Articles
- Page Path
- HOME > Endocrinol Metab > Volume 35(3); 2020 > Article
-
Review ArticleMetabolically Healthy and Unhealthy Normal Weight and Obesity
-
Norbert Stefan1,2,3

-
Endocrinology and Metabolism 2020;35(3):487-493.
DOI: https://doi.org/10.3803/EnM.2020.301
Published online: August 20, 2020
1Department of Internal Medicine IV, University Hospital of Tübingen, Tübingen, Germany
2Institute of Diabetes Research and Metabolic Diseases (IDM) of the Helmholtz Center Munich, Tübingen, Germany
3German Center for Diabetes Research (DZD), Neuherberg, Germany
- Corresponding author: Norbert Stefan. Department of Internal Medicine IV, University Hospital of Tübingen, OtfriedMüller Str. 10, 72076 Tubingen, Germany Tel: +49-7071-2980390, Fax: +49-7071-295974, E-mail: norbert.stefan@med.uni-tuebingen.de
• Received: June 23, 2020 • Revised: July 15, 2020 • Accepted: July 29, 2020
Copyright © 2020 Korean Endocrine Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- ABSTRACT
- INTRODUCTION
- METABOLICALLY HEALTHY OBESITY
- METABOLICALLY UNHEALTHY NORMAL WEIGHT
- METABOLIC HEALTH AS A TRANSIENT STATE
- CARDIOMETABOLIC RISK IN PERISTENT METABOLIC HEALTH
- PREDICTORS OF THE CHANGE OF METABOLIC HEALTH
- THE CONCEPT OF METABOLIC HEALTH IN CLINICAL PRACTICE
- CONCLUSIONS
- Article information
- References
ABSTRACT
- Increased fat mass is an established risk factor for the cardiometabolic diseases type 2 diabetes and cardiovascular disease (CVD) and is associated with increased risk of all-cause and CVD mortality. However, also very low fat mass associates with such an increased risk. Whether impaired metabolic health, characterized by hypertension, dyslipidemia, hyperglycemia, insulin resistance, and subclinical inflammation, may explain part of the elevated risk of cardiometabolic diseases that is found in many subjects with very low fat mass, as it does in many obese subjects, is unknown. An important pathomechanism of impaired metabolic health is disproportionate fat distribution. In this article the risk of cardiometabolic diseases and mortality in subjects with metabolically healthy and unhealthy normal weight and obesity is summarized. Furthermore, the change of metabolic health during a longer period of follow-up and its impact on cardiometabolic diseases is being discussed. Finally, the implementation of the concept of metabolic health in daily clinical practice is being highlighted.
- Worldwide there is an increase in the prevalence of overweight and obesity [1]. Both, overweight and particularly obesity are associated with an increased risk of the cardiometabolic diseases type 2 diabetes and cardiovascular disease (CVD), certain types of cancer and mortality [2-5]. In most studies that investigated the relationships of fat mass with all cause and cause-specific mortality, fat mass has been estimated by the calculation of the body mass index (BMI) [2-4]. In this respect the nadir of allcause mortality risk was found to be 23 kg/m2 at age younger than 70 years, rising to 25 kg/m2 at age 70 years and older, while a BMI in the range of 21 to 25 kg/m2 was associated with the lowest risk of cancer, cardiovascular, and respiratory mortality [4]. In all of these studies a J-shaped relationship of BMI with all-cause and disease-specific mortality was found [2-4]. The increased mortality, that has been observed at a BMI <21 kg/m2, and particularly in the range of underweight (<18.5 kg/m2), may be largely attributed to increased mortality from mental and behavioural, neurological, and external causes [4,6]. However, as patients with lipodystrophy [7] or a lipodystrophy-like phenotype [8,9] have an increased cardiometabolic risk, it cannot be excluded that particularly a low amount of fat mass in the subcutaneous region may also increase mortality in the general population with a low BMI. Recent analyses from the Women’s Health Initiative study in postmenopausal women with normal BMI (18.5 to <25kg/m2) showed that lower gluteofemoral fat mass, estimated using dual-energy X-ray absorptiometry (DEXA), was associated with a higher incidence of CVD and that this increased risk was independent of increased trunk fat mass [10]. Furthermore, there is emerging evidence from precise phenotyping studies and from genetic studies showing that increased gluteofemoral and leg fat mass is protective of cardiometabolic diseases [11-13].
INTRODUCTION
- Because it is often not feasible to precisely measure fat distribution using DEXA, computed tomography or magnetic resonance imaging, during the last decade estimation of the cardiometabolic risk using insulin resistance and/or parameters of the metabolic syndrome has been the focus of intense research. Stefan et al. [14] found in 2008 that insulin sensitive obese subjects have a very characteristic body fat distribution that is characterized by a low amount of visceral fat and, more so, by a lower amount of liver fat content. In a back-to-back publication with our study, Wildman et al. [15] found in the National Health and Nutrition Examination Surveys 1999 to 2004 a similar prevalence of metabolically healthy obese subjects, when using parameters of the metabolic syndrome, a high homeostasis model assessment of insulin resistance (HOMA-IR) and an elevated high-sensitivity C-reactive protein (hs-CRP) level. Importantly, assuming that subjects cannot be considered ‘metabolically healthy’ when setting the cut-off for ‘health’ <3 parameters of the metabolic syndrome, they considered individuals being metabolically healthy when <2 parameters of the metabolic syndrome, except waist circumference, and high HOMA-IR and hs-CRP levels were present. Thus, while currently there is no broad agreement among the researchers and clinicians on how to define metabolic health (MH) [16-21], the definition proposed by Wildman et al. [15] appears to be most useful (Table 1). Nevertheless, many studies having investigated the relationship of metabolic healthy obesity (MHO) with cardiometabolic risk or cardiometabolic mortality used the definition of the metabolic syndrome. In meta-analyses of these studies subjects with MHO were found to have a moderately increased (~30%) risk of CVD and CVD mortality, when compared to subjects with metabolically healthy normal weight (MHNW). However, for a comparable BMI, the same risk was much higher (~150%) in subjects with metabolically unhealthy obesity (MUHO) [22-25]. While these studies mostly included Caucasians, similar relationships were also found in Asian populations. For example in an analysis of 323,175 adults from the large Korean National Health Insurance System (NHIS) database, who were followedup for a median of 96 months, subjects with MHO even had a lower all-cause (–19%) and cardiovascular (–27%) mortality risk, when compared to non-obese and metabolically healthy subjects. Of note, in that study the authors used a very strict definition of MH, in that subjects with MH were required to have none of three metabolic disease components (hypertension, diabetes, dyslipidemia) [26]. Thus, in general, subjects with MHO cannot be considered being truly healthy, but of having a much lower cardiometabolic risk, than most of the obese individuals.
METABOLICALLY HEALTHY OBESITY
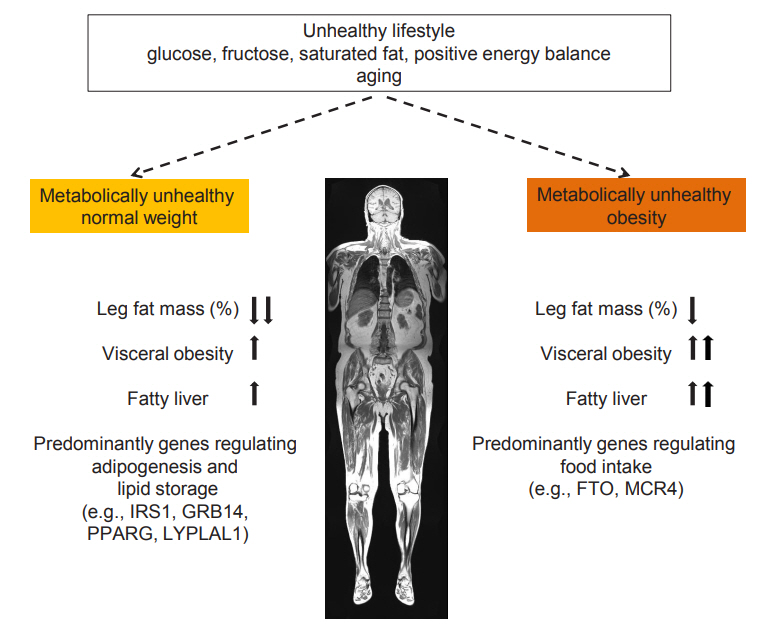
- In the course of the investigation of the cardiometabolic risk of subjects with MHO, for a long time the unexpected finding of very high risk (~120%) of CVD and CVD mortality of subjects with metabolically unhealthy normal weight (MUHNW), compared to subjects with MHNW, was not well recognized [22- 25]. When investigating the body fat distribution phenotypes of subjects with MUHNW Stefan et al. [8] found that it somewhat differed from the one observed in subjects with MUHO. While subjects with MUHO mostly have a very high visceral fat mass and an elevated liver fat content, subjects with MUHNW are predominantly characterized by a low amount of gluteofemoral fat mass [8]. Recently data from the Women’s Health Initiative Study support that a low gluteofemoral fat mass, measured by DEXA, associates with a high risk of CVD in lean women, and that this risk is independent of elevated trunk fat mass, which was also measured using DEXA [10]. It is widely accepted that adipose tissue in the gluteofemoral region serves as a healthy sink to store excess fat. This is mostly due to the fact that under excess energy intake this fat compartment responds predominantly by hyperplasia, which is accompanied by lower lipolytic activity. Thus, there is less spill-over of fatty acids into the circulation and less storage of fat ectopically in metabolic relevant organs, such as the liver and the pancreas [11]. Genetic studies further suggest that while the MUHO phenotype is predominantly characterized by variability in genes regulating food intake, the MUHNW phenotype is strongly characterized by variability in genes regulating adipocyte differentiation, lipogenesis and lipolysis. Furthermore, genes regulating hepatic de novo lipogenesis and lipid release from the liver and lifestyle parameters of the individual and its parents, impact on the pathogenesis of fatty liver (Fig. 1) [8,27,28].
METABOLICALLY UNHEALTHY NORMAL WEIGHT
- During ageing there is body fat redistribution, predominantly from the gluteofemoral area to the upper body and, in most cases, a decrease in physical activity. This may result in a decline of the prevalence of MH. In agreement, in the Whitehall II study, about 50% of initially healthy obese individuals converted to an unhealthy phenotype over 20 years of follow-up [29]. In the largest study, also with the longest period of follow-up, that investigated the change of MH over time, the Nurses’ Health Study, among the initially metabolically healthy obese women only 16% remained metabolically healthy over 20 years of follow-up. This number decreased to 6% after 30 years of followup. However, also in initially metabolically healthy women with normal weight MH declined and after 30 years of follow-up only 15% of the women remained metabolically healthy [30].
METABOLIC HEALTH AS A TRANSIENT STATE
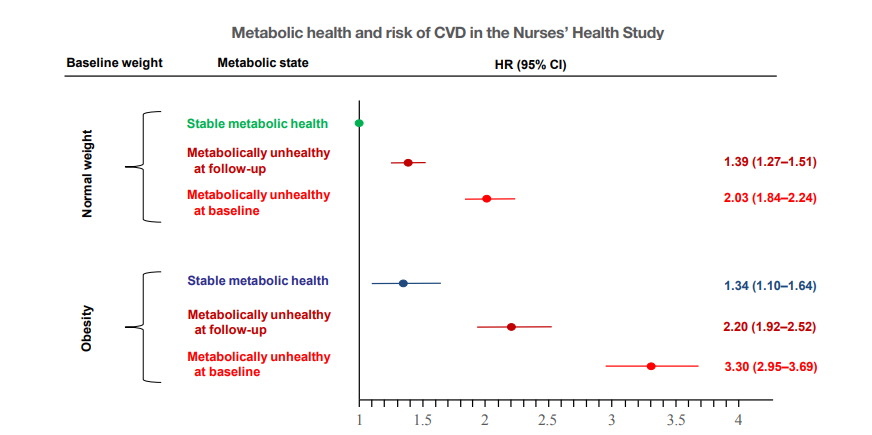
- An important question is whether and to what extent the cardiometabolic risk differs in obese subjects who can retain MH over a longer period of follow-up. Data from the North West Adelaide Health Study suggest that risk of diabetes, CVD, or stroke is not increased in people with MHO compared to people with MHNW, if the metabolically healthy obesity phenotype is maintained during a natural follow-up over 5.5 to 10.3 years [31]. In the Nurses’ Health Study, women who maintained the MHO phenotype over 20 years still had a higher (34%) risk of CVD over the following 10 years of follow-up, when compared with normal weight women who were metabolically healthy over the same time period. However, this risk was much higher (120%) in obese women who could not retain MH (Fig. 2) [30]. These relationships were found in studies that predominantly included Caucasians. However, they may also be present in Asian populations. For example in a community-based population in Shanghai, China, 46.8% of individuals with MHO developed a metabolically unhealthy status during a follow-up period of 4.4 years. While subjects with transient MHO had an increased risk (152%) of a composite subclinical atherosclerosis endpoint, the risk was statistically not different (8%) in subjects with stable MHO, when compared to subjects who were metabolically healthy and non-obese (MHNO) [32].
- Of interest, persistent MH in obesity does not protect from heart failure as was most recently shown in an analysis of data from the Korean NHIS datasets from 2002 to 2017 [33]. When compared to stable MHNO individuals, subjects who retained MHO during a mean follow-up of 3.70 years, had a 17.3% increased hazard of hospitalization for heart failure (hHF). Furthermore, individuals who shifted from MHO to MHNO had a 34.3% lower hazard of hHF [33]. This is in agreement with previous observations showing that subjects with MHO may have no or only a moderately increased risk of myocardial infarction, but are not protected from heart failure [34], which is also strongly driven by obesity [35,36].
CARDIOMETABOLIC RISK IN PERISTENT METABOLIC HEALTH
- An important question related to the concept of MH is how can a metabolically healthy state be retained or achieved in subjects with impaired MH. In this respect weight loss that is brought about by a lifestyle intervention, may be very effective. In support of this hypothesis in the Tübingen Lifestyle Intervention Program, in subjects with MUHO, a median weight loss of 9.2 kg over a median period of 9 months was associated with the conversion to MH. Furthermore, among several of the baseline parameters tested (age, sex, BMI, waist circumference, systolic and diastolic blood pressure, fasting glucose, high density lipoprotein cholesterol and triglycerides, and MRI- and 1 HMRspectroscopy-derived measurements of visceral fat mass and liver fat content), only BMI and liver fat content remained independent predictors of the conversion from MUHO to MH [37]. In agreement with an important role of fatty liver in the pathogenesis of cardiometabolic diseases, involving insulin resistance, subclinical inflammation, increased hepatic glucose production, dyslipidemia and dysregulated hepatokine production [38-43], fatty liver was also found to be a stronger determinant of subclinical atherosclerosis than visceral fat mass and hyperglycemia [44].
- However, can such a beneficial effect also be observed with a lifestyle intervention that is not accompanied by weight loss? In this respect data from the Prevención con Dieta Mediterránea (PREDIMED) study show that even in the absence of a large amount of weight loss, adherence to a Mediterranean diet can promote the transition to MH in subjects with MUHO and protect against deterioration of MH in subjects with MHO [45]. Similar results were also found in other studies involving healthy diets [18].
PREDICTORS OF THE CHANGE OF METABOLIC HEALTH
- It is undisputed that all obese individuals, independent of the status of MH, should aim at achieving normal weight. For this several weight-loss programs have been recommended by the medical guidelines focusing on diet, exercise, pharmacological therapy and bariatric surgery [46]. However, based on the limited resources that are available for weight-loss programs, it may be reasonable to allocate resources predominantly to the MUHO people. In support of this assumption a study investigating the effect of phentermine/topiramate-induced weight loss compared to placebo on the prevention of diabetes in subjects who were stratified by the Cardiometabolic Disease Staging (CMDS) score in those with a high or a low cardiometabolic risk, provided interesting information. This score incorporates the parameters of the metabolic syndrome and, thus, is very similar to the MHO/MUHO concept. In that study the baseline mean BMI ranged from 34.2 to 41.2 kg/m2 among the subgroups and the percentage of weight loss was with around 10% almost identical in the verum groups. Although the treatment reduced diabetes risk compared to placebo in the high-risk group, in the latter group a risk as low as baseline in the low-risk was not achieved. Thus, substantially larger weight loss may be necessary to achieve a similarly low diabetes risk as the low-risk group. Second, still, the numbers needed to treat to prevent one case of diabetes over about 1 year were 120 in the low-risk group but only 24 in the high risk group. Thus, this study provided support that targeting high-risk patients for specific weight-loss strategies may improve the cost-benefit ratio of such interventions. Furthermore, while during the COVID-19 pandemic obesity emerged as an important and independent risk factor for a more severe course of the disease [47-49], a metabolically unhealthy condition is still considered to substantially increase this risk [49,50].
THE CONCEPT OF METABOLIC HEALTH IN CLINICAL PRACTICE
- MH is not an entirely new concept. The parameters that are being used for its definition are well-known parameters of the metabolic syndrome [51]. However, there is agreement among the researchers and health care providers that a more stringent exclusion of risk parameters, such as is being done when defining MH, may improve the cardiometabolic risk prediction [11,18,20]. Furthermore, while intense research was done to understand the risk of diseases in subjects with MHO compared to people with MUHO, the widely neglected increased risk of cardiometabolic diseases in some of the lean people, became a major focus of recent cardiometabolic studies. Finally, as for many overweight and obese people MH may be an interesting and easily reachable ‘low hanging fruit’ on the path of weight-loss [37], implementation of the concept of MH in the communication of the health care providers with their patients may be of great motivational value.
CONCLUSIONS
-
CONFLICTS OF INTEREST
No potential conflict of interest relevant to this article was reported.
Article information
-
Acknowledgements
- This work was funded by the German Research Foundation (STE 1096/1-3) and the German Federal Ministry of Education and Research to the German Centre of Diabetes Research.
Fig. 1.Fat distribution, fatty liver and main lifestyle and genetic determinants of metabolically unhealthy normal weight and obesity. The arrows indicate whether the prevalence of fat distribution and fatty liver is increased or decreased in the metabolically unhealthy condition. Modified from Stefan et al. [8]. IRS1, insulin receptor substrate 1; GRB14, growth factor receptor-bound protein 14; PPARG, peroxisome proliferator activated receptor gamma; LYPLAL1, lysophospholipase like 1; FTO, fat mass and obesity-associated; MC4R, melanocortin-4 receptor.


Fig. 2.Transition from metabolic healthy to unhealthy phenotypes and association with cardiovascular disease (CVD) risk across body mass index categories in 90,257 women of the Nurses’ Health Study. Risk of CVD in women with normal weight and obesity, stratified by metabolic health status. Hazard ratios (HRs) are adjusted for age, race, highest degree, alcohol consumption, postmenopausal status, physical examinations for screening purposes, family history of myocardial infarction and diabetes, aspirin use, smoking status change, physical activity. The data are from Eckel et al. [30], with permission from Elsevier. CI, confidence interval.
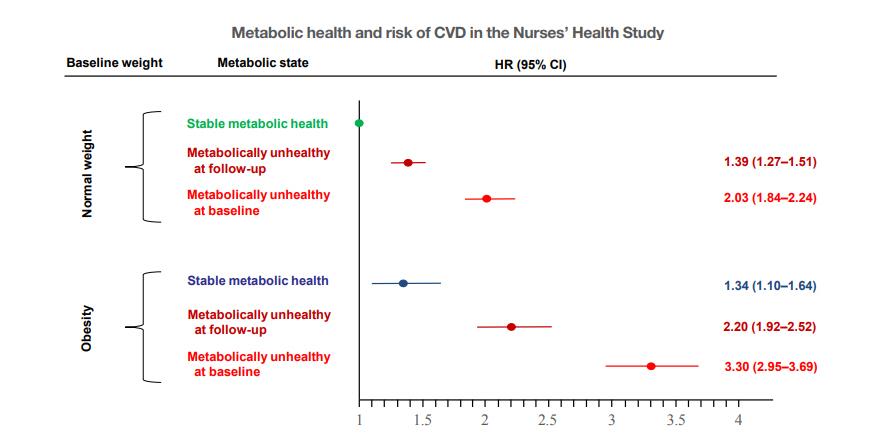

Table 1.Definition of Metabolic Risk as Proposed by Wildman et al. [15]
- 1. NCD Risk Factor Collaboration (NCD-RisC). Trends in adult body-mass index in 200 countries from 1975 to 2014: a pooled analysis of 1698 population-based measurement studies with 19·2 million participants. Lancet 2016;387:1377–96.ArticlePubMedPMC
- 2. Global BMI Mortality Collaboration, Di Angelantonio E, Bhupathiraju ShN, Wormser D, Gao P, Kaptoge S, et al. Body-mass index and all-cause mortality: individual-participant-data meta-analysis of 239 prospective studies in four continents. Lancet 2016;388:776–86.ArticlePubMedPMC
- 3. GBD 2015 Obesity Collaborators, Afshin A, Forouzanfar MH, Reitsma MB, Sur P, Estep K, et al. Health effects of overweight and obesity in 195 countries over 25 years. N Engl J Med 2017;377:13–27.ArticlePubMedPMC
- 4. Bhaskaran K, Dos-Santos-Silva I, Leon DA, Douglas IJ, Smeeth L. Association of BMI with overall and cause-specific mortality: a population-based cohort study of 3·6 million adults in the UK. Lancet Diabetes Endocrinol 2018;6:944–53.ArticlePubMedPMC
- 5. Calle EE, Rodriguez C, Walker-Thurmond K, Thun MJ. Overweight, obesity, and mortality from cancer in a prospectively studied cohort of U.S. adults. N Engl J Med 2003;348:1625–38.ArticlePubMed
- 6. Tobias DK, Hu FB. The association between BMI and mortality: implications for obesity prevention. Lancet Diabetes Endocrinol 2018;6:916–7.ArticlePubMed
- 7. Mann JP, Savage DB. What lipodystrophies teach us about the metabolic syndrome. J Clin Invest 2019;129:4009–21.ArticlePubMedPMC
- 8. Stefan N, Schick F, Haring HU. Causes, characteristics, and consequences of metabolically unhealthy normal weight in humans. Cell Metab 2017;26:292–300.ArticlePubMed
- 9. Eigentler T, Lomberg D, Machann J, Stefan N. Lipodystrophic nonalcoholic fatty liver disease induced by immune checkpoint blockade. Ann Intern Med 2020;172:836–7.ArticlePubMed
- 10. Chen GC, Arthur R, Iyengar NM, Kamensky V, Xue X, Wassertheil-Smoller S, et al. Association between regional body fat and cardiovascular disease risk among postmenopausal women with normal body mass index. Eur Heart J 2019;40:2849–55.ArticlePubMedPMCPDF
- 11. Stefan N. Causes, consequences, and treatment of metabolically unhealthy fat distribution. Lancet Diabetes Endocrinol 2020;8:616–27.ArticlePubMed
- 12. Lotta LA, Wittemans LBL, Zuber V, Stewart ID, Sharp SJ, Luan J, et al. Association of genetic variants related to gluteofemoral vs abdominal fat distribution with type 2 diabetes, coronary disease, and cardiovascular risk factors. JAMA 2018;320:2553–63.ArticlePubMedPMC
- 13. Loos RJ. The genetics of adiposity. Curr Opin Genet Dev 2018;50:86–95.ArticlePubMedPMC
- 14. Stefan N, Kantartzis K, Machann J, Schick F, Thamer C, Rittig K, et al. Identification and characterization of metabolically benign obesity in humans. Arch Intern Med 2008;168:1609–16.ArticlePubMed
- 15. Wildman RP, Muntner P, Reynolds K, McGinn AP, Rajpathak S, Wylie-Rosett J, et al. The obese without cardiometabolic risk factor clustering and the normal weight with cardiometabolic risk factor clustering: prevalence and correlates of 2 phenotypes among the US population (NHANES 1999-2004). Arch Intern Med 2008;168:1617–24.ArticlePubMed
- 16. Stefan N, Haring HU, Hu FB, Schulze MB. Metabolically healthy obesity: epidemiology, mechanisms, and clinical implications. Lancet Diabetes Endocrinol 2013;1:152–62.ArticlePubMed
- 17. Samocha-Bonet D, Dixit VD, Kahn CR, Leibel RL, Lin X, Nieuwdorp M, et al. Metabolically healthy and unhealthy obese: the 2013 Stock Conference report. Obes Rev 2014;15:697–708.ArticlePubMedPMC
- 18. Schulze MB. Metabolic health in normal-weight and obese individuals. Diabetologia 2019;62:558–66.ArticlePubMedPDF
- 19. Magkos F. Metabolically healthy obesity: what’s in a name? Am J Clin Nutr 2019;110:533–9.ArticlePubMedPDF
- 20. Smith GI, Mittendorfer B, Klein S. Metabolically healthy obesity: facts and fantasies. J Clin Invest 2019;129:3978–89.ArticlePubMedPMC
- 21. Bluher M. Metabolically healthy obesity. Endocr Rev 2020;41:405–20.ArticlePDF
- 22. Kramer CK, Zinman B, Retnakaran R. Are metabolically healthy overweight and obesity benign conditions?: a systematic review and meta-analysis. Ann Intern Med 2013;159:758–69.ArticlePubMed
- 23. Hamer M, Stamatakis E. Metabolically healthy obesity and risk of all-cause and cardiovascular disease mortality. J Clin Endocrinol Metab 2012;97:2482–8.ArticlePubMedPMC
- 24. Eckel N, Meidtner K, Kalle-Uhlmann T, Stefan N, Schulze MB. Metabolically healthy obesity and cardiovascular events: a systematic review and meta-analysis. Eur J Prev Cardiol 2016;23:956–66.ArticlePubMed
- 25. Lassale C, Tzoulaki I, Moons KGM, Sweeting M, Boer J, Johnson L, et al. Separate and combined associations of obesity and metabolic health with coronary heart disease: a pan-European case-cohort analysis. Eur Heart J 2018;39:397–406.ArticlePubMedPMCPDF
- 26. Yang HK, Han K, Kwon HS, Park YM, Cho JH, Yoon KH, et al. Obesity, metabolic health, and mortality in adults: a nationwide population-based study in Korea. Sci Rep 2016;6:30329.ArticlePubMedPMCPDF
- 27. Eslam M, Valenti L, Romeo S. Genetics and epigenetics of NAFLD and NASH: clinical impact. J Hepatol 2018;68:268–79.ArticlePubMed
- 28. Stefan N, Haring HU, Hu FB, Schulze MB. Divergent associations of height with cardiometabolic disease and cancer: epidemiology, pathophysiology, and global implications. Lancet Diabetes Endocrinol 2016;4:457–67.ArticlePubMed
- 29. Bell JA, Hamer M, Sabia S, Singh-Manoux A, Batty GD, Kivimaki M. The natural course of healthy obesity over 20 years. J Am Coll Cardiol 2015;65:101–2.ArticlePubMed
- 30. Eckel N, Li Y, Kuxhaus O, Stefan N, Hu FB, Schulze MB. Transition from metabolic healthy to unhealthy phenotypes and association with cardiovascular disease risk across BMI categories in 90 257 women (the Nurses’ Health Study): 30 year follow-up from a prospective cohort study. Lancet Diabetes Endocrinol 2018;6:714–24.ArticlePubMed
- 31. Appleton SL, Seaborn CJ, Visvanathan R, Hill CL, Gill TK, Taylor AW, et al. Diabetes and cardiovascular disease outcomes in the metabolically healthy obese phenotype: a cohort study. Diabetes Care 2013;36:2388–94.ArticlePubMedPMC
- 32. Lin L, Zhang J, Jiang L, Du R, Hu C, Lu J, et al. Transition of metabolic phenotypes and risk of subclinical atherosclerosis according to BMI: a prospective study. Diabetologia 2020;63:1312–23.ArticlePubMedPDF
- 33. Lee YB, Kim DH, Kim SM, Kim NH, Choi KM, Baik SH, et al. Hospitalization for heart failure incidence according to the transition in metabolic health and obesity status: a nationwide population-based study. Cardiovasc Diabetol 2020;19:77.ArticlePubMedPMCPDF
- 34. Morkedal B, Vatten LJ, Romundstad PR, Laugsand LE, Janszky I. Risk of myocardial infarction and heart failure among metabolically healthy but obese individuals: HUNT (Nord-Trøndelag Health Study), Norway. J Am Coll Cardiol 2014;63:1071–8.ArticlePubMed
- 35. Lavie CJ, Milani RV, Ventura HO. Disparate effects of metabolically healthy obesity in coronary heart disease and heart failure. J Am Coll Cardiol 2014;63:1079–81.ArticlePubMed
- 36. Stefan N, Fritsche A, Haring HU. Mechanisms explaining the relationship between metabolically healthy obesity and cardiovascular risk. J Am Coll Cardiol 2014;63:2748–9.ArticlePubMed
- 37. Stefan N, Haring HU, Schulze MB. Metabolically healthy obesity: the low-hanging fruit in obesity treatment? Lancet Diabetes Endocrinol 2018;6:249–58.ArticlePubMed
- 38. Friedman SL, Neuschwander-Tetri BA, Rinella M, Sanyal AJ. Mechanisms of NAFLD development and therapeutic strategies. Nat Med 2018;24:908–22.ArticlePubMedPMCPDF
- 39. Stefan N, Haring HU, Cusi K. Non-alcoholic fatty liver disease: causes, diagnosis, cardiometabolic consequences, and treatment strategies. Lancet Diabetes Endocrinol 2019;7:313–24.ArticlePubMed
- 40. Tilg H, Moschen AR, Roden M. NAFLD and diabetes mellitus. Nat Rev Gastroenterol Hepatol 2017;14:32–42.ArticlePubMedPDF
- 41. Stefan N, Haring HU. The role of hepatokines in metabolism. Nat Rev Endocrinol 2013;9:144–52.ArticlePubMedPDF
- 42. Pal D, Dasgupta S, Kundu R, Maitra S, Das G, Mukhopadhyay S, et al. Fetuin-A acts as an endogenous ligand of TLR4 to promote lipid-induced insulin resistance. Nat Med 2012;18:1279–85.ArticlePubMedPDF
- 43. Stefan N, Haring HU. Circulating fetuin-A and free fatty acids interact to predict insulin resistance in humans. Nat Med 2013;19:394–5.ArticlePubMedPDF
- 44. Stefan N, Fritsche A, Schick F, Haring HU. Phenotypes of prediabetes and stratification of cardiometabolic risk. Lancet Diabetes Endocrinol 2016;4:789–98.ArticlePubMed
- 45. Konieczna J, Yanez A, Monino M, Babio N, Toledo E, Martinez-Gonzalez MA, et al. Longitudinal changes in Mediterranean diet and transition between different obesity phenotypes. Clin Nutr 2020;39:966–75.ArticlePubMed
- 46. Bluher M. Obesity: global epidemiology and pathogenesis. Nat Rev Endocrinol 2019;15:288–98.ArticlePubMedPDF
- 47. Petrilli CM, Jones SA, Yang J, Rajagopalan H, O’Donnell L, Chernyak Y, et al. Factors associated with hospital admission and critical illness among 5279 people with coronavirus disease 2019 in New York City: prospective cohort study. BMJ 2020;369:m1966.ArticlePubMedPMC
- 48. Cariou B, Hadjadj S, Wargny M, Pichelin M, Al-Salameh A, Allix I, et al. Phenotypic characteristics and prognosis of inpatients with COVID-19 and diabetes: the CORONADO study. Diabetologia 2020;1–16.ArticlePubMedPMCPDF
- 49. Stefan N, Birkenfeld AL, Schulze MB, Ludwig DS. Obesity and impaired metabolic health in patients with COVID-19. Nat Rev Endocrinol 2020;16:341–2.ArticlePubMedPMCPDF
- 50. Zhu L, She ZG, Cheng X, Qin JJ, Zhang XJ, Cai J, et al. Association of blood glucose control and outcomes in patients with COVID-19 and pre-existing type 2 diabetes. Cell Metab 2020;31:1068–77.ArticlePubMedPMC
- 51. Eckel RH, Alberti KG, Grundy SM, Zimmet PZ. The metabolic syndrome. Lancet 2010;375:181–3.ArticlePubMed
References
Figure & Data
References
Citations
Citations to this article as recorded by 

- Phenotyping obesity: A focus on metabolically healthy obesity and metabolically unhealthy normal weight
Rachel Agius, Nikolai P. Pace, Stephen Fava
Diabetes/Metabolism Research and Reviews.2024;[Epub] CrossRef - Comparing verum and sham acupoint catgut embedding for adults with obesity: A systematic review and meta-analysis of randomized clinical trials
Jin-huan Yue, Xiao-ling Li, Yu-ying Zhang, Guan-hu Yang, Jeffrey Zhong-xue Mah, Ang Li, Wei-wei Zhao, Yu-lin Wang, Qin-hong Zhang, Jia-qi Huang
Medicine.2024; 103(4): e36653. CrossRef - Association between Weight Change and Incidence of Dyslipidemia in Young Adults: A Retrospective Cohort Study of Korean Male Soldiers
Joon-Young Yoon, Won Ju Park, Hee Kyung Kim, Ho-Cheol Kang, Cheol-Kyu Park, Wonsuk Choi
Journal of Obesity & Metabolic Syndrome.2024; 33(1): 36. CrossRef - Metabolically Healthy Obesity: An Eye-opener
Purushothaman Padmanabhan, Nagendram Dinakaran, Somnath Verma, S Keerthana
Gastroenterology, Hepatology and Endoscopy Practice.2023; 3(1): 1. CrossRef - Effect of metabolic health and obesity on all-cause death and CVD incidence in Korean adults: a retrospective cohort study
Ye-Seul Kim, Sang-Jun Shin, Yonghwan Kim, Joungyoun Kim, Hee-Taik Kang
Scientific Reports.2023;[Epub] CrossRef - Coffee and metabolic phenotypes: A cross-sectional analysis of the Japan multi-institutional collaborative cohort (J-MICC) study
Takeshi Watanabe, Kokichi Arisawa, Tien Van Nguyen, Masashi Ishizu, Sakurako Katsuura-Kamano, Asahi Hishida, Takashi Tamura, Yasufumi Kato, Rieko Okada, Rie Ibusuki, Chihaya Koriyama, Sadao Suzuki, Takahiro Otani, Teruhide Koyama, Satomi Tomida, Kiyonori
Nutrition, Metabolism and Cardiovascular Diseases.2023; 33(3): 620. CrossRef - Metabolically unhealthy phenotype in adults with normal weight: Is cardiometabolic health worse off when compared to adults with obesity?
Myong-Won Seo, Joon Young Kim
Obesity Research & Clinical Practice.2023; 17(2): 116. CrossRef - Association between metabolic obesity phenotypes and multiple myeloma hospitalization burden: A national retrospective study
Yue Zhang, Xiude Fan, Chunhui Zhao, Zinuo Yuan, Yiping Cheng, Yafei Wu, Junming Han, Zhongshang Yuan, Yuanfei Zhao, Keke Lu
Frontiers in Oncology.2023;[Epub] CrossRef - Metabolically healthy obesity: Misleading phrase or healthy phenotype?
Cem Tanriover, Sidar Copur, Abduzhappar Gaipov, Batu Ozlusen, Rustu E. Akcan, Masanari Kuwabara, Mads Hornum, Daniel H. Van Raalte, Mehmet Kanbay
European Journal of Internal Medicine.2023; 111: 5. CrossRef - Prevalence of combined metabolic health and weight status by various diagnosis criteria and association with cardiometabolic disease in Korean adults
Myong-Won Seo, Jung-Min Lee, Hyun Chul Jung
Obesity Research & Clinical Practice.2023; 17(2): 137. CrossRef - Precision medicine in complex diseases—Molecular subgrouping for improved prediction and treatment stratification
Åsa Johansson, Ole A. Andreassen, Søren Brunak, Paul W. Franks, Harald Hedman, Ruth J. F. Loos, Benjamin Meder, Erik Melén, Craig E. Wheelock, Bo Jacobsson
Journal of Internal Medicine.2023; 294(4): 378. CrossRef - Lipid droplet biogenesis and functions in health and disease
Armella Zadoorian, Ximing Du, Hongyuan Yang
Nature Reviews Endocrinology.2023; 19(8): 443. CrossRef - Molecular Mechanisms for the Vicious Cycle between Insulin Resistance and the Inflammatory Response in Obesity
Dariusz Szukiewicz
International Journal of Molecular Sciences.2023; 24(12): 9818. CrossRef - Insulin Resistance Is the Main Characteristic of Metabolically Unhealthy Obesity (MUO) Associated with NASH in Patients Undergoing Bariatric Surgery
Sophia M. Schmitz, Sebastian Storms, Alexander Koch, Christine Stier, Andreas Kroh, Karl P. Rheinwalt, Sandra Schipper, Karim Hamesch, Tom F. Ulmer, Ulf P. Neumann, Patrick H. Alizai
Biomedicines.2023; 11(6): 1595. CrossRef - Hyperleptinemia as a marker of various phenotypes of obesity and overweight in women with rheumatoid arthritis and systemic lupus erythematosus
L. V. Kondrateva, Yu. N. Gorbunova, T. A. Panafidina, T. V. Popkova
Rheumatology Science and Practice.2023; 61(3): 339. CrossRef - Predictable Representation of Metabolic Synthesis Pathways of Vitamins and Short-Chain Fatty Acids in Obese Adults
A. V. Shestopalov, L. A. Ganenko, I. M. Kolesnikova, T. V. Grigoryeva, I. Yu. Vasilyev, Yu. L. Naboka, N. I. Volkova, O. V. Borisenko, S. A. Roumiantsev
Journal of Evolutionary Biochemistry and Physiology.2023; 59(5): 1510. CrossRef - Metabolically unhealthy individuals, either with obesity or not, have a higher risk of critical coronavirus disease 2019 outcomes than metabolically healthy individuals without obesity
Nam Hoon Kim, Kyeong Jin Kim, Jimi Choi, Sin Gon Kim
Metabolism.2022; 128: 154894. CrossRef - Associations between obesity, metabolic syndrome, and endometrial cancer risk in East Asian women
Boyoung Park
Journal of Gynecologic Oncology.2022;[Epub] CrossRef - Insulin and cancer: a tangled web
Brooks P. Leitner, Stephan Siebel, Ngozi D. Akingbesote, Xinyi Zhang, Rachel J. Perry
Biochemical Journal.2022; 479(5): 583. CrossRef - Relationships Between Metabolic Body Composition Status and Rapid Kidney Function Decline in a Community-Based Population: A Prospective Observational Study
Shao-Chi Chu, Po-Hsi Wang, Kuan-Ying Lu, Chia-Chun Ko, Yun-Hsuan She, Chin-Chan Lee, I-Wen Wu, Chiao-Yin Sun, Heng-Jung Hsu, Heng-Chih Pan
Frontiers in Public Health.2022;[Epub] CrossRef - Dissecting the clinical relevance of polygenic risk score for obesity—a cross-sectional, longitudinal analysis
Eun Kyung Choe, Manu Shivakumar, Seung Mi Lee, Anurag Verma, Dokyoon Kim
International Journal of Obesity.2022; 46(9): 1686. CrossRef - Metabolic and Obesity Phenotype Trajectories in Taiwanese Medical Personnel
Hsin-Yun Chang, Jer-Hao Chang, Yin-Fan Chang, Chih-Hsing Wu, Yi-Ching Yang
International Journal of Environmental Research and Public Health.2022; 19(13): 8184. CrossRef - Sex Differences in Cardiovascular Impact of Early Metabolic Impairment: Interplay between Dysbiosis and Adipose Inflammation
Haneen S. Dwaib, Ibrahim AlZaim, Ghina Ajouz, Ali H. Eid, Ahmed El-Yazbi
Molecular Pharmacology.2022; 102(1): 60. CrossRef - Reduced leukocyte mitochondrial copy number in metabolic syndrome and metabolically healthy obesity
Rachel Agius, Nikolai Paul Pace, Stephen Fava
Frontiers in Endocrinology.2022;[Epub] CrossRef - Changes in BMI and physical activity from youth to adulthood distinguish normal-weight, metabolically obese adults from those who remain healthy
A. Viitasalo, K. Pahkala, T. Lehtimäki, JSA. Viikari, TH. Tammelin, O. Raitakari, TO. Kilpeläinen
Frontiers in Endocrinology.2022;[Epub] CrossRef - Pathogenesis, Murine Models, and Clinical Implications of Metabolically Healthy Obesity
Yun Kyung Cho, Yoo La Lee, Chang Hee Jung
International Journal of Molecular Sciences.2022; 23(17): 9614. CrossRef - Metabolically healthy obesity: it is time to consider its dynamic changes
Yun Kyung Cho, Chang Hee Jung
Cardiovascular Prevention and Pharmacotherapy.2022; 4(4): 123. CrossRef - Obesity as a Risk Factor for Breast Cancer—The Role of miRNA
Karolina Hanusek, Jakub Karczmarski, Anna Litwiniuk, Katarzyna Urbańska, Filip Ambrozkiewicz, Andrzej Kwiatkowski, Lidia Martyńska, Anita Domańska, Wojciech Bik, Agnieszka Paziewska
International Journal of Molecular Sciences.2022; 23(24): 15683. CrossRef - Propensity Score–Matching Sleeve Gastrectomy (SG) vs. Gastric Bypass (RYGB) in Patients ≥ 60 Years
Omar Thaher, Stefanie Wolf, Martin Hukauf, Christine Stroh
Obesity Surgery.2021; 31(6): 2682. CrossRef - Associations between obesity, metabolic health, and the risk of breast cancer in East Asian women
Boyoung Park, Soyeoun Kim, Hayoung Kim, Chihwan Cha, Min Sung Chung
British Journal of Cancer.2021; 125(12): 1718. CrossRef

 KES
KES
 PubReader
PubReader ePub Link
ePub Link Cite
Cite