Articles
- Page Path
- HOME > Endocrinol Metab > Volume 34(3); 2019 > Article
-
Original ArticleClinical Study Changes in Thyroid Peroxidase and Thyroglobulin Antibodies Might Be Associated with Graves' Disease Relapse after Antithyroid Drug Therapy
 Audioslide
Audioslide -
Yun Mi Choi
 , Mi Kyung Kwak, Sang Mo Hong, Eun-Gyoung Hong
, Mi Kyung Kwak, Sang Mo Hong, Eun-Gyoung Hong
-
Endocrinology and Metabolism 2019;34(3):268-274.
DOI: https://doi.org/10.3803/EnM.2019.34.3.268
Published online: September 26, 2019
Department of Internal Medicine, Hallym University Dongtan Sacred Heart Hospital, Hallym University College of Medicine, Hwaseong, Korea.
- Corresponding author: Eun-Gyoung Hong. Department of Internal Medicine, Hallym University Dongtan Sacred Heart Hospital, Hallym University College of Medicine, 7 Keunjaebong-gil, Hwaseong 18450, Korea. Tel: +82-31-8086-2493, Fax: +82-31-8086-2499, hegletter@hallym.or.kr
• Received: March 29, 2019 • Revised: May 20, 2019 • Accepted: June 18, 2019
Copyright © 2019 Korean Endocrine Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
-
Background
- Graves' disease (GD) is an autoimmune thyroid disorder caused by antibodies stimulating the thyrotropin (TSH) receptor. TSH receptor antibody (TRAb) measurement is useful for predicting GD relapse after antithyroid drug (ATD) treatment. However, the association of other thyroid autoantibodies with GD relapse remains obscure.
-
Methods
- This retrospective study enrolled patients with GD who were initially treated with ATD. TRAb, thyroid peroxidase antibody (TPOAb), and thyroglobulin antibody (TgAb) were measured at the initial diagnosis and at the time of ATD discontinuation.
-
Results
- A total of 55 patients were enrolled. The mean age was 49.7 years, and 39 patients (70.9%) were female. Antibody positivity at diagnosis was 90.9%, 69.1%, and 61.9% for TRAb, TPOAb, TgAb, respectively. Median ATD treatment period was 15.1 months. At the time of ATD withdrawal, TRAb titers decreased uniformly overall. Conversely, TPOAb and TgAb showed various changes. After withdrawal of ATD, 19 patients (34.5%) experienced relapse. No clinical features or laboratory results were significantly related to relapse in the overall patient group. However, in the TPOAb positive group at diagnosis, increasing titer of TPOAb or TgAb after ATD treatment was significantly and independently related to relapse free survival (TPOAb: hazard ratio [HR], 17.99; 95% confidence interval [CI], 1.66 to 195.43; P=0.02) (TgAb: HR, 5.73; 95% CI, 1.21 to 27.26; P=0.03).
-
Conclusion
- Changes in TPOAb or TgAb titers during treatment might be useful for predicting relapse after ATD treatment in patients with positive TPOAb at diagnosis.
- Graves' disease (GD) is an autoimmune thyroid disorder caused by antibodies stimulating the thyrotropin (TSH) receptor on thyroid follicular cells [1]. One standard treatment for GD includes antithyroid drugs (ATD) usage for 12 to 18 months course [2]. This offers the possibility of disease remission without post-interventional hypothyroidism. However, risk for relapse is high and patients may experience side effects [2].
- ATD may also have immunomodulating effects associated with normalization of TSH receptor antibody (TRAb) levels over time, which is potentially important in mediating remissions after therapy [3]. In addition, thyroid peroxidase antibody (TPOAb) or thyroglobulin antibody (TgAb) changes during ATD treatment also had been reported [4]. The remission rate varies among ethnicities and geographic areas [5]. In patients with normalized TRAb, relapse rates are 20% to 30% over 3 to 5 years of follow-up [1]. Overall, the risk of relapse after a 1 to 2 year course of ATD therapy is around 50% [67].
- Some previously identified predictors of relapse are younger age, larger goiters, smoking, male sex, severe hyperthyroidism, higher concentration of antibodies, and presence of Graves' ophthalmopathy [2789]. The usefulness of TRAb measurement at the time of ATD withdrawal for predicting a later relapse of hyperthyroidism has been studied intensively [10111213]. However, the effect of TPOAb or TgAb on the course of GD after ATD withdrawal remains unclear.
- The aim of this study was to investigate the effect of autoimmune antibodies on risk of GD relapse in patients who underwent ATD treatment.
INTRODUCTION
- Subjects
- This retrospective study enrolled patients with GD who were treated with ATD between 2009 and 2017 at Hallym University Dongtan Sacred Heart Hospital and Hallym University Kangnam Sacred Heart Hospital, Korea. A total of 55 patients with newly diagnosed GD were enrolled. GD was diagnosed on the basis of medical history, clinical findings, serum thyroid hormone levels, serum TRAb titer, and increased thyroidal 99m-technetium uptake [14]. Clinical characteristics and autoantibody status were evaluated at the time of diagnosis and at the time of ATD withdrawal. Patients treated with methimazole or carbimazole were analyzed together. The study was approved by the Institutional Review Board of Hallym Sacred Heart Hospital, Korea (IRB No. 2018-04-015). The IRB approved the waiver of informed consent.
- Definition
- ATD therapy was administered using a dose titrating regimens. ATD was withdrawn when TSH, free thyroxine (T4), and TRAb were normalized for 6 months period. If TRAb have not reached normal range, ATD was withdrawn when euthyroid state was maintained over 6 months with the minimum maintenance dose of ATDs (1 tablet or less in a day of each drug [methimazole 5 mg, carbimazole 5 mg, and propylthiouracil 50 mg]). Remission was defined as euthyroid status for more than 6 months after ATD withdrawal. Recurrence was defined as reappearance of hyperthyroidism (including subclinical) with or without increasing TRAb.
- Laboratory measurements
- Thyroid function tests were measured with an automated direct chemiluminescent method (Advia Centaur XP, Siemens Healthcare Diagnostics, Munich, Germany; normal range: TSH 0.55 to 4.78 µIU/mL, free T4 0.89 to 1.76 ng/dL, and total triiodothyronine [T3] 60 to 181 ng/dL). Thyroid autoantibody assays were also performed by an automated chemiluminescent method (Liaison XL, DiaSorin, Saluggia, Italy; normal range: TRAb 0 to 1.75 IU/L, TPOAb 0 to 60 U/mL, and TgAb 0 to 100 IU/mL).
- TRAb, TPOAb, and TgAb were measured at the initial diagnosis and at the time of ATD withdrawal.
- Statistical analyses
- R version 3.0 and R libraries prodlim, car, Cairo, and survival were used to analyze data and draw graphs (R Foundation for Statistical Computing, Vienna, Austria, https://www.r-project.org/). Continuous variables are presented as medians with interquartile ranges (IQRs). Categorical variables are presented as numbers with percentages. The Student's t test or Wilcoxon rank-sum test was used to compare continuous variables. The chi-square test or Fisher's exact test was used to compare categorical variables. Survival curves were constructed using the Kaplan-Meier method, and the log-rank test was used to evaluate differences in survival between groups. A Cox proportional hazards model with and 95% confidence intervals (CIs) was used to evaluate the risk of recurrence. All P values were 2-sided, and P<0.05 was considered statistically significant.
METHODS
- Baseline characteristics and evolution of autoantibodies
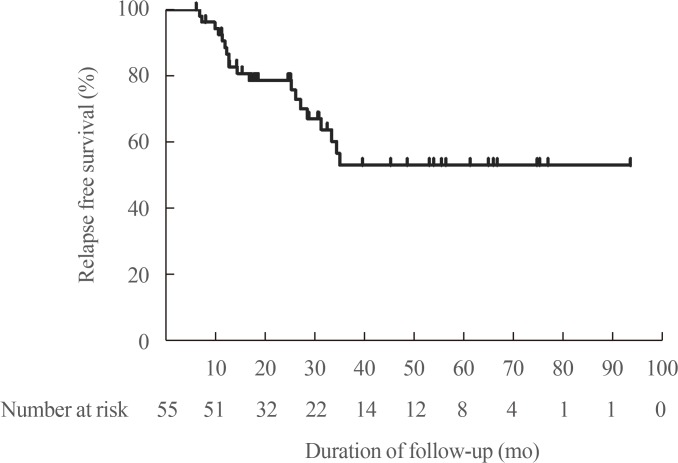
- This retrospective study initially screened patients with TRAb, TPOAb, and TgAb measured simultaneously at the time of diagnosis and, if possible, at the time of ATD withdrawal. A total of 108 patients were screened. Among them, only newly diagnosed patients were enrolled (n=103). Patients who could not stop ATD (n=6) or who were lost before ATD withdrawal (n=9) were excluded. And those who did not reached 6 months after ATD withdrawal (n=11) or recurred in less than 6 months (n=22) were also excluded. Finally, a total of 55 subjects were enrolled for this study. The mean age was 49.7 years, and 39 patients (70.9%) were female. Thyroid function tests and the prevalence of each autoantibody are summarized in Table 1 according to time point in the treatment course. Methimazole, carbimazole, and propylthiouracil was used in 80%, 7.3%, and 12.7% of patients, respectively. The median follow-up duration was 56.3 months (IQR, 37.0 to 97.3). The median duration of ATD therapy to remission was 15.1 months (IQR, 11.3 to 24.8). During a median of 32.9 months of observation after ATD withdrawal, 19 patients (34.5%) had a relapse of hyperthyroidism.
- At the time of diagnosis, almost all patients had positive TRAb level. Conversely, TPOAb or TgAb positivity was not that high. At the time of ATD withdrawal, TRAb titers decreased uniformly overall. Conversely, TPOAb and TgAb showed various changes (Fig. 1).
- Clinical and laboratory features associated with relapse of Graves' disease
- After withdrawal of ATD, 19 patients (34.5%) experienced relapse. The median duration from ATD withdrawal to relapse was 14.7 months (Fig. 2). Table 2 shows clinical and laboratory features of patients according to relapse status. Table 3 shows clinical and laboratory factors associated with GD relapse. There were no clinical features or laboratory results significantly related to relapse. Autoantibody positive status at diagnosis and at ATD withdrawal both had no influence on relapse of GD. Thirteen patients respectively showed increasing TPOAb and TgAb titer during treatment. But increasing antibody titers after treatment with ATD (not applicable for TRAb) did not significantly relate to GD relapse in overall patients (TPOAb: hazard ratio [HR], 2.22; 95% CI, 0.75 to 6.64; P=0.49) (TgAb: HR, 2.05; 95% CI, 0.65 to 6.53; P=0.96) (Table 3).
- Clinical and laboratory features associated with relapse of Graves' disease in subjects with positive TPOAb at diagnosis
- In the TPOAb positive group at diagnosis (n=38), increased TPOAb and TgAb titer were shown in five and eight patients. Four out of five (increasing TPOAb) and four out of eight (increasing TgAb) experienced recurrence, compared with six out of 23 and five out of 19 in stable or decreased TPOAb and TgAb titer group, respectively. This increased TPOAb (n=5) or TgAb (n=8) titers after treatment with ATD were significantly related to GD relapse independent of thyroid function and autoantibody status (TPOAb: HR, 17.99; 95% CI, 1.66 to 195.43; P=0.02) (TgAb: HR, 5.73; 95% CI, 1.21 to 27.26; P=0.03) (Table 4). Patients with increasing TPOAb or TgAb titers showed significantly shorter relapse free survival (Fig. 3).
- In the TgAb positive group at diagnosis (n=34), increased TPOAb (n=10) or TgAb (n=9) titer after treatment with ATD was not significantly related to GD relapse (TPOAb increase: HR, 2.38; 95% CI, 0.57 to 9.97; P=0.24) (TgAb increase: HR, 1.30; 95% CI, 0.31 to 5.50; P=0.72).
RESULTS
- The risk of GD relapse after withdrawal of ATD therapy is highly variable among patients [6]. In patients with high risk of disease recurrence, definitive ablative therapy is recommended. Identification of patients who require definitive therapy at an early stage may be beneficial [15]. Numerous previous studies reported factors that may be associated with risk of hyperthyroidism relapse. These studies mostly focused on the usefulness of TRAb or thyroid stimulating antibody (TSAb) for predicting relapse [1011]. However, the role of TPOAb or TgAb in GD relapse is not well known. This study shows that TPOAb and TgAb do not change uniformly during treatment with ATD. In TPOAb positive patients at diagnosis, increased TPOAb or TgAb titers during treatment might be related to shorter relapse free survival, independent of thyroid function or TRAb change.
- The main clinical manifestations of autoimmune thyroid diseases are GD and Hashimoto's thyroiditis [15]. Although the presence of TPOAb or TgAb is a hallmark of Hashimoto's thyroiditis, both antibodies are also present in many patients with GD [16]. In some previous studies, GD patients with high TPOAb titers had a high relapse rate [161718]. These studies suggest that in GD, TPOAb may represent a secondary immune process rather than a sign of antibody-dependent cell-mediated cytotoxicity [16]. The immunosuppressive action of ATD was previously reported [319]. One study reported that during ATD treatment, TPOAb values significantly decreased, as observed with TSAb, but they increased once again after drug withdrawal [4]. Increasing TPOAb titers during ATD therapy, even with decreasing TRAb, could represent greater disease severity and higher likelihood of relapse. Another study reported that patients with well-controlled hyperthyroidism had a greater decrease in TPOAb level than patients who were still hyperthyroid during ATD treatment [2021]. Marcocci et al. [20] also showed that increased TPOAb titer after withdraw of ATD is associated with increased risk of relapse. This may indicate that TSAb increases TPOAb titers by mediating an increase in microsomal antigen [1721]. However, some other studies showed opposite results [142223]. Those reports showed decreased recurrence or even progression to hypothyroidism after ATD therapy in patients with high TPOAb or TbAb titers. GD may evolve gradually into chronic thyroiditis, leading to hypothyroidism. Another study showed significantly shorter T3 normalization time in the group with higher TPOAb [24]. The discrepancy between studies on the role of TPOAb and TgAb for predicting relapse might be due to several reasons. First, the duration and treatment protocols of ATD therapy differed. Second, the initial autoantibody status was not considered in previous studies.
- Relapse of hyperthyroidism is a prominent drawback of ATD therapy [6]. Patients may suffer from repeated recurrence. It is important to identify factors that may be associated with relapse after withdrawal of ATD therapy. Previous studies did not focus on TPOAb or TgAb status or changes regarding disease relapse. In Korea, repeated measurement of TPOAb or TgAb during GD treatment is not recommended because it is not cost-effective. The current study showed new insights into the role of TPOAb or TgAb on GD relapse. These factors might help to guide early ablative treatment for those with very high relapse risk. Our study had an inherent limitation because of its retrospective design. There was no uniform protocol for the titration and duration of ATD treatment. There was a relatively small number of study subjects. Lastly, we did not consider other clinical factors such as smoking status, goiter size, and presence of thyroid-associated ophthalmopathy that might affect disease recurrence.
- In summary, we conclude that increase of TPOAb or TgAb titer during ATD therapy, in patients with positive TPOAb at diagnosis, might have a higher likelihood of disease relapse.
DISCUSSION
-
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.
-
AUTHOR CONTRIBUTIONS:
Article information
- 1. Burch HB, Cooper DS. Management of Graves disease: a review. JAMA 2015;314:2544–2554. ArticlePubMed
- 2. Struja T, Fehlberg H, Kutz A, Guebelin L, Degen C, Mueller B, et al. Can we predict relapse in Graves' disease? Results from a systematic review and meta-analysis. Eur J Endocrinol 2017;176:87–97. ArticlePubMed
- 3. Laurberg P. Remission of Graves' disease during anti-thyroid drug therapy. Time to reconsider the mechanism? Eur J Endocrinol 2006;155:783–786. ArticlePubMed
- 4. Guilhem I, Massart C, Poirier JY, Maugendre D. Differential evolution of thyroid peroxidase and thyrotropin receptor antibodies in Graves' disease: thyroid peroxidase antibody activity reverts to pretreatment level after carbimazole withdrawal. Thyroid 2006;16:1041–1045. ArticlePubMed
- 5. Moon JH, Yi KH. The diagnosis and management of hyperthyroidism in Korea: consensus report of the Korean Thyroid Association. Endocrinol Metab (Seoul) 2013;28:275–279. ArticlePubMedPMC
- 6. Laurberg P, Krejbjerg A, Andersen SL. Relapse following antithyroid drug therapy for Graves' hyperthyroidism. Curr Opin Endocrinol Diabetes Obes 2014;21:415–421. ArticlePubMed
- 7. Wiersinga WM. Graves' disease: can it be cured? Endocrinol Metab (Seoul) 2019;34:29–38. ArticlePubMedPMC
- 8. Smith TJ, Hegedus L. Graves' disease. N Engl J Med 2016;375:1552–1565. ArticlePubMed
- 9. Liu J, Fu J, Xu Y, Wang G. Antithyroid drug therapy for Graves' disease and implications for recurrence. Int J Endocrinol 2017;2017:3813540. ArticlePubMedPMCPDF
- 10. Kwon H, Kim WG, Jang EK, Kim M, Park S, Jeon MJ, et al. Usefulness of measuring thyroid stimulating antibody at the time of antithyroid drug withdrawal for predicting relapse of Graves disease. Endocrinol Metab (Seoul) 2016;31:300–310. ArticlePubMedPMC
- 11. Carella C, Mazziotti G, Sorvillo F, Piscopo M, Cioffi M, Pilla P, et al. Serum thyrotropin receptor antibodies concentrations in patients with Graves' disease before, at the end of methimazole treatment, and after drug withdrawal: evidence that the activity of thyrotropin receptor antibody and/or thyroid response modify during the observation period. Thyroid 2006;16:295–302. ArticlePubMed
- 12. Choi HS, Yoo WS. Free thyroxine, anti-thyroid stimulating hormone receptor antibody titers, and absence of goiter were associated with responsiveness to methimazole in patients with new onset Graves' disease. Endocrinol Metab (Seoul) 2017;32:281–287. ArticlePubMedPMC
- 13. Huh JE, Suk JH, Kim MK, Choi IJ, Son SM, Kim IJ, et al. Clinical usefulness of the second generation TSH-binding inhibitory immunoglobulin assay using recombinant human TSH receptor in patients with Graves' disease. J Korean Endocr Soc 2008;23:179–185.Article
- 14. Takaichi Y, Tamai H, Honda K, Nagai K, Kuma K, Nakagawa T. The significance of antithyroglobulin and antithyroidal microsomal antibodies in patients with hyperthyroidism due to Graves' disease treated with antithyroidal drugs. J Clin Endocrinol Metab 1989;68:1097–1100. ArticlePubMedPDF
- 15. Smycznyska J, Cyniak-Magierska A, Stasiak M, Karbownik-Lewinska M, Lewinski A. Persistent remission of Graves' disease or evolution from Graves' disease to Hashimoto's thyroiditis in childhood: a report of 6 cases and clinical implications. Neuro Endocrinol Lett 2014;35:335–341. PubMed
- 16. Schott M, Eckstein A, Willenberg HS, Nguyen TB, Morgenthaler NG, Scherbaum WA. Improved prediction of relapse of Graves' thyrotoxicosis by combined determination of TSH receptor and thyroperoxidase antibodies. Horm Metab Res 2007;39:56–61. ArticlePubMedPDF
- 17. Hamada N, Ito K, Mimura T, Ishikawa N, Momotani N, Noh J, et al. Retrospective reevaluation of the significance of thyroid microsomal antibody in the treatment of Graves' disease. Acta Endocrinol (Copenh) 1987;114:328–335. ArticlePubMed
- 18. Romaldini JH, Bromberg N, Werner RS, Tanaka LM, Rodrigues HF, Werner MC, et al. Comparison of effects of high and low dosage regimens of antithyroid drugs in the management of Graves' hyperthyroidism. J Clin Endocrinol Metab 1983;57:563–570. ArticlePubMedPDF
- 19. Weetman AP. The immunomodulatory effects of antithyroid drugs. Thyroid 1994;4:145–146. ArticlePubMed
- 20. Marcocci C, Chiovato L, Mariotti S, Pinchera A. Changes of circulating thyroid autoantibody levels during and after the therapy with methimazole in patients with Graves' disease. J Endocrinol Invest 1982;5:13–19. ArticlePubMedPDF
- 21. Chiovato L, Vitti P, Lombardi A, Kohn LD, Pinchera A. Expression of the microsomal antigen on the surface of continuously cultured rat thyroid cells is modulated by thyrotropin. J Clin Endocrinol Metab 1985;61:12–16. ArticlePubMedPDF
- 22. Stefanic M, Karner I. Thyroid peroxidase autoantibodies are associated with a lesser likelihood of late reversion to hyperthyroidism after successful non-ablative treatment of Graves' disease in Croatian patients. J Endocrinol Invest 2014;37:71–77. ArticlePubMedPDF
- 23. Katahira M, Ogata H. Thyroglobulin autoantibodies are associated with refractoriness to antithyroid drug treatment for Graves' disease. Intern Med 2016;55:1519–1524. ArticlePubMed
- 24. Hwang SM, Kim MS, Lee DY. Predictive factors for early response to methimazole in children and adolescents with Graves disease: a single-institute study between 1993 and 2013. Ann Pediatr Endocrinol Metab 2016;21:70–74. ArticlePubMedPMCPDF
References
Fig. 1
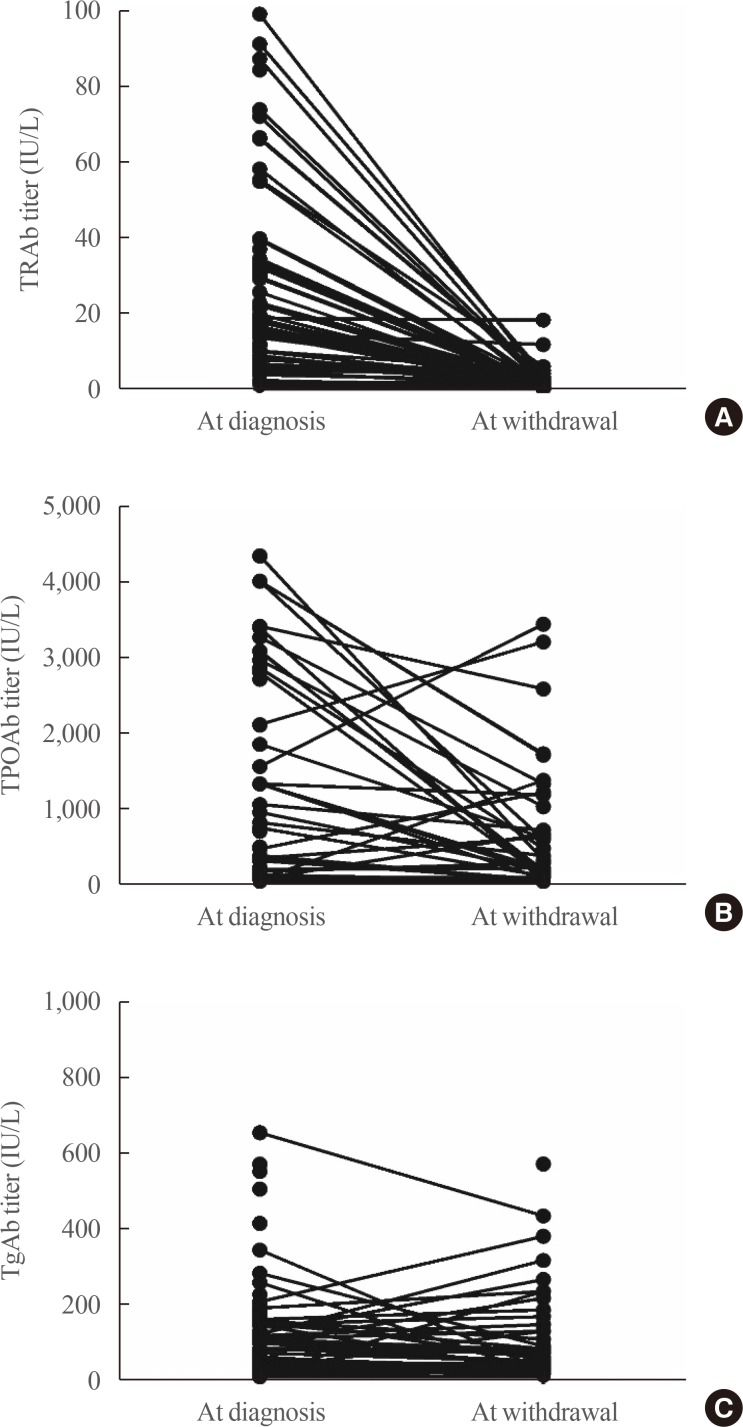
Changes in (A) thyrotropin (TSH) receptor antibody (TRAb), (B) thyroid peroxidase antibody (TPOAb), and (C) thyroglobulin antibody (TgAb) during treatment with antithyroid drug in patients with Graves' disease.

Fig. 3
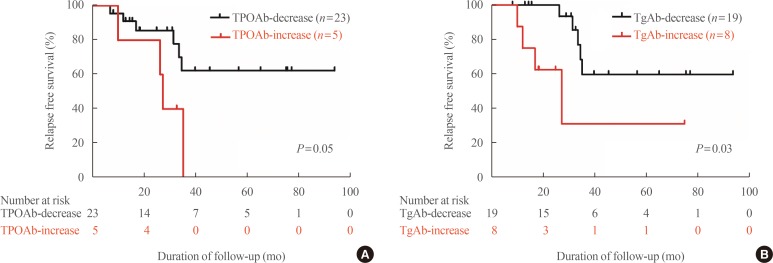
Relapse free survival of Graves' disease patients with positive thyroid peroxidase antibody (TPOAb) at diagnosis, according to (A) TPOAb changes and (B) thyroglobulin antibody (TgAb) changes.

Table 1
![enm-34-268-i001.jpg]()
Baseline Characteristics
Table 2
![enm-34-268-i002.jpg]()
Clinical and Laboratory Features Associated with Relapse of Graves' Disease
Table 3
![enm-34-268-i003.jpg]()
Clinical Features and Changes in Autoantibodies Associated with Relapse of Graves' Disease
Table 4
![enm-34-268-i004.jpg]()
Clinical Features and Changes in Autoantibodies Associated with Relapse of Graves' Disease in Initially TPOAb Positive Patients (n=38)
Figure & Data
References
Citations
Citations to this article as recorded by 

- Enhanced predictive validity of integrative models for refractory hyperthyroidism considering baseline and early therapy characteristics: a prospective cohort study
Xinpan Wang, Tiantian Li, Yue Li, Qiuyi Wang, Yun Cai, Zhixiao Wang, Yun Shi, Tao Yang, Xuqin Zheng
Journal of Translational Medicine.2024;[Epub] CrossRef - Clinical significance of thyroglobulin antibodies and thyroid peroxidase antibodies in Graves’ disease: a cross-sectional study
Masahito Katahira, Taku Tsunekawa, Akira Mizoguchi, Mariko Yamaguchi, Kahori Tsuru, Hiromi Takashima, Ryoma Terada
Hormones.2023; 22(2): 253. CrossRef - The Clinical Implications of Anti-thyroid Peroxidase Antibodies in Graves’ Disease in Basrah
Emad S Alhubaish, Nassar T Alibrahim, Abbas A Mansour
Cureus.2023;[Epub] CrossRef - Influence of Thyroid Peroxidase Antibodies Serum Levels in Graves' Disease: A Retrospective Cohort Study
Maria L Guia Lopes, Carlos Tavares Bello, José P Cidade, Clotilde Limbert, Joao Sequeira Duarte
Cureus.2023;[Epub] CrossRef - Interpretation of Thyroid Autoantibodies in Hyperthyroidism
Han-Sang Baek, Dong-Jun Lim
The Korean Journal of Medicine.2023; 98(3): 132. CrossRef - Usefulness of Real-Time Quantitative Microvascular Ultrasonography for Differentiation of Graves’ Disease from Destructive Thyroiditis in Thyrotoxic Patients
Han-Sang Baek, Ji-Yeon Park, Chai-Ho Jeong, Jeonghoon Ha, Moo Il Kang, Dong-Jun Lim
Endocrinology and Metabolism.2022; 37(2): 323. CrossRef - Differential Diagnosis of Thyrotoxicosis by Machine Learning Models with Laboratory Findings
Jinyoung Kim, Han-Sang Baek, Jeonghoon Ha, Mee Kyoung Kim, Hyuk-Sang Kwon, Ki-Ho Song, Dong-Jun Lim, Ki-Hyun Baek
Diagnostics.2022; 12(6): 1468. CrossRef - The relationship between atherosclerotic disease and relapse during ATD treatment
Xinxin Zhu, Yaguang Zhang, Xiaoyu Zhao, Xiaona Zhang, Zixuan Ru, Yanmeizhi Wu, Xu Yang, Boyu Hou, Hong Qiao
Frontiers in Cardiovascular Medicine.2022;[Epub] CrossRef - Programmed Cell Death-Ligand 1 (PD-L1) gene Single Nucleotide Polymorphism in Graves’ Disease and Hashimoto’s Thyroiditis in Korean Patients
Jee Hee Yoon, Min-ho Shin, Hee Nam Kim, Wonsuk Choi, Ji Yong Park, A Ram Hong, Hee Kyung Kim, Ho-Cheol Kang
Endocrinology and Metabolism.2021; 36(3): 599. CrossRef - Low frequency of IL-10-producing B cells and high density of ILC2s contribute to the pathological process in Graves’ disease, which may be related to elevated-TRAb levels
Xiaoyun Ji, Jie Wan, Rong Chen, Huixuan Wang, Lan Huang, Shwngjun Wang, Zhaoliang Su, Huaxi Xu
Autoimmunity.2020; 53(2): 78. CrossRef - Implication of VDR rs7975232 and FCGR2A rs1801274 gene polymorphisms in the risk and the prognosis of
autoimmune thyroid diseases in the Tunisian population
S Mestiri, I Zaaber, I Nasr, H Marmouch
Balkan Journal of Medical Genetics.2020; 23(1): 69. CrossRef - Thyroid Peroxidase Antibody Positivity is Associated With Relapse-Free Survival Following Antithyroid Drug Treatment for Graves Disease
Christopher A. Muir, Graham R.D. Jones, Jerry R. Greenfield, Andrew Weissberger, Katherine Samaras
Endocrine Practice.2020; 26(9): 1026. CrossRef - Predicting the Risk of Graves Disease Relapse: Commentary on “Thyroid Peroxidase Antibody Positivity is Associated with Relapse-Free Survival Following Antithyroid Drug Treatment for Graves Disease”
D. Gallo, M.L. Tanda, E. Piantanida
Endocrine Practice.2020; 26(9): 1039. CrossRef

 KES
KES
 PubReader
PubReader ePub Link
ePub Link Cite
Cite