Articles
- Page Path
- HOME > Endocrinol Metab > Volume 30(1); 2015 > Article
-
Original ArticleDetermination of Mother Centriole Maturation in CPAP-Depleted Cells Using the Ninein Antibody
- Miseon Lee, Kunsoo Rhee
-
Endocrinology and Metabolism 2015;30(1):53-57.
DOI: https://doi.org/10.3803/EnM.2015.30.1.53
Published online: March 27, 2015
Department of Biological Sciences, Seoul National University, Seoul, Korea.
- Corresponding author: Kunsoo Rhee. Department of Biological Sciences, Seoul National University, 1 Gwanak-ro, Gwanak-gu, Seoul 151-747, Korea. Tel: +82-2-880-5751, Fax: +82-2-873-5751, rheek@snu.ac.kr
• Received: May 12, 2014 • Revised: June 2, 2014 • Accepted: June 3, 2014
Copyright © 2015 Korean Endocrine Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
-
Background
- Mutations in centrosomal protein genes have been identified in a number of genetic diseases in brain development, including microcephaly. Centrosomal P4.1-associated protein (CPAP) is one of the causal genes implicated in primary microcephaly. We previously proposed that CPAP is essential for mother centriole maturation during mitosis.
-
Methods
- We immunostained CPAP-depleted cells using the ninein antibody, which selectively detects subdistal appendages in mature mother centrioles.
-
Results
- Ninein signals were significantly impaired in CPAP-depleted cells.
-
Conclusion
- The results suggest that CPAP is required for mother centriole maturation in mammalian cells. The selective absence of centriolar appendages in young mother centrioles may be responsible for asymmetric spindle pole formation in CPAP-depleted cells.
- The centrosome is a major microtubule organization center in animal cells, and regulates cell polarity, shape, motility, and division; cilia also originate from the centrosome in nondividing cells. The centrosome is composed of a pair of centrioles surrounded by a protein matrix, referred to as the percentriolar material. Centrioles duplicate and segregate in tight association with the cell cycle. Procentrioles begin to form adjacent to mother centrioles when DNA is replicated during the S phase of the cycle. Procentrioles are elongated in the S and G2 phases, and eventually disengage from the mother centriole during the end stage of mitosis. Centriole disengagement is considered important for licensing a new round of centriole duplication [1,2]. A new procentriole is formed perpendicular to the disengaged centriole. However, this young mother centriole is still structurally immature; for example, it lacks distal and subdistal appendages until the latter stages of the G2 phase [3]. Therefore, one-and-a-half cell cycles are necessary for a procentriole to become a fully matured mother centriole.
- Mutations in centrosomal protein genes have been identified in a number of genetic diseases in brain development. Primary microcephaly is a rare, recessive genetic disease in which prenatal brain growth is significantly reduced while the brain structure is left intact [4]. It has been proposed that a reduced number of neuronal cells during development underlies microcephaly [5]. To date, seven genes in microcephaly patients have been discovered to possess mutations; the majority of these genes encode centrosome proteins [4].
- Centrosomal P4.1-associated protein (CPAP) is one of the causal genes implicated in primary microcephaly [5]. However, the reason why the number of neural cells is reduced in individuals with CPAP mutations is not well understood. We previously proposed that CPAP is essential for mother centriole maturation during mitosis [6]. To test this theory, we immunostained CPAP-depleted cells with the ninein antibody, which specifically detects subdistal appendages of mature mother centrioles.
INTRODUCTION
- Antibodies
- The ninein antibody was raised against GST-Ninein381-689, in which residues 381-689 of the human ninein protein were linked to the GST protein. The centrin-2 rabbit polyclonal antibody was raised against GST-Centrin-21-172. Antibodies against γ-tubulin (C-20, Santa Cruz Biotechnology, Santa Cruz, CA, USA), acetylated tubulin (T6793, Sigma, St. Louis, MO, USA), and cyclin B1 (GNS1, Santa Cruz) were purchased.
- Cell culture
- HeLa cells were cultured at 37℃ and 5% CO2 in high glucose DMEM supplemented with 10% fetal bovine serum. The cell cycle was synchronized; HeLa cells were arrested during the G1/S phase using a double-thymidine block, and released for 0, 4, 8, and 10 hours.
- RNA interference
- siCTL (scrambled sequence for control), (5'-GCA AUC GAA GCU CGG CUA CTT-3'), and siCPAP (5'-GGA CUG ACC UUG AAG AGA ATT-3') were used for RNAi experiments. The siRNAs were transfected into HeLa cells using RNAi MAX reagents (Invitrogen, Carlsbad, CA, USA).
- Immunoblot analysis
- HeLa cells were lysed in 1×SDS sample buffer (50 mM Tris-HCl, pH 6.8, 100 mM dithiothreitol, 2% SDS, 0.1% bromophenol blue, 10% glycerol). Samples were loaded on 4% polyacrylamide gels, and subsequently transferred onto nitrocellulose membranes. The membranes were blocked in blocking solution (5% skim milk in Tris-buffered saline with Triton X-100 [TBST]: 20 mM Tris, 150 mM NaCl, 0.3% Triton X-100) for 30 minutes, and incubated with anti-Ninein (1:200) overnight at 4℃. After washing three times with TBST for 7 minutes, the membranes were incubated with antirabbit secondary antibodies (1:10,000) for 30 minutes. Peroxidase activity was detected with X-ray film using ECL solutions.
- Immunofluorescence and microscopy
- HeLa cells on 12 mm coverslips were briefly washed with phosphate buffered saline (PBS) and fixed in cold 100% methanol for 10 minutes. The cells were then permeabilized and blocked in 3% bovine serum albumin (BSA) in PBS with 0.5% Triton X-100 (PBST) for 15 minutes. Primary antibodies were diluted in 3% BSA in 0.5% PBST, and incubated at room temperature for 2 hours. Coverslips were then washed three times with 0.1% PBST. Secondary antibodies were diluted in 3% BSA in 0.5% PBST, incubated for 30 minutes at room temperature, and washed an additional three times with 0.1% PBST. DNA was stained with 4',6-diamidino-2-phenylindole solution for 4 minutes. The coverslips were mounted on slides and observed using a fluorescence microscope (Olympus IX51, Olympus, Tokyo, Japan) equipped with a CCD camera (Qicam fast 1394, Qimaging, Surrey, BC, Canada). Cells were analyzed using a 60× lens. Images were processed using ImagePro 5.0 (Media Cybernetics, Rockville, MD, USA). Statistical analyses were performed with Sigma Plot (Systat Software, San Jose, CA, USA).
METHODS
- We began our study by generating a ninein-specific polyclonal antibody. Ninein is a large protein with 2096 amino acid residues (Fig. 1A). During antigen preparation, a ninein fragment (381-689 amino acid residues) was fused with the glutatione S-transferase (GST) tag and expressed in bacteria (Fig. 1A). Affinity-purified rabbit antiserum was used for immunoblot analysis. As reported previously, the ninein-specific band of the 240 kDa protein was detected in HeLa cell lysates (Fig. 1B) [7,8]. Accordingly, we used this antibody for immunostaining analysis of ninein in cultured cells.
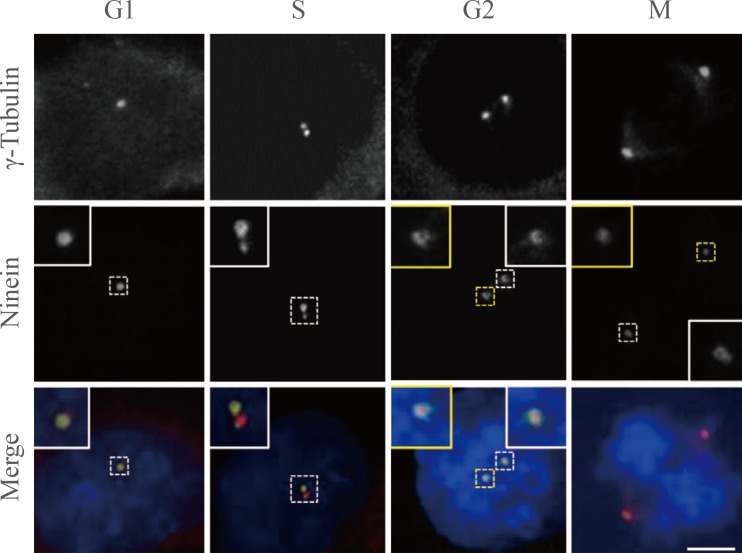
- We performed immunostaining analysis using HeLa cells whose cell cycle was synchronized with a double thymidine block and release. γ-Tubulin signals were detected as a single dot in G1 phase cells (Fig. 2). The second γ-tubulin dot was visible during the S phase, and became more prominent in G2 and M phase cells, suggesting that γ-tubulin dots represent centrosomes with mother centrioles (Fig. 2). The ninein signal was exclusively detected at the centrosome. Ninein formed either a ring or three distinct dots at the centrosome in G1 phase cells (Fig. 2). A ring-like structure of ninein was prominent at the one of the two γ-tubulin signal, and a tiny dot of ninein also appeared at the other γ-tubulin signal in S phase cells (Fig. 2). Ring-like signals of ninein were observed in G2 phase centrosomes, and became weaker in M phase centrosomes (Fig. 2). This suggests that ninein is primarily localized in mature, mother centrioles [9].
- To determine the precise localization of ninein within the centriole, we coimmunostained HeLa cells with ninein and centrin-2 antibodies. Ninein was localized at only one of the two unduplicated centrioles in G1 phase cells (Fig. 3). Ninein was detected in both the proximal and distal regions of the mother centriole, as either a ring or three dots (Fig. 3). A second ninein signal appeared as a dot at the daughter centriole during late-stage G1 (Fig. 3). Strong and weak ninein signals were located at the old and young mother centrioles, respectively (Fig. 3). This staining pattern was maintained until the young mother centriole reached maturity during late-stage G2 (data not shown).
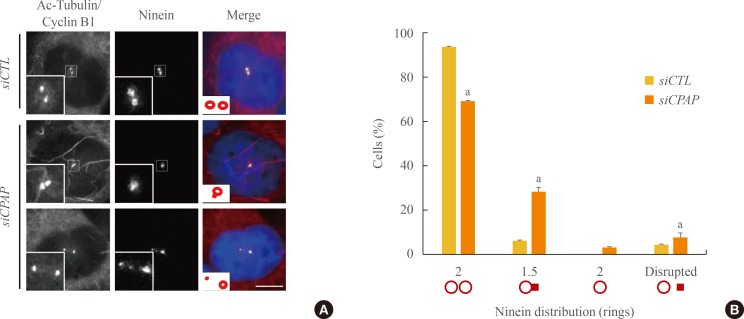
- CPAP is an essential component of centriole assembly, because CPAP depletion results in defects in centriole duplication [10]. We previously proposed that CPAP is also critical for the maturation of mother centrioles [6]. Therefore, we immunostained CPAP-depleted cells with the ninein antibody to determine the structure of mature mother centrioles. Ninein signals were detected in cyclinB1-positive G2 cells, which became weaker and dispersed in M phase cells [11,12]. The majority of the control cells possessed two ninein rings corresponding to two mother centrioles (Fig. 4A). In CPAP-depleted cells, however, the number of cells with two intact ninein rings was significantly reduced (Fig. 4A); more than 30% of the CPAP-depleted cells included an intact ninein ring with a dot representing an immature centriole appendage (Fig. 4B). These results support the notion that CPAP is required for maturation of the young mother centriole.
RESULTS
- Ninein is known to serve as a microtubule anchor at the subdistal appendages of mother centrioles in dividing cells [13]. In fact, we observed ninein signals at the distal end of mature mother centrioles. The ring-like staining pattern of ninein was observed in young mother centrioles only during the latter stages of the G2 phase, suggesting that the ninein antibody could be used as a marker for mature centrioles with subdistal appendages.
- In this study, we demonstrated that intact ninein signals were significantly reduced in CPAP-depleted cells. This suggests that CPAP is required for mother centriole maturation in mammalian cells. The selective absence of centriolar appendages in young mother centrioles may be responsible for asymmetric spindle pole formation in CPAP-depleted cells [6]. An absence of ninein at the mother centriole results in a reduction of the microtubule organizing activity of the centrosome [13]. In fact, removal of ninein disrupted asymmetric segregation and resulted in premature reduction of the progenitor during Amouse neocortex development [14]. A recent report also revealed that ninein is critical for maintenance of the cortical progenitor [15]. Our results revealed that the two microcephaly genes are functionally related.
DISCUSSION
-
Acknowledgements
- This study was supported by grants from the Basic Research Program (NRF-2012R1A2A2A01003512) and the Basic Research Laboratory (NRF-2014R1A4A1005259) of the Ministry of Education, Science and Technology.
ACKNOWLEDGMENTS
-
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.
Article information
- 1. Tsou MF, Stearns T. Mechanism limiting centrosome duplication to once per cell cycle. Nature 2006;442:947–951. ArticlePubMedPDF
- 2. Mardin BR, Schiebel E. Breaking the ties that bind: new advances in centrosome biology. J Cell Biol 2012;197:11–18. ArticlePubMedPMCPDF
- 3. Loncarek J, Khodjakov A. Ab ovo or de novo? Mechanisms of centriole duplication. Mol Cells 2009;27:135–142. ArticlePubMedPMCPDF
- 4. Thornton GK, Woods CG. Primary microcephaly: do all roads lead to Rome? Trends Genet 2009;25:501–510. ArticlePubMedPMC
- 5. Bond J, Roberts E, Springell K, Lizarraga SB, Scott S, Higgins J, Hampshire DJ, Morrison EE, Leal GF, Silva EO, Costa SM, Baralle D, Raponi M, Karbani G, Rashid Y, Jafri H, Bennett C, Corry P, Walsh CA, Woods CG. A centrosomal mechanism involving CDK5RAP2 and CENPJ controls brain size. Nat Genet 2005;37:353–355. ArticlePubMedPDF
- 6. Lee M, Chang J, Chang S, Lee KS, Rhee K. Asymmetric spindle pole formation in CPAP-depleted mitotic cells. Biochem Biophys Res Commun 2014;444:644–650. ArticlePubMedPMC
- 7. Bouckson-Castaing V, Moudjou M, Ferguson DJ, Mucklow S, Belkaid Y, Milon G, Crocker PR. Molecular characterisation of ninein, a new coiled-coil protein of the centrosome. J Cell Sci 1996;109(Pt 1):179–190. ArticlePubMedPDF
- 8. Chen CH, Howng SL, Cheng TS, Chou MH, Huang CY, Hong YR. Molecular characterization of human ninein protein: two distinct subdomains required for centrosomal targeting and regulating signals in cell cycle. Biochem Biophys Res Commun 2003;308:975–983. ArticlePubMed
- 9. Mogensen MM, Malik A, Piel M, Bouckson-Castaing V, Bornens M. Microtubule minus-end anchorage at centrosomal and non-centrosomal sites: the role of ninein. J Cell Sci 2000;113(Pt 17):3013–3023. ArticlePubMedPDF
- 10. Chang J, Cizmecioglu O, Hoffmann I, Rhee K. PLK2 phosphorylation is critical for CPAP function in procentriole formation during the centrosome cycle. EMBO J 2010;29:2395–2406. ArticlePubMedPMC
- 11. Cheng TS, Hsiao YL, Lin CC, Hsu CM, Chang MS, Lee CI, Yu RC, Huang CY, Howng SL, Hong YR. hNinein is required for targeting spindle-associated protein Astrin to the centrosome during the S and G2 phases. Exp Cell Res 2007;313:1710–1721. ArticlePubMed
- 12. Ou YY, Mack GJ, Zhang M, Rattner JB. CEP110 and ninein are located in a specific domain of the centrosome associated with centrosome maturation. J Cell Sci 2002;115(Pt 9):1825–1835. ArticlePubMedPDF
- 13. Delgehyr N, Sillibourne J, Bornens M. Microtubule nucleation and anchoring at the centrosome are independent processes linked by ninein function. J Cell Sci 2005;118(Pt 8):1565–1575. ArticlePubMed
- 14. Wang X, Tsai JW, Imai JH, Lian WN, Vallee RB, Shi SH. Asymmetric centrosome inheritance maintains neural progenitors in the neocortex. Nature 2009;461:947–955. ArticlePubMedPMCPDF
- 15. Shinohara H, Sakayori N, Takahashi M, Osumi N. Ninein is essential for the maintenance of the cortical progenitor character by anchoring the centrosome to microtubules. Biol Open 2013;2:739–749. ArticlePubMedPMC
References
Fig. 1

Generation of a ninein polyclonal antibody. (A) Ninein is a 2096-chain amino acid protein. The 381-689 fragment of ninein (solid bar) was used to generate the ninein antibody. (B) HeLa lysates were subjected to immunoblot analysis using the affinity-purified ninein antibody. The estimated size of ninein is approximately 240 kDa.

Fig. 2

Cell cycle stage-specific localization of ninein at the centrosome. HeLa cells were synchronized with a double thymidine block and release. Cells representative of each stage were coimmunostained with ninein (green) and γ-tubulin (red) antibodies. DNA was stained with 4',6-diamidino-2-phenylindole. Scale bar=10 µm. Insets are magnified views of the centrosomes.

Fig. 3
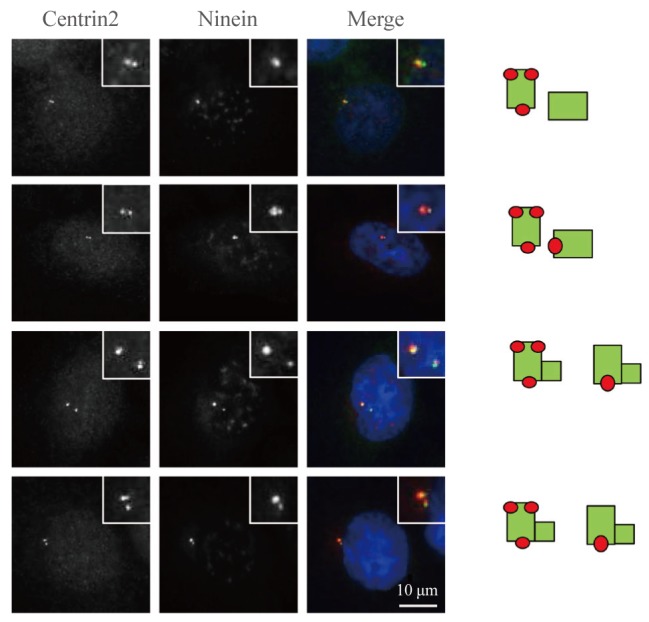
Ninein distribution within the centriole. Asynchronous HeLa cells were coimmunostained with centrin-2 (green) and ninein (red) antibodies. DNA was stained with 4',6-diamidino-2-phenylindole. Scale bar=10 µm. Insets are magnified views of the centrosomes. The staining patterns of ninein on centrioles were taken from the right side.

Fig. 4

Centrosomal ninein distribution in centrosomal P4.1-associated protein (CPAP)-depleted mitotic cells. (A) The cell cycle of CPAP-depleted cells was synchronized during the G2 phase using a double thymidine block and release. The cells were placed on ice for 90 minutes to disrupt microtubules, and coimmunostained with acetylated-tubulin (red), cyclin B1 (red), and ninein (green) antibodies. DNA was stained with 4',6-diamidino-2-phenylindole. Scale bar=10 µm. (B) Ninein staining patterns at the spindle poles were categorized into four groups: paired ring; ring with a bar; single ring; and disrupted. More than 300 cells were analyzed across three independent experiments. The data are means±SE. aP<0.05 was considered statistically significant compared to the controls.

Figure & Data
References
Citations
Citations to this article as recorded by 

- The intercentriolar fibers function as docking sites of centriolar satellites for cilia assembly
Sungjin Ryu, Donghee Ko, Byungho Shin, Kunsoo Rhee
Journal of Cell Biology.2024;[Epub] CrossRef - CPAP insufficiency leads to incomplete centrioles that duplicate but fragment
Alejandra Vásquez-Limeta, Kimberly Lukasik, Dong Kong, Catherine Sullenberger, Delgermaa Luvsanjav, Natalie Sahabandu, Raj Chari, Jadranka Loncarek
Journal of Cell Biology.2022;[Epub] CrossRef - Loss of CPAP causes sustained EGFR signaling and epithelial-mesenchymal transition in oral cancer
Radhika R. Gudi, Harinarayanan Janakiraman, Philip H. Howe, Viswanathan Palanisamy, Chenthamarakshan Vasu
Oncotarget.2021; 12(8): 807. CrossRef - NANOG/NANOGP8 Localizes at the Centrosome and is Spatiotemporally Associated with Centriole Maturation
Erika Mikulenkova, Jakub Neradil, Ondrej Vymazal, Jan Skoda, Renata Veselska
Cells.2020; 9(3): 692. CrossRef - ODF2/Cenexin maintains centrosome cohesion by restricting β-catenin accumulation
Kefei Yang, Marco Andreas Tylkowski, Daniela Hüber, Constanza Tapia Contreras, Sigrid Hoyer-Fender
Journal of Cell Science.2018;[Epub] CrossRef

 KES
KES
 PubReader
PubReader Cite
Cite