Indications for Fine Needle Aspiration in Thyroid Nodules
Article information
Abstract
Thyroid nodules are a common clinical problem with the widespread use of ultrasonography. Fine needle aspiration (FNA) is the mainstay for diagnosing a thyroid malignancy. There have been several guidelines on when to perform FNA in thyroid nodules. This review is based on several published recommendations and helps physicians easily understand the factors favoring FNA.
INTRODUCTION
With the increase in worldwide use of neck ultrasonography (US), the detection of thyroid nodules has rapidly increased to up to 67% of patients who undergo neck US [1]. Among them, only 4% to 7% of patients have palpable nodules on physical examination [2]. In South Korea, the yearly incidence has rapidly increased from 6.3 per 100,000 in 1999 to 47.5 per 100,000 in 2009 [3].
Fine needle aspiration (FNA) is the standard tool for detecting thyroid cancer and has led to a decrease in the number of thyroid surgeries and an increase in cancer detected during thyroid surgery [4-6]. The overall incidence of thyroid cancer is about 9.2% to 13% in patients with thyroid nodules who undergo FNA [7-9]. Also, the high detection rate of US makes FNA for all US-detected nodules impractical, if not impossible. Therefore, deciding which nodules should be biopsied is an important medical issue to ensure that no clinically significant thyroid cancers are missed. There are several guidelines for the indications of FNA in thyroid nodules [10-14]. This work demonstrates these guidelines and compares their merits.
CONSIDERATION FACTORS TO PERFORM FNAs
Size
Thyroid nodule size itself is not a predictive factor of malignancy [7,9-11]. Nevertheless, most guidelines recommend FNA for nodules larger than 10 mm [11,13,15], unless patients have high risk factors [15] or suspicious US features [13]. High risk factors include a history of thyroid cancer in one or more first-degree relatives, a history of exposure to external beam radiation or ionizing radiation in childhood or adolescence, prior hemithyroidectomy with discovery of thyroid cancer, fluorodeoxyglucose (18F) avidity on positron emission tomography scanning, multiple endocrine neoplasia (MEN) 2/familial medullary thyroid cancer-associated RET proto-oncogene mutation, or calcitonin levels >100 pg/mL [15]. Based on the revised American Thyroid Association (ATA) guidelines, FNA is recommended in thyroid nodules larger than 5 mm with suspicious US features in high risk patients. This guideline does not recommend FNA in thyroid nodules smaller than 5 mm. The size criterion for FNA is slightly different between the ATA and the American Association of Clinical Endocrinologists/Associazione Medici Endocrinologi (AACE/AME) guidelines. The AACE/AME guidelines suggests that FNA be performed regardless of lesion size when patients have a history of neck irradiation, a family history of medullary thyroid cancer or MEN2, extracapsular growth, or metastatic cervical lymph nodes [13]. The Korean Society of Thyroid Radiology (KSThR) has an even more strict size criteria [14]. They recommend FNA in thyroid nodules larger than 5 mm with suspicious US features, even when a patient does not have high risk factors. They also recommend selective FNA in thyroid nodules smaller than 5 mm according to risk factors and according to the performing radiologist's experience.
This diversity in size criteria stems from the uncertainty regarding the clinical meaning of early diagnosis for tiny thyroid cancers in patients at low risk. The rationale behind the recommendations for FNA in larger nodules is based on a high rate of false positives and nondiagnostic cytology in thyroid nodules smaller than 5 mm [16,17].
Number of nodules
Traditionally, single nodules are considered to be at more risk for malignancy than multiple nodules. However, patients with multiple thyroid nodules do not have a decreased likelihood of thyroid cancer when compared to patients with solitary nodules [7,18]. The incidence of thyroid cancer in patients with multiple nodules is the same as that in patients with a solitary nodule, although the cancer rate per nodule decreases in patients with multiple nodules [19]. Thus, an FNA operator should focus on the US features of each individual nodule to decide whether or not to perform FNA.
Interval growth
Nodule growth is defined when a nodule shows more than a 50% increase in volume or a 20% increase in at least two nodule dimensions with a minimal increase of 2 mm in solid or in the solid portion of mixed nodules [15] in order to minimize interobserver bias of each measurement (Fig. 1) [20]. Although the growth itself is not a pathognomonic feature of malignancy [21] and the risk of malignancy is very low in a thyroid nodule with benign cytology [13], repeated FNA is usually indicated in nodules that increase in size [14,15].
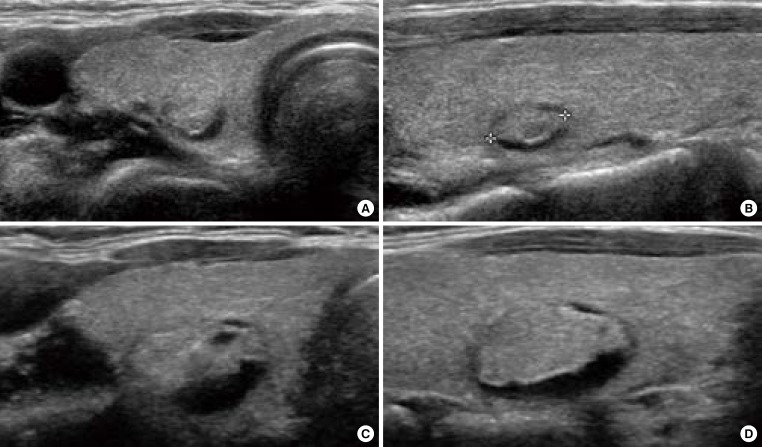
Ultrasonography (US) of growing benign mass in a 40-year-old woman who underwent fine needle aspiration twice. (A) Transverse and (B) longitudinal US images demonstrate a 0.7-cm-sized isoechoic nodule with the cystic portion of the posterior portion of the nodule in the right thyroid gland. (C, D) After 4 years, the nodule was enlarged. The cytology was benign, twice.
US features
Internal content of nodules
Depending on the components of the internal part of the thyroid nodule, nodules can be classified into cystic, mixed (both solid and cystic components), and solid nodules [22-24]. In cases where microcysts are aggregated in mixed nodules, the nodule is further defined as spongiform [25].
An anechoic cyst is definitely benign and can contain hyperechoic spots with comet tail artifacts. The comet tail artifacts are related to microcrystals inside colloid cysts which should be differentiated from the microcalcifications of malignant nodules.
Solidity itself is not considered a suspicious US feature. However, several guidelines recommend FNA in solid nodules larger than 10 mm and mixed echoic nodules larger than 15 mm with some variation in the defining size depending on the guidelines used [11,15].
Echogenicity
Traditionally, hypoechogenicity is a well-known US feature related to malignancy, and most thyroid carcinomas are hypoechoic compared with surrounding thyroid parenchyma [10,26,27]. Hypoechogenicity is a very sensitive sign of malignancy but is less specific than other features. Marked hypoechogenicity is defined as decreased echogenicity less than that of the adjacent strap muscle [10] and is a specific sign of thyroid cancer.
The ATA and AACE/AME consider hypoechogenicity as a suspicious US feature [13,15]. In comparison, the KSThR considers marked hypoechogenicity as a suspicious US feature [14].
Calcification
Microcalcification is a very specific US finding that suggests thyroid malignancy [11,13,15,22,24]. It is defined as a prominent echogenic focus with or without posterior shadowing. Unlike other reports [11,13,15,22,24], the KSThR considers both microcalcification and macrocalcification as suspicious US features [14].
Margin
The analysis of nodule margins is very subjective, resulting in very low interobserver agreement [28-32]. Microlobulated, irregular, infiltrative, or speculated margins have been considered as suspicious US features.
Shape
A taller than wide shape was first described by Kim et al. [10], after which it has been considered a suspicious US feature [13-15]. Although the ATA guidelines suggests that a taller than wide shape on transverse view alone is a suspicious US feature, a recent study demonstrates that a taller than wide shape in either the transverse or longitudinal plane can be accurate and sensitive for predicting thyroid malignancy [33].
Vascularity
Intranodular and chaotic vascularity have been considered as suspicious US features [13,15]. Recently, Moon et al. [34] published a study with color Doppler US performed on 1,083 nodules and showed that there was actually more blood flow distribution in benign nodules than in malignant nodules, and that vascularity was not helpful in differentiating benign and malignant nodules. The KSThR does not consider vascular pattern as a suspicious US feature [14].
Combinations of several suspicious US features
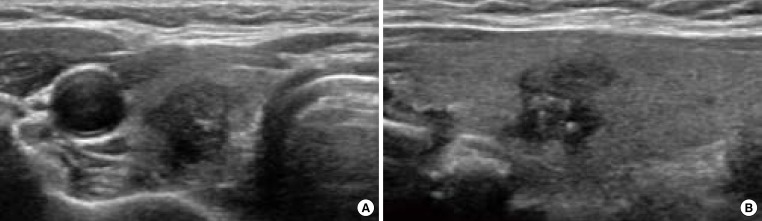
All of the US features explained thus far cannot alone diagnose thyroid nodules. The risk of malignancy increases as the number of suspicious US features increases (Fig. 2) [13,24].
CONCLUSIONS
Current guidelines vary on how to differentiate thyroid nodules into benign or malignant nodules through US. Reported guidelines show substantial overlap in what defines benign and malignant nodules, and what might be considered benign by one recommendation might be considered malignant by another. Another issue is that US assessment is subjective and operator-dependent. Therefore, US performers should continuously compare their readings with confirmed diagnostic results to maintain and improve proficient diagnostic abilities.
Notes
No potential conflict of interest relevant to this article was reported.