Site-Specific Difference of Bone Geometry Indices in Hypoparathyroid Patients
Article information
Abstract
Background
Hypoparathyroid patients often have a higher bone mineral density (BMD) than the general population. However, an increase in BMD does not necessarily correlate with a solid bone microstructure. This study aimed to evaluate the bone microstructure of hypoparathyroid patients by using hip structure analysis (HSA).
Methods
Ninety-five hypoparathyroid patients >20 years old were enrolled and 31 of them had eligible data for analyzing bone geometry parameters using HSA. And among the control data, we extracted sex-, age-, and body mass index-matched three control subjects to each patient. The BMD data were reviewed retrospectively and the bone geometry parameters of the patients were analyzed by HSA.
Results
The mean Z-scores of hypoparathyroid patients at the lumbar spine, femoral neck, and total hip were above zero (0.63±1.17, 0.48±1.13, and 0.62±1.10, respectively). The differences in bone geometric parameters were site specific. At the femoral neck and intertrochanter, the cross-sectional area (CSA) and cortical thickness (C.th) were higher, whereas the buckling ratio (BR) was lower than in controls. However, those trends were opposite at the femoral shaft; that is, the CSA and C.th were low and the BR was high.
Conclusion
Our study shows the site-specific effects of hypoparathyroidism on the bone. Differences in bone components, marrow composition, or modeling based bone formation may explain these findings. However, further studies are warranted to investigate the mechanism, and its relation to fracture risk.
INTRODUCTION
Hypoparathyroidism, the deficiency or absence of parathyroid hormone (PTH), is a unique condition; it is managed with calcium, active vitamin D, or dichlozid supplements [1], whereas most other conditions of hormone deficiencies are treated by replacing the deficient hormone itself.
PTH is an important hormone for regulating calcium and phosphate homeostasis, and also plays a key role in bone remodeling. It increases bone resorption by stimulating the receptor activator for nuclear factor κ B ligand (RANKL) expression of osteoblasts, osteocytes, and T cells [23], and when given intermittently with at a low dose, has been reported to have anabolic effects on the bone [4]. The exact mechanism of this anabolic effect is uncertain; however, presumably, it is induced by increasing insulin-like growth factor 1 and suppressing apoptosis and sclerostin (SOST) expression [4].
The effects of a PTH excess or deficiency on the bone mineral density (BMD) have been described in previous studies. In case of hyperparathyroidism, the BMD is usually preserved at the lumbar spine where the trabecular bone is dominant. However, at the femur, where the cortical bone is dominant, the BMD is decreased and tunneling effects are noted, which lead to a high risk of fracture [5]. In contrast, patients with permanent hypoparathyroidism were reported to have higher BMD than do age-and sex-matched controls [6]. Takamura et al. [7] reported higher BMDs in postmenopausal women with postoperative transient hypoparathyroidism, and they attributed this finding to attenuated bone turnover. However, as a high BMD does not necessarily reflect high bone quality, the risk of fracture in patients with hypoparathyroidism is controversial [8910]. Furthermore, studies on the quantitative and qualitative change of bone in hypoparathyroid patients and patients receiving treatment with PTH are lacking.
Recently, Cusano et al. [11] reported that cortical volumetric BMD was increased and cortical porosity was reduced in hypoparathyroid patients at the radius and tibia. However, despite the increased BMD and decreased cortical porosity, the ultimate stress and failure load were not changed compared with those in controls. Noteworthy, the authors reported a trend toward a decrease in cortical thickness (C.th) at the radius and the tibia even though the etiology for the decreased C.th in their hypoparathyroid cohort was unclear. Therefore, in this study, we measured bone geometric parameters by using hip structure analysis (HSA). Here, we find that there is a site-specific difference of bone geometry indices in patients with hypoparathyroidism.
METHODS
Subjects
Hypoparathyroid patients >20 years old were enrolled. We defined hypoparathyroidism as a state of low PTH level below the lower limits of normal (15 pg/mL) on at least two prior occasions, and the use of calcium or vitamin D treatment to maintain a normal calcium level. Patients who had been receiving treatment with bisphosphonates, estrogens, progestins, calcitonin, diuretics, statins, or methotrexate, which are agents that could affect bone metabolism, were excluded. Patients were also excluded if they had a diagnosis of diabetes, chronic kidney disease, Cushing's syndrome, or other malignancies. Ninety-five patients with hypoparathyroidism were enrolled, and 31 females of them had eligible dual-energy X-ray absorptiometry (DXA) data for HSA.
The Severance Hospital Institutional Review Board approved the study protocol, and no consent was given because the data were collected and analyzed anonymously.
Control subjects
We collected the control data from a previous study by Kim et al. [12]. The study data comprise measurements of bone geometry of 3,580 Korean people who participated in the Fourth Korean National Health and Nutrition Examination Survey. To compare the bone geometry of these controls with those of the patients, normal subjects aged from 20 to 80 years were selected. From the collected data, we extracted three sex-, age-, and body mass index (BMI)-matched control subjects to each patient.
Biochemical measurement
The serum calcium, phosphate, and intact PTH levels, which were measured at the time of BMD measurement, were reviewed. The serum concentration of intact PTH was measured by immunoradiometric assay (Nichols Institute Diagnostics, Sacramento, CA, USA). The reference rage of serum intact PTH was 15 to 65 pg/mL.
Imaging evaluation
The BMD data were reviewed retrospectively. The BMD of hypoparathyroid patients had been measured at the lumbar spine, femoral neck, and total hip, and by using the same densitometer at the same center (DXA, Hologic Discovery A, Waltham, MA, USA). The Z-score was used for analysis because the subjects in this study had a wide range of age. Mean values and standard deviations (SD) for each decade from the Korean reference data were used to calculate Z-score as previously reported [13]. The bone geometry parameters of the patients and were analyzed with an HSA program included in APEX software of Hologic, as previously described [121415]. For control subjects, their BMD and bone geometry were analyzed by using DXA scan (QDR 4500 A, Hologic, Bedford, MA, USA) and HSA program, respectively, and we obtained their data from the study by Kim et al. [12]. By using HSA, the geometric properties of the cross section can be analyzed at specific locations of the femur [16]. Three sites at the proximal femur were evaluated: narrow neck, intertrochanter, and shaft. The narrow neck indicates the narrowest point of the femoral neck; the intertrochanter region is the bisector of the neck-shaft angle; and the femoral shaft is located 2 cm distal from the midpoint of the lesser trochanter [1617]. The narrow neck and shaft region were modeled as circular annuli with a cortical bone content of 60% and 100%, respectively. In contrast, the intertrochanter region was modeled as an elliptical annulus with a cortical and trabecular bone content of 70% and 30%, respectively [1517]. At each location, the cross-sectional area (CSA), cross-sectional moment of inertia (CSMI), C.th, section modulus (SM), and buckling ratio (BR) were measured. The CSMI indicates structural rigidity and the distribution of mass about the center of a structural element. SM is an index of the bending strength for maximum bending stress, and BR means relative thickness of the cortex reflecting cortical stability in buckling [16]. Those bone geometry indices of the patient group were compared with those of the age-, sex-, and BMI-matched control subjects.
Statistical analysis
All statistical analyses were performed with the SPSS version 20.0 (IBM Co., Armonk, NY, USA) and GraphPad Prism 5 (GraphPad Software Inc., San Diego, CA, USA). Values are expressed as mean±SD or medians with interquartile range. Descriptive statistics were used to show the Z-scores of the BMD of hypoparathyroid patients. Between-group differences of means of baseline variables and Z-scores were tested by unpaired t tests. For geometric parameters, because of the small number of the subjects in each group, nonparametric Mann-Whitney U tests were used. For all statistical analysis, P<0.05 was considered statistically significant.
RESULTS
The baseline characteristics are shown in Table 1. The mean age of the hypoparathyroid patients was 53.89±13.51 years, and most of them were women (78.9%). Their calcium, PTH, and 25-OH-vitamin D levels were below normal. Of those 95 patients with hypoparathyroidism, 74 patients had postsurgical hypoparathyroidism, and the others had idiopathic hypoparathyroidism such as immune-mediated parathyroid destruction or genetic disorders. The baseline characteristics were similar between the two groups except for disease duration, calcium level, and β-cross laps. The mean duration of the disease was significantly longer in patients with postsurgical hypoparathyroidism and calcium and β-cross laps were significantly lower in the idiopathic group.
The mean Z-scores of hypoparathyroid patients were above zero at all sites where BMD was measured—lumbar spine, femoral neck, and total hip (0.63±1.17, 0.48±1.13, and 0.62±1.10, respectively) (Fig. 1A). The mean value of BMD was slightly higher at the lumbar spine than at the femoral neck or total hip; however, no statistical significance was found. We also compared the BMD according to the etiology of the disease, and there was no difference between the two groups (Fig. 1B).
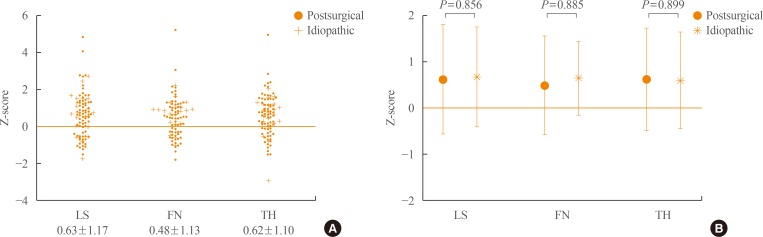
Z-scores of the hypoparathyroid patients. (A) Distribution of the Z-scores of hypoparathyroid patients. (B) Z-scores according to disease etiology. Symbols represent mean±SD. LS, lumbar spine; FN, femoral neck; TH, total hip.
The difference in bone geometry between the patient group and the control group was analyzed at three sites (femoral neck, intertrochanter, and femoral shaft) for five indexes (CSA, CSMI, SM, C.th, and BR). Thirty-one females had eligible data for analyzing bone geometry. The number of patients in each age group was four (age 30 to 39 years), six (age 40 to 49 years), eight (age 50 to 59 years), eight (age 60 to 69 years), and five (age 70 to 79 years) (Table 2). We matched three control female subjects to each patient, and the number of controls for each age group was 12, 18, 24, 24, and 15, respectively. The mean age of control subjects was 55.45±12.50 years and their mean BMI was 23.11±3.11 kg/m2. In HSA, although the statistical significance varied within the age groups, we found a similar trend of indices in the patient group (Fig. 2). At the femoral neck and intertrochanter, the CSA and C.th were higher, whereas the BR was lower than those of the control subjects. Of interest, those findings for the CSA, C.th, and BR were inversely noted at the femoral shaft. At the femoral shaft, the CSA and C.th were low, whereas BR was high. Although the trend of the CSA was not statistically significant, the C.th and BR had a significant trend in the 40 to 49 and 70 to 79 years age groups. In patients with idiopathic hypoparathyroidism, the same trend was noted (Fig. 2, cross). In addition, the CSMI and SM were higher at the intertrochanter in hypoparathyroid patients group; however, this trend was not found at the femoral neck (Table 2). Of note, at femoral neck, CSMI was higher in age group of 30 to 39 and 70 to 79, whereas lower in age group of 40 to 49, 50 to 59, and 60 to 69. However as it was only significant in age group of 50 to 59 and 60 to 69, we may need further data to make any conclusive explanation.
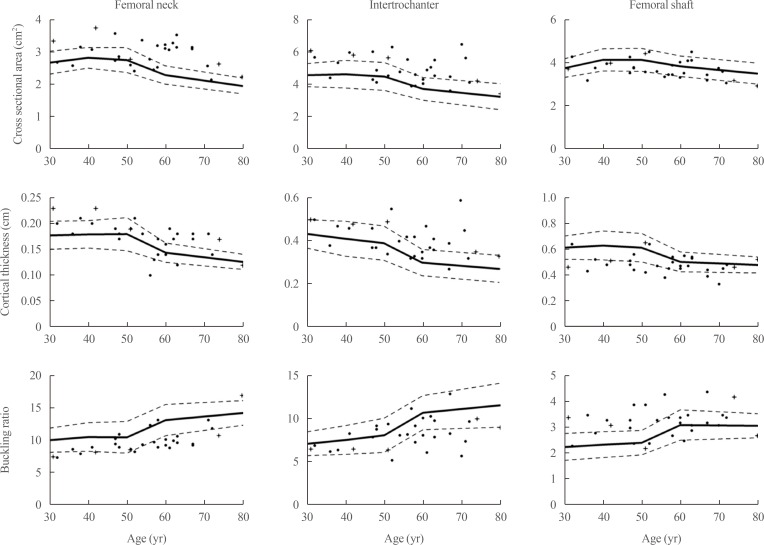
Bone geometric parameters of the control subjects and hypoparathyroid patients. The values of the sex-, age-, and body mass index-matched control subjects are depicted as lines shown as mean±SD; those of the hypoparathyroid patients are shown as circles (postsurgical hypoparathyroidism) or cross (idiopathic hypoparathyroidism).
DISCUSSION
In this cross-sectional study, we showed that bone geometric parameters differ depending on the site. It is well known that hypoparathyroid patients have a higher BMD than the general population; however, their bone geometric characteristics have not been well understood. This study provides a better understanding of the bone structure of hypoparathyroid patients.
PTH has a crucial role in regulating bone remodeling. It induces osteoclastogenesis by stimulating osteoblasts and subsequently, the bone matrix is resorbed and calcium is released [18]. In case of a low level of PTH, bone remodeling is suspended and, thereby, the bone mass is increased. This idea has been supported by previous studies. A study of 33 hypoparathyroid patients showed that the patients had a higher BMD at the lumbar spine and proximal femur than did the control group [19]; other studies also reported similar results [620]. However, whether increased BMD has a protective effect against fractures in hypoparathyroid patients, as it has in the general population, is uncertain. According to Underbjerg et al. [8] increased BMD in hypoparathyroidism has a protective effect against fractures at the upper extremities; however, some other studies could not find any protective role, and even revealed an increased risk of fracture [910]. These inconsistent results raised the question about the bone structure in hypoparathyroid patients. A previous study with iliac biopsy noted that the bone structure and dynamic properties are markedly atypical in hypoparathyroid patients [19]. However, these studies were limited by the small number of evaluated patients and their use of an invasive procedure that cannot be easily applicable in clinics. In this study, the BMD of 95 hypoparathyroid patients was reviewed, and the bone geometry of 31 patients was analyzed by using HSA.
On baseline characteristics, the mean duration of the disease differs between the postsurgical group and idiopathic group, which might be due to the period of nondetection of the disease. Postsurgical PTH deficiency is usually detected during the early stage of disease, because patients usually obtain regular checkups after the operation. On the other hand, patients with idiopathic hypoparathyroidism usually seek medical attention only after they develop symptoms of hypocalcemia; hence, the disease is usually recognized late. Therefore, the actual duration of disease in the idiopathic hypoparathyroidism group could have been longer than what has been stated. Calcium and β-cross laps were significantly lower in the idiopathic group, and again it could be explained by the longer nontreatment period before the diagnosis. Regarding lower β-cross laps, genetic mutation or autoimmune factor of idiopathic hypoparathyroidism could play a role in suppressing bone turnover. However, role of genetic or autoimmune factor regarding bone remodeling was not dealt in this study, which can be a limitation of this study.
The mean Z-scores in hypoparathyroid patients were above zero, which indicate higher BMDs than those in sex- and age-matched Korean population. In terms of the sites, the Z-score of the lumbar spine was slightly higher than that of the femoral neck or total hip; however, no significant difference was found. Furthermore, when the patients were grouped according to disease etiology, there was no difference in the Z-score between the groups, which is consistent with a previous study [6]. Of the 95 patients, the DXA data of 31 patients were used to analyze bone geometry by using the HSA program, because we only included female data and the HSA program was available only after 2011. The bone geometry of those 31 patients was compared with that of the control subjects. The structural indices including C.th, CSA, CSMI, SM, and BR were analyzed. Those parameters were previously demonstrated to be risk factors for fracture [21]. It was reported that subjects with a history of fracture had lower CSA, C.th, CSMI, and SM, and higher BR than subjects without a history of fracture. In this regard, if hypoparathyroidism increases the BMD with a lower risk of fracture similar to the general population, we should have seen higher CSA, C.th, CSMI, and SM, and lower BR in the patient group. Interestingly, the results were rather different depending on the measurement site. At the femoral neck and intertrochanter, the bone geometry parameters showed a similar trend, as expected; the C.th and CSA were high, whereas the BR was low. However, at the femoral shaft, the C.th and CSA were found to be lower, and the BR higher, than those in the control subjects.
The specific reason for the different trends at different sites is unclear. Previously, it was illustrated that increased BMD in hypoparathyroid patients differed depending on the site [6]. Although it was not statistically significant, increased BMD was more prominent at the lumbar spine than at the femoral neck. We assumed that this difference might be attributed to the difference in bone composition. In the same context, the site-specific difference of bone geometry parameters can also be due to inconsistent bone structure. The lumbar spine and femoral neck have considerable amount of trabecular bone, whereas the femoral shaft mostly consists of cortical bone [22]. In this study, at the femoral neck where the proportion of trabecular bone is estimated as 40% [23], the C.th in hypoparathyroid patients was higher; however, this finding was not observed at the femoral shaft, which is mostly composed of cortical bone. As mentioned above, the increased BMD in hypoparathyroid patients resulted from decreased bone turnover and remodeling; thereby, it affects mostly the trabecular bone. At the femoral neck where trabecular bone is abundant, the excavated cavities at the trabecular bone adjacent to the cortical bone receive infilling, which can lead to thicker cortical bone. In contrast, the femoral shaft mostly consists of cortical bone, which means less chance of being affected by decreased bone turnover. Hence, it is less likely to accrue C.th. We also assumed that different marrow compositions among locations could be a potential factor for different reactions to PTH deficiency. At birth, the red marrow occupies most of the skeletal bones except epiphyses and apophyses. During growth, the red marrow becomes confined to the axial skeleton, and the femoral neck is the only site in the femur where residual red marrow exists. The yellow marrow replaces the red marrow in the rest of the femur [24]. As it is mainly made of fat tissues, it is plausible that the bone part consisting of yellow marrow does not actively respond to bone modeling. Finally, less modeling-based bone formation (MBF) would induce the site-specific response to PTH deficiency. A well-known antiresorptive agent, denosumab, was reported to continuously increase BMD by maintaining MBF [25]. What was intriguing about this finding was that this effect was only confined to some part of the bone—the superior endocortex and inferior periosteum of the femoral neck. These two sites are where mechanical loading is increased. Because PTH deficiency also reduces bone turnover and remodeling like denosumab does, we propose that PTH also maintains MBF only at sites where loading is increased, like the femoral neck in this study. With the above-mentioned mechanisms, we could partly explain why the geometric indices at the femoral neck and intertrochanter changed as we expected. However, the changes at the femoral shaft are still unclear. A similar result was reported in a recent study [11] that analyzed the skeletal microstructure of the distal radius and tibia by using high-resolution peripheral quantitative computed tomography, and the authors found a trend toward a decline in C.th. An underlying disease and lower physical activity level of the patients were raised as factors for a decreased C.th; however, the exact mechanism is still uncertain.
This study has some limitations. Although we analyzed BMD in 95 hypoparathyroid patients, which, to our knowledge, is the largest number of study patients, only female patients (n=31) were evaluated for bone geometric parameters. Moreover, the study population might not be enough to represent all hypoparathyroid patients. Second, bone biopsy was not assessed. In the scan images, edge margins are often blurred, and there may be difficulties in differentiating cortical bone from trabecular bone [26]. Lastly, this was a retrospective study. However, a prospective study is difficult to perform because the exact onset of hypoparathyroidism is difficult to define.
In conclusion, we demonstrated that hypoparathyroidism affects the site of bone remodeling specifically. Further studies are needed to investigate the exact mechanism of the site-specific response of the bone to PTH deficiency.
ACKNOWLEDGMENTS
We thank Eun Ae Kim and Jaekyu Ko for their technical assistance.
Notes
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.




