Cardiovascular Effects of Glucagon-Like Peptide-1 Receptor Agonists
Article information
Abstract
Glucagon-like peptide-1 (GLP-1) is a member of the proglucagon incretin family, and GLP-1 receptor agonists (RAs) have been introduced as a new class of antidiabetic medications in the past decade. The benefits of GLP-1 RAs are derived from their pleiotropic effects, which include glucose-dependent insulin secretion, suppressed glucagon secretion, and reduced appetite. Moreover, GLP-1 RAs also exert beneficial roles on multiple organ systems in which the GLP-1 receptors exist, including the cardiovascular system. Cardiovascular effects of GLP-1 RAs have been of great interest since the burden from cardiovascular diseases (CVD) has been unbearably increasing in a diabetic population worldwide, despite strict glycemic control and advanced therapeutic techniques to treat CVD. Preclinical studies have already demonstrated the beneficial effects of GLP-1 on myocardium and vascular endothelium, and many clinical studies evaluating changes in surrogate markers of CVD have suggested potential benefits from the use of GLP-1 RAs. Data from numerous clinical trials primarily evaluating the antihyperglycemic effects of multiple GLP-1 RAs have also revealed that changes in most CVD risk markers reported as secondary outcomes have been in favor of GLP-1 RAs treatment. However, to date, there is only one randomized clinical trial of GLP-1 RAs (the ELIXA study) evaluating major cardiovascular events as their primary outcomes, and in this study, a neutral cardiovascular effect of lixisenatide was observed in high-risk diabetic subjects. Therefore, the results of ongoing CVD outcome trials with the use of GLP-1 RAs should be awaited to elucidate the translation of benefits previously seen in CVD risk marker studies into large clinical trials with primary cardiovascular outcomes.
INTRODUCTION
Because CVD is the leading cause of death in patients with type 2 diabetes (T2D), T2D is often referred as a 'cardiovascular disease (CVD)' [1]. Despite a significant improvement in the treatment of T2D and CVD over the past few decades, reducing CVD in patients with T2D solely with an optimal glycemic control has been challenging [2]. Therefore, current treatment guidelines recommend maintenance of an optimal weight and stringent management of lipid levels in addition to glycemic control [3]. Clearly, there has been a need for a new class of antidiabetic agents with advantages over currently marketed drugs that can lower glucose via mechanisms resulting in either neutral or protective effects on the cardiovascular system.
In the last 10 to 15 years, discoveries on certain gut peptides that influence insulin secretion related to meal ingestion, namely incretins, have opened up a new paradigm in the treatment of diabetes [4]. The levels of incretins including glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic hormone rise in response to a meal, and they are rapidly degraded by the enzyme dipeptidyl peptidase 4 (DPP-4) [5]. Accordingly, incretin-based therapies, such as DPP-4 inhibitors and GLP-1 receptor agonists (GLP-1 RAs) that are resistant to degradation by DPP-4, have been introduced in the market within the past decade to overcome and extend the short half-life of the incretin effect.
GLP-1 RAs exert an antihyperglycemic effect by enhancing pancreatic β-cell–mediated glucose-dependent insulin secretion, suppressing glucagon secretion, delaying gastric emptying, and reducing food intake in patients with T2D [6]. The key beneficial features of GLP-1 RAs are weight loss and a relatively low risk of hypoglycemia compared with other antidiabetic agents [7]. Moreover, additional roles for GLP-1 have been revealed since the discovery of an extrapancreatic distribution of GLP-1 receptors in multiple organs such as the gastrointestinal tract, lung, heart, liver, and kidneys [58910], and evidence shows that administration of GLP-1 or GLP-1 RAs result in beneficial effects in these organs, indeed [11].
Of particular interest, the discovery of GLP-1 receptor expression in the myocardium and in the vascular endothelium suggested possible roles of GLP-1 in the cardiovascular system and multiple preclinical studies support cardioprotective effects of native GLP-1 (Fig. 1) [1213141516171819202122232425]. For example, Gros et al. [16] found that the lack of GLP-1 receptors resulted in impaired left ventricular (LV) contractility and a decreased resting heart rate (HR) in GLP-1 receptor knockout mouse models. Also, Nikolaidis et al. [18] showed that intravenous infusion of GLP-1 for 48 hours in animal models with induced dilated cardiomyopathy significantly improved LV contractility, stroke volume, and cardiac output. Furthermore, many studies of ischemia/reperfusion animal models consistently revealed beneficial effects of GLP-1 on post-ischemia recovery and myocardial viability [13172224]. Regarding the effect of GLP-1 on vascular endothelium, numerous in vitro studies and animal studies showed either nitric oxide-dependent or -independent vasodilative effects [121415192021]. Lastly, in hypertension-prone animal models, GLP-1 infusion exerted its diuretic and natriuretic effects and resulted in a reduction of hypertension and albuminuria development, and an improved endothelial function [23]. These beneficial preclinical roles of GLP-1 in the cardiovascular system might translate to the clinical setting. Therefore, attempts have been made to elucidate the cardioprotective effects of GLP-1 RAs in humans.
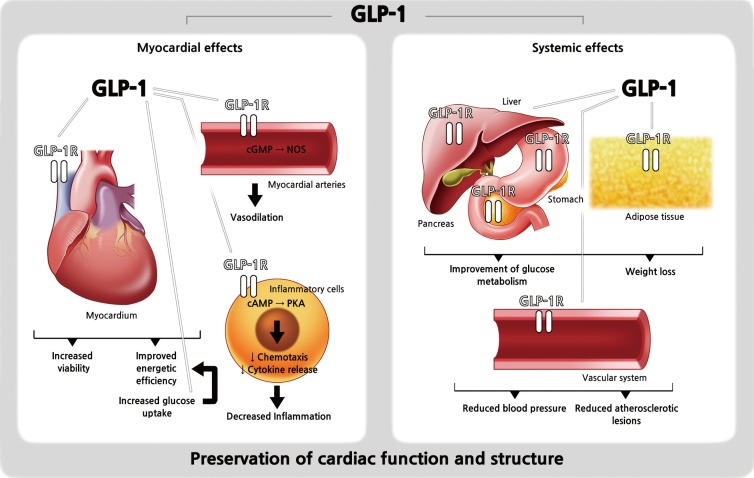
Schematic representation of the distribution of glucagon-like peptide-1 (GLP-1) receptors (GLP-1Rs) and the cardiovascular and systemic effects of GLP-1. Modified from Ravassa et al. [25], with permission from Oxford University Press. cAMP, cyclicadenosine monophosphate; cGMP, cyclic guanosine monophosphate; NOS, nitric oxide synthase; PKA, protein kinase A.
Numerous GLP-1RAs are currently available on the market, and each agent possesses a unique biochemical profile, depending the alterations made to its backbone, human GLP-1, or exendin-4 (Fig. 2) [26]. Exenatide and liraglutide were released onto the market relatively early, whereas newer agents, such as lixisenatide, dulaglutide, and albiglutide, were approved more recently (Table 1). Accordingly, large clinical trials on the safety and efficacy of these earlier GLP-1 RAs are available. However, concerns have recently emerged about the long-term cardiovascular safety of antidiabetic medications, and U.S. Food and Drug Administration published guidelines to assess the cardiovascular risk of novel antidiabetic drugs [27]. Therefore, the role of GLP-1 RAs specifically in the cardiovascular system has been of particular interest in the past several years, and multiple clinical trials have examined the cardiovascular effects and safety of GLP-1 RAs (Table 1). Most trials are still ongoing and only one prospective randomized clinical trial with a primary outcome of major cardiovascular events in patients with T2D has reported its results [28]. Therefore, this present review examines the cardiovascular effects of the GLP-1 RAs in T2D by revisiting the results of clinical studies evaluating CVD surrogate markers and cardiovascular events.
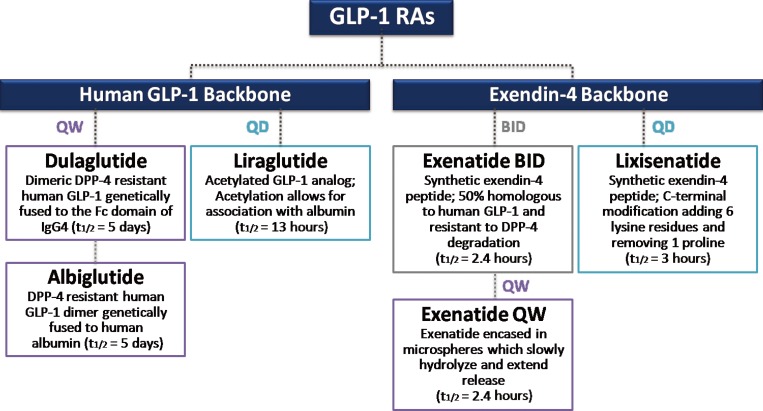
Classification of currently available glucagon-like peptide-1 receptor agonists (GLP-1 RAs) by structure and duration of action. Adapted from Kuritzky et al. [26], with permission from Taylor & Francis. QW, weekly; QD, daily; BID, twice a day; DPP-4, dipeptidyl peptidase 4; IgG4, immunoglobulin G4.
CARDIOVASCULAR EFFECTS OF GLP-1 RAs IN HUMAN STUDIES
Studies using surrogate markers of CVD risks
Blood pressure and heart rate
Hypertension affects almost three-quarters of T2D patients [29] and is a powerful independent predictor of CVD mortality [30]. Accordingly, better control of cardiovascular risk factors such as blood pressure (BP) in patients with T2D is required to achieve favorable cardiovascular safety goals. Indeed, GLP-1 RAs have consistently showed BP-lowering effects in human subjects, although the exact mechanisms are not fully understood [31323334]. For example, Wang et al. [34] showed that treatment with the GLP-1 RAs exenatide and liraglutide reduced both systolic and diastolic BP by 1 to 5 mm Hg in patients with T2D, compared with some other anti-diabetic drugs, including insulin, glimepiride, and placebo.
For most GLP-1 trials, HR is included as a secondary safety endpoint, and the results have been inconsistent. Nonetheless, the effect of GLP-1 RAs on HR should not be overlooked, because studies have shown that an elevated HR was potentially associated with a higher cardiovascular risk [353637]. Although the subjects were non-diabetic, one meaningful study observed acute changes of cardiovascular risk markers such as HR and BP upon intravenous administration of GLP-1 RA [38]. In this study, Smits et al. [38] performed automated oscillometric BP measurements and a finger photoplethysmography during the intravenous administration of placebo (saline 0.9%), exenatide, or a combination of exenatide and the nitric oxide-synthase inhibitor (L-NMMA) in 10 healthy overweight males. As a result, exenatide increased HR by a mean maximum of 6.8 beats per minute (bpm), as well as systolic BP by 9.8 mm Hg [38]. Simultaneously, sympathetic nervous system activity measured by HR variability and the rate-pressure-product were also elevated, and increases in HR, systolic BP and sympathetic nervous system activity were still observed even with a concomitant L-NMMA-infusion [38]. On the other hand, Mendis et al. [39] recruited healthy male subjects and found that HR was elevated by 8.2 in the exenatide group compared with the placebo group. The exenatide group in this study, however, showed a significant increase in the mean cardiac output by 1.2 L/min, a reduced total peripheral resistance by the mean of 120 dyn·s/cm–5, and a significantly reduced urinary sodium: creatinine ratio without a significant change in the BP [39]. Therefore, the authors postulated that the GLP-1-induced vasodilation might reduce total peripheral resistance, leading to a reflex tachycardia [39]. They also suggested that the natriuretic effect of GLP-1 RAs they have seen in their study might be one of the mechanisms behind the BP-lowering effect of GLP-1 RAs, although the BP reduction was not significant in their study [39]. Nonetheless, the exact mechanisms underlying the GLP-1 RA-associated HR acceleration are still unclear and these preliminary study results involving healthy male subjects cannot be over-generalized to a diabetic population using GLP-1 RAs. However, the results clearly suggest the need for further investigation of the effect of GLP-1 RAs on HR and BP and their long-term consequences.
Table 2 summarizes the cardiovascular risk-relevant outcomes of large clinical trials such as the Diabetes therapy Utilization: Researching changes in A1c, weight and other factors Through Intervention with exenatide Once weekly (DURATION) trials of exenatide once-weekly, the Liraglutide Effect and Action in Diabetes (LEAD) studies of liraglutide, the Assessment of Weekly Administration of LY2189265 in Diabetes-1 (AWARD) studies of dulaglutide, the HARMONY studies of albiglutide, and the GetGoal studies of lixisenatide. Most clinical trials on the efficacy of GLP-1 RAs found a reduced systolic BP, and this beneficial effect was somewhat more apparent in studies of liraglutide and exenatide (Table 2). Although data on HR were not provided in most studies of twice-daily exenatide, HR elevation was generally more apparent in the GLP-1 RA treatment arms, compared with the comparator arms (Table 2). Although these changes in HR upon GLP-1 RA administration were subtle and the underlying mechanisms are unclear, further studies are necessary to elucidate the long-term effects of GLP-1 RAs primarily on HR and other related variables to clarify whether the elevated HR can be regarded as a transient reflex mechanism or whether it might in fact lead to poor cardiovascular outcomes in a diabetic population.
Circulating biomarkers and lipid profiles
Several studies have primarily evaluated the changes in cardiovascular risk biomarkers upon the use of GLP-1 RAs. Courreges et al. [40] evaluated the effects of liraglutide on cardiovascular risk biomarkers such as adiponectin, leptin, high-sensitivity C-reactive protein (hs-CRP), interleukin 6 (IL-6), tumor necrosis factor α (TNF-α), plasminogen activator inhibitor 1 (PAI-1), and B-type natriuretic peptide (BNP). Of these parameters, PAI-1 and BNP levels were significantly decreased following a 14-week treatment with liraglutide (–29% for PAI-1 with 1.25 mg liraglutide and –38% for hs-CRP with 1.90 mg liraglutide) versus placebo [40]. Because the level of PAI-1 is correlated with atherosclerosis formation [41] and is markedly elevated in T2D patients with CVD compared with healthy or T2D patients without CVD [42], this result implies more fibrinolytic thereby less atherogenic processes. In this study, a dose-dependent reduction of hs-CRP levels was seen, albeit nonsignificant [40]. Fourteen weeks of liraglutide treatment in 59 overweight or obese T2D patients whose glycemic control was inadequate (hemoglobin A1c >7% with metformin and/or sulfonylurea) at the initiation period resulted in a nonsignificant decrease in pro-inflammatory factors such as TNF-α and IL-6 [43]. These decreased pro-inflammatory markers suggest liraglutide might attenuate inflammatory cascade which leads to the development of atherosclerosis [44].
In addition, several cardiovascular marker studies were conducted also with the exenatide treatment [4546]. Gurkan et al. [45] enrolled 34 insulin and incretin-naïve T2D patients on metformin and randomized subjects to exenatide or insulin glargine treatment arms. After 26 weeks, the levels of hs-CRP and endothelin-1 decreased (27.5% and 18.75%, respectively) in the exenatide arm whereas the levels of fibrinogen, monocyte chemoattractant protein-1, leptin and endothelin-1 increased in the insulin glargine arm (13.4%, 30.2%, 47.5%, and 80%, respectively) [45]. Blonde et al. [47] observed that 82 weeks of adjunctive exenatide administration in 314 T2D patients with sulfonylurea and/or metformin resulted in reduced triglyceride concentrations (–0.43 mmol/L) and elevated high density lipoprotein cholesterol concentrations (0.12 mmol/L). Other parameters such as total cholesterol, low density lipoprotein cholesterol, and apolipoprotein B showed trends toward improvement, albeit nonsignificant [47].
Likewise, multiple large clinical studies with various GLP-1 RAs have shown a more favorable trend in lipid profiles with GLP-1 RAs, although these were not evaluated as the primary outcomes (Table 2). Moreover, some trials revealed improvements in cardiovascular risk markers such as BNP and CRP. For instance, Bunck et al. [48] and Derosa et al. [49] observed significant reduction in hs-CRP with twice-daily exenatide, and DURATION-2, DURATION-3, DURATION-3 extension, and DURATION-5 studies observed considerable reductions in inflammatory markers such as hs-CRP or CRP (Table 2) [484950515253545556575859606162636465666768697071727374757677787980818283848586]. Notably, the 65% reduction in hs-CRP levels observed in one study was statistically significant, independent of body fat mass and body weight [48]. These results imply that the anti-inflammatory and potentially anti-atherosclerotic effects of GLP-1 RAs are not attributable to improved adiposity, which is often associated with a low-grade inflammation [87].
Carotid intima-media thickness
Carotid artery wall thickening is one of the earliest detectable manifestations of CVD, and is significantly correlated with future CVD events [88]. Intima-media thickness (IMT) measured at a common carotid artery level is widely used as an effective, non-invasive surrogate measure of atherosclerosis [89]. A few longitudinal studies evaluated the anti-atherosclerotic effects of GLP-1 RAs using carotid IMT data [9091]. Hopkins et al. [90] enrolled 11 severely obese T2D patients, treated them with either exenatide twice-daily (nine patients) or liraglutide (two patients) for 6 months, and compared the carotid IMT before and after the GLP-1 RA treatment. However, there was no change in carotid IMT with GLP-1 RA treatment despite an improved glycemic profile and a significant adipose tissue reduction [90]. This result might be attributable to the small number of patients and the long duration and severity of T2D, as well as the presence of comorbidities. In contrast, an 8-month prospective pilot study in which 64 T2D patients with no prior CVD history were treated with liraglutide revealed that the carotid IMT decreased from 1.19±0.47 to 0.94±0.21 mm (P<0.01) [91]. This study was the first to show a beneficial effect of GLP-1 RAs on carotid IMT in human subjects. Although there are many ongoing large CVD outcome trials of GLP-1 RA treatment (Table 1), more simultaneous investigation of the causal relationship between GLP-1 RA administration and useful surrogate measures of CVD, including carotid IMT, would help us understand the potential mechanisms through which GLP-1 RAs may result in favorable cardiovascular outcomes in patients with T2D.
LV function and reperfusion injury in patients with coronary artery disease
In 14 patients with coronary artery disease and good LV function, intravenous GLP-1 infusion improved global and regional wall LV function on dobutamine stress echocardiography compared with control [92]. Similarly, Lonborg et al. [9394] found that intravenous infusion of GLP-RA exenatide prior to revascularization in patients with ST-segment elevation myocardial infarction (MI) reduced reperfusion injury and final infarct size, compared with placebo treatment and Woo et al. [95] obtained similar results with subcutaneous injection of exenatide.
With subcutaneous liraglutide treatment, Chen et al. [96] observed a mild improvement in LV ejection fraction in ST-segment elevation MI patients who were not necessarily diabetic. On the other hand, in a retrospective study with acute MI patients who were diabetic, Nozue et al. [97] observed that liraglutide use was an independent negative predictor of LV mass index, suggesting liraglutide might prevent the progression of post-MI LV remodeling. Taken together, the results of numerous small studies equivocally suggest that GLP-1 RA treatment in coronary artery disease patients would be beneficial, regardless of the presence of diabetes. However, more large-scale clinical trial data are needed for a clear causal relationship to be established.
Cardiovascular outcome studies
Retrospective studies on cardiovascular outcomes
Although multiple clinical trials evaluating cardiovascular events associated with GLP-1 RA use as the primary outcome are currently ongoing (Table 1), abundant non-primary outcome data are available from many clinical trials on the efficacy of GLP-1 RAs marketed earlier, such as exenatide (DURATION studies) and liraglutide (LEAD studies). In addition, some studies on recently marketed GLP-1 RAs such as dulaglutide (AWARD studies), albiglutide (HARMONY studies), and lixisenatide (GetGoal studies) included cardiovascular parameters as a part of their non-primary endpoints (Table 2). Furthermore, we could get some information on the effect of GLP-1 RAs on cardiovascular outcomes through a few retrospective studies published in the literature [289899100101]. Best et al. [98], for instance, led a retrospective study of 39,275 patients with T2D on exenatide twice-daily and 381,218 patients on other glucose-lowering therapies. In this study, twice-daily exenatide treatment was associated with a lower risk of CVD events including MI, ischemic stroke, or coronary revascularization procedure (hazard ratio [HR], 0.81; 95% confidence interval [CI], 0.68 to 0.95; P=0.01) and hospitalizations (HR, 0.88; 95% CI, 0.79 to 0.98; P=0.02) than treatment with other glucose-lowering therapies [98]. It is worth noting that this cardioprotective effect of exenatide outweighed the fact that a greater proportion of patients on exenatide treatment possessed CVD risk factors such as prior ischemic heart disease, obesity, hyperlipidemia, and hypertension [98]. Monami et al. [100] reported a meta-analysis of 33 published and unpublished trials that evaluated the effect of GLP-1 RAs and had information on major adverse cardiovascular events (MACE), mortality, and cardiovascular risk factors. Although the difference in the incidence of MACE between GLP-1 RA and comparators failed to show statistical significance (Mantel-Haenzel odds ratio, 0.78; 95% CI, 0.54 to 1.13; P=0.18), GLP-1 RA were associated with a significant reduction in the incidence of MACE compared with placebo and pioglitazone, with a non-significant trend towards reduction in DPP-4 inhibitor-controlled studies [100]. There was no significant effect of GLP-1 RAs on mortality, although a nonsignificant favorable trend was observed in comparisons with placebo [100]. A retrospective longitudinal analysis of ambulatory care data with a median follow-up of 3.5 years showed that twice-daily exenatide use with or without insulin was associated with significantly fewer cardiovascular events than insulin treatment [101]. In addition, a meta-analysis of the HARMONY studies of albiglutide revealed that the risk of cardiovascular events was not significantly higher in subjects on albiglutide than in subjects on all other comparators including placebo and oral antidiabetic drugs such as glimepiride, insulin glargine, insulin lispro, liraglutide, pioglitazone, and sitagliptin [99].
A prospective randomized clinical trial with primary cardiovascular outcome: the ELIXA study
Of the numerous prospective randomized clinical trials evaluating the influence of GLP-1 RA use on cardiovascular outcomes (Table 1), only the ELIXA study of lixisenatide [28] has thus far published its results. In the ELIXA study, 6,068 patients with T2D who had an MI or who had been hospitalized for unstable angina within the previous 180 days were randomized to receive lixisenatide or placebo in addition to baseline treatment. In this study, with a median follow-up of 25 months, the primary composite end point including cardiovascular death, MI, stroke, and hospitalization for unstable angina occurred in 406 patients (13.4%) in the lixisenatide group and in 399 (13.2%) in the placebo group (HR, 1.02; 95% CI, 0.89 to 1.17) [28]. In addition, the results of the ELIXA study answered the question of cardiovascular safety related to heart failure by displaying no significant between-group differences in the rate of hospitalization for heart failure (HR, 0.96; 95% CI, 0.75 to 1.23) or the rate of death (HR, 0.94; 95% CI, 0.78 to 1.13) in the lixisenatide group [28].
Although the analysis revealed the non-inferiority of lixisenatide to placebo (P<0.001), it did not display superiority (P=0.81) [28]. The lack of superiority is probably attributable to the short follow-up period and high CVD risk in the enrolled subjects. However, there was no clear explanation for the neutral cardiovascular profile of lixisenatide, rather than the expected cardioprotective effect. Upcoming CVD outcome trials, such as the EXSCEL study of exenatide weekly, the LEADER study of liraglutide and the REWIND study of dulaglutide might provide further insight in this matter (Table 1).
CONCLUSIONS
Preclinical and preliminary clinical evidence has consistently revealed potential cardiovascular benefits from the administration of GLP-1 RAs. Likewise, the results of large clinical trials employing cardiovascular surrogate markers as their secondary outcomes also imply the potential beneficial effects of GLP-1 RAs in the cardiovascular system in human subjects. Although the ELIXA study—the only CVD outcome trial of GLP-1 RAs published to date—reported a neutral cardiovascular effect of lixisenatide, the potential cardiovascular benefits of GLP-1 RAs should not be overlooked, because high-risk subjects with established CVD were recruited in this study to delineate its cardiovascular safety within a short-term period. As depicted in Table 1, multiple large-scale clinical trials are ongoing to clarify the cardiovascular efficacy and safety of GLP-1 RAs and the results have been awaited with bated breath. The accumulation of the results of these ongoing cardiovascular outcome trials should be able to answer questions about cardiovascular benefits of GLP-1 RAs that were strongly suggested by previous preclinical and clinical studies and their possibility to lower the excessive cardiovascular burden in patients with T2D.
Notes
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.



