaf Alharbi, May Salem Alnbaheen, Fawiziah Khalaf Alharbi, Rana M. Hasanato, Imran Ali Khan
Annals of Saudi Medicine.2017; 37(6): 425. CrossRef

 KES
KES
 PubReader
PubReader Cite
Cite