A Comparison of Ultrasound-Guided Fine Needle Aspiration versus Core Needle Biopsy for Thyroid Nodules: Pain, Tolerability, and Complications
Article information
Abstract
Background
To compare pain, tolerability, and complications associated with fine needle aspiration (FNA) versus core needle biopsy (CNB).
Methods
FNAs were performed using 23-gauge needles and CNBs were performed using 18-gauge double-action spring-activated needles in 100 patients for each procedure. Patients were asked to record a pain score using a 10-cm visual analog scale and procedure tolerability. Complications and number of biopsies were recorded.
Results
The median pain scores were similar for the FNA and CNB approaches during and 20 minutes after the biopsy procedures (3.7 vs. 3.6, P=0.454; 0.9 vs. 1.1, P=0.296, respectively). The procedure was tolerable in all 100 FNA patients and in 97 CNB patients (P=0.246). The mean number of biopsies was fewer in the CNB group (1.4 vs. 1.2, P=0.002). By subgroup analysis (staff vs. non-staff), no significant difference was detected in any parameter. There were no major complications in either group, but three patients who underwent CNB had minor complications (P=0.246).
Conclusion
FNA and CNB show no significant differences for diagnosing thyroid nodules in terms of pain, tolerability, or complications.
INTRODUCTION
Ultrasound (US)-guided fine needle aspiration (FNA) has been suggested to be a cost-effective and safe diagnostic procedure for assessing thyroid nodules and US-guided FNA has been established as the method of choice for this purpose [1]. However, FNA shows non-diagnostic results in 10% to 42% of cases [234], and atypia of undetermined significance or follicular lesions of undetermined cause in 3% to 18% of the thyroid nodules tested [456]. Despite these known limitations of FNA, the optimal management of these nodules has not yet been clearly established.
Recently, core needle biopsy (CNB) has been suggested as an alternative to FNA. CNB has shown efficacy for testing thyroid nodules that show non-diagnostic [7], or indeterminate [89] FNA results, and for clinically suspected lymphoma or anaplastic carcinoma [10]. Additionally, CNB is effective in the diagnosis of calcified thyroid nodules [11] and follicular neoplasms [1213]. Although CNB has been suggested to be an effective procedure [14], assessments of its safety and tolerability in previous publications have been limited [1516]. On our present study therefore we evaluated the pain, tolerability, and complications associated with CNB in comparison with FNA.
METHODS
Patients
This retrospective study was approved by the Institutional Review Board of Asan Medical Center (S2013-1765-0001) and informed consent was obtained from all patients before each procedure. From September to November 2013, 100 consecutive patients (male:female, 17:83; mean age, 51.3 years [range, 28 to 78]) who underwent FNA and 100 consecutive patients (male: female, 13:87; mean age, 49.7 years [range, 26 to 77]) who received CNB at our hospital (total cohort: male:female, 30:170; mean age, 50.5 years [range, 26 to 78]) were retrospectively evaluated.
US-guided FNA and CNB procedures
US examinations were performed using one of three systems: an iU 22 (Philips Medical Systems, Bothell, WA, USA); an EUB-7500 (Hitachi Medical Systems, Tokyo, Japan); or an Aixplorer (Super Sonic Imagine S.A., Aix-en-Provence, France) equipped with a linear, high frequency probe (5 to 14 MHz). All US examinations and US-guided FNA or CNB procedures were performed by experienced staff, and by non-staff (fellows, or resident radiologists) under the supervision of two experienced thyroid radiologists with 18 and 12 years of thyroid US experience, respectively. Whether to perform CNB or FNA was determined according to the referring physicians' preference. After evaluation of thyroid and perithyroidal structures, including the vessels or esophagus, we measured the size of the nodule [17]. FNAs were performed using a 10-mL plastic syringe attached to a conventional 23-gauge needle with a combination of capillary and aspiration techniques that were selected according to the nodule characteristics. No local anesthesia was performed before performing FNA. US-guided CNB was performed using a disposable 18-gauge double-action spring-activated needle (1.1- or 1.6-cm excursion; TSK Ace-cut, Create Medic, Yokohama, Japan). Local anesthesia with 1% lidocaine was selectively performed before performing CNB [7918]. Using a freehand technique, the core of the needle was advanced from the isthmus of the thyroid towards the target nodule using the transisthmic approach [19]. When the needle tip was advanced into the edge of the nodule, the stylet and cutting cannula of the needle were sequentially fired. A second FNA or CNB was performed when adequate tissue was not obtained based on a visual inspection [7920]. The number of biopsies and types of core needles were recorded in our radiology US-guided biopsy report.
Questionnaire
Immediately after the FNA or CNB procedure, patients were asked by one author to rate their pain on a 10-cm visual analog scale (0 to 10 cm) with “0” representing “no pain” and “10” representing the “the worst pain imaginable.” After compression of the biopsy site for 20 minutes, patients were asked again to rate their pain on a 10-cm visual analog scale (0 to 10 cm) and also whether the procedure was tolerable or not.
Statistical analyses
Statistical analyses were performed using SPSS version 18.0 for Windows (SPSS Inc., Chicago, IL, USA). We analyzed and compared parameters that included pain score, the number of biopsies, complications, tolerability, and non-diagnostic pathological results between the FNA and CNB groups. We also performed subgroup analyses between staff and non-staff groups. The chi-square test or Fisher exact test for categorical variables and the Mann-Whitney U test for quantitative variables were used to compare study groups. Statistical significance was accepted for P<0.05.
RESULTS
All patients underwent FNA or CNB for one nodule respectively. All 200 patients also completed questionnaires to evaluate the level of pain associated with FNA or CNB. Both staff and non-staff performed the FNA (75 by non-staff, 25 by staff) or CNB (30 by non-staff, 70 by staff) procedures.
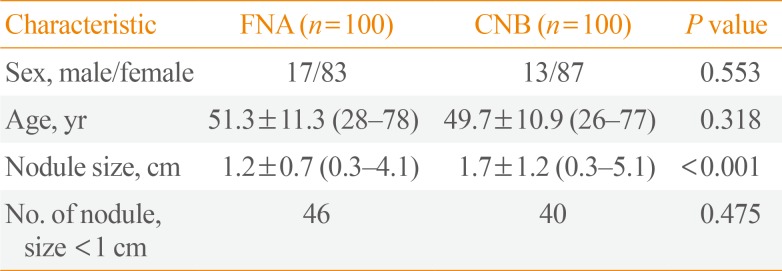
Demographic data for the patients who underwent FNA or CNB are summarized in Table 1. There were no significant differences in either gender or age between the two groups (P=0.553 and P=0.318, respectively). The mean size of nodules of FNA group was statistically significantly smaller than that of CNB group (1.2 cm vs. 1.7 cm, P<0.001). However, the number of nodules <1 cm between the FNA and CNB patients showed no significant difference (46 vs. 40, P=0.475).
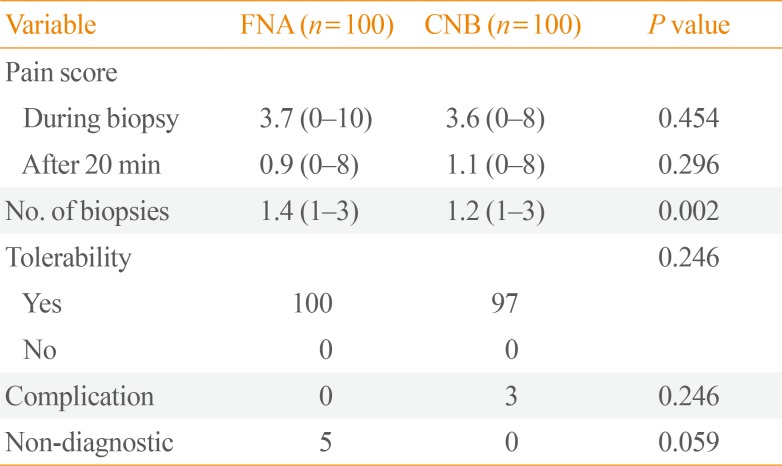
Table 2 details the questions and patient responses following both FNA and CNB procedures. Regarding the mean pain score during and at 20 minutes after the procedures, there were no significant differences between the two groups (3.7 vs. 3.6, P=0.454; 0.9 vs. 1.1, P=0.296, respectively). Regarding procedure-related complications and tolerability, there was also no difference detected between the groups. Regarding the mean number of biopsies, this was greater in the FNB group (1.4 vs. 1.2, P=0.002). Although we detected a tendency for FNA to be more frequently associated with non-diagnostic pathological results than CNB, this was not significant (5 vs. 0, P=0.059).
We performed subgroup analysis stratified by staff and nonstaff (Table 3). There were no significant differences for any parameter analyzed for the two biopsy methods, including pain score, biopsy number, tolerability, complications, and non-diagnostic pathological results. However, the number of biopsies for non-staff was less than that for staff in the FNA group (P=0.04).
Complications
There were no major complications in either the FNA or CNB groups, and no patient required hospital admission or interventional treatment. In the case of the CNB group, there were three minor complications, including perithyroidal hemorrhage and parenchyma edema; however, there were no significant differences with regard to this parameter between the groups (P=0.246). In the patients with hemorrhage and parenchymal edema, the symptoms were relieved following manual compression from 30 minutes to 2 hours. After compression, US evaluations showed the nearly complete disappearance of the hematoma or edema. There have been no delayed complications reported during the follow-up period in our current patient series.
DISCUSSION
We observed no significant differences in terms of pain, tolerability, or complications between the FNA and CNB procedures. The rate of non-diagnostic pathologic result was lower in CNB, even though CNB achieved a fewer number of biopsies than FNA. In our subgroup analysis (staff vs. non-staff), we detected no significant difference in any parameter in terms of efficacy or safety. Our present findings thus suggest that FNA and CNB are equivalent in terms of pain, tolerability, and possible complications.
Two previous studies have also compared the tolerability of FNA versus CNB [1516] and suggested that the two procedures are similar in terms of tolerability and pain. Nasrollah et al. [16] found that the occurrence of pain during the first few minutes following CNB was significantly higher than FNA, although there was no significant difference in pain detected at later time points for either procedure. The patients in that study were asked to evaluate the degree of tolerability for both procedures, and no significant difference was reported. Stangierski et al. [15] reported that CNB was slightly more painful than FNA but remained tolerable for most patients. In our present study, we found no differences in either pain or tolerability for either procedure during and at 20 minutes after the biopsy. We further found that CNB required fewer numbers of biopsies than FNA. Considering the different experience level of the staff versus non-staff who performed these procedures, we also performed subgroup analysis, but again found no significant differences in terms of pain, tolerability, or complications for both procedures.
In recent studies, CNB using 18 to 22 gauges cutting needles with US guidance has allowed operators to obtain thyroid tissue samples without major complications. From 1994 to 2014, many CNB studies have reported associated complications (Table 4) [7891112162122232425262728293031323334353637383940,41]. The total number of nodules reported in that period was 4,201, for which there were 60 recorded complications (1.43%) including hematoma, parenchyma edema, hemoptysis, infection, hoarseness, vasovagal reaction, and dysphagia. Hematoma was the most common complication, which was reported in 35/4,201 cases (0.83%). There were three patients described with minor soft tissue infections after CNB that were successfully managed by orally administered antibiotics. In our present study, only three of the cases in the CNB series (3%) experienced minor hematoma after the procedure and there was no significant difference in the complication rate between the FNA and CNB groups. All of the reported symptoms among our current study subjects were mild and recovery occurred in all cases with simple compression within 2 hours. Furthermore, additional treatments or hospitalization were unnecessary in these cases. Although CNB showed no major complications and similar pain or discomfort when compared with FNA in our study, it remains important for investigators to understand the anatomy of the thyroid and perithyroidal areas to minimize adverse events after either procedure [19].
In terms of the advantages of CNB, it provides a larger tissue sample and can facilitate a more precise histological diagnosis. At the microscopic level, the core of the CNB nodule sample offers the possibility of evaluating the general architecture of the lesion, alterations of the follicular structures, and the integrity of the capsule of the nodule along with its relationship with adjacent tissues. Specifically, CNB core samples allow for the differentiation of cancer in up to 98% of thyroid nodules that showed previously indeterminate FNA results [8]. Additionally, Na et al. [7] reported that CNB is more useful for reducing the number of inconclusive diagnoses than FNA, and that this method will serve a useful complementary diagnostic role for the optimal management of thyroid nodules with previous non-diagnostic findings or atypia from FNA readings. The devices and techniques associated CNB have been developed over the past 20 years. Unlike the old technique of palpation-guided large needle biopsy, US-guided CNB, using a modern spring-activated biopsy needle, has been reported to be a safe and well tolerated procedure [162122]. Furthermore, the guidelines of the National Cancer Institute [14] also suggest that CNB is safe, well tolerated, and associated with a low incidence of complications.
Our study had several limitations of note. First, it was retrospective in design, which could have introduced an inherent bias regarding patient selection. However, all patients were consecutively enrolled. Second, multiple operators with different levels of experience were involved. However, the inclusion of multiple operators reflects the real practice in our hospital and we performed subgroup analysis between staff and non-staff. Third, we used relatively small sample size to assess the low incidence of complication of FNA and CNB. Although CNB has been known as a safe procedure [424344], a further prospective investigation with a larger sample size is necessary to verify the results of our study. Finally, we performed local anesthesia with 2% lidocaine when performing the CNB procedure, but not with FNA procedure. It can be influence the pain score of the patients who underwent CNB procedure. However, 70% of patients, who underwent CNB by staff, did not undergo local anesthesia. Thus majority of patients who underwent CNB actually did not have received local anesthesia.
In conclusion, FNA and CNB show no significant differences in terms of pain, tolerability, or complications for diagnosing thyroid nodules.
Notes
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.
AUTHOR CONTRIBUTIONS: Conception or design: J.H.B., E.J.J., Y.J.C., J.H.L., J.K.K., S.R.C. Acquisition, analysis, or interpretation of data: J.H.B., E.J.J., S.R.C. Drafting the work or revising: J.H.B., E.J.J., S.R.C. Final approval of the manuscript: J.H.B., E.J.J., Y.J.C., J.H.L., J.K.K., S.R.C. Statistical analysis: J.K.K.