Genetic Studies on Diabetic Microvascular Complications: Focusing on Genome-Wide Association Studies
Article information
Abstract
Diabetes is a common metabolic disorder with a worldwide prevalence of 8.3% and is the leading cause of visual loss, end-stage renal disease and amputation. Recently, genome-wide association studies (GWASs) have identified genetic risk factors for diabetic microvascular complications of retinopathy, nephropathy, and neuropathy. We summarized the recent findings of GWASs on diabetic microvascular complications and highlighted the challenges and our opinion on future directives. Five GWASs were conducted on diabetic retinopathy, nine on nephropathy, and one on neuropathic pain. The majority of recent GWASs were underpowered and heterogeneous in terms of study design, inclusion criteria and phenotype definition. Therefore, few reached the genome-wide significance threshold and the findings were inconsistent across the studies. Recent GWASs provided novel information on genetic risk factors and the possible pathophysiology of diabetic microvascular complications. However, further collaborative efforts to standardize phenotype definition and increase sample size are necessary for successful genetic studies on diabetic microvascular complications.
INTRODUCTION
Diabetes mellitus is a chronic metabolic disorder that can result in multiple long-term micro- and macrovascular complications. Microvascular complications include retinopathy, nephropathy and neuropathy. Diabetes is well known as the leading cause of blindness, end-stage renal disease (ESRD) and limb amputation. In Korea, approximately 18.6% of diabetic patients have retinopathy, 27.3% albuminuria, and 33.5% diabetic neuropathy [1]. In a nationwide survey in 2012, diabetes accounted for 50.6% of new-onset ESRD in Korea [2]. Microvascular complications significantly affect the quality of life and impose a major burden on the healthcare system and economy.
The development of microvascular complications is related to several environmental risk factors, including duration of diabetes, degree of hyperglycemia, blood pressure, and dyslipidemia. In the landmark U.K. Prospective Diabetes Study, which enrolled newly diagnosed type 2 diabetes mellitus (T2DM) patients, participants randomized to intensive glucose control (median hemoglobin A1c [HbA1c] 7.0%) had a 25% reduction in microvascular complications including vitreous hemorrhage, retinal photocoagulation and ESRD compared with those in the conventional treatment group (median HbA1c 7.9%) after 10 years of follow-up [3]. In the recent Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial, intensive glycemic control targeting HbA1c <6.0% resulted in 23% reduction of retinopathy progression [4] and delayed onset of albuminuria and peripheral neuropathy [5]. Treatment with fenofibrate also decreased the risk of retinopathy in the ACCORD Eye Study [4].
Despite various interventions to control these environmental factors, large individual variations in the outcome of diabetic microvascular complications exist. Some patients with a short duration of diabetes develop microvascular complications although they had relatively good glycemic control. In contrast, some people do not develop microvascular complications even with a prolonged disease duration and with poor glycemic control. These clinical findings suggest that genetic factors play a role in the pathogenesis of microvascular complications. Familial aggregation in diabetic microvascular complications provides further evidence of genetic predisposition. For example, the heritability for diabetic retinopathy was estimated at 18% [6] and for proliferative diabetic retinopathy (PDR) at 52% [7]. The development of diabetic nephropathy also differs based on ethnicity and African Americans and Asians have 1.9- and 1.8-fold increased risks of ESRD, respectively, compared with European diabetic patients [8]. Consequently, efforts have been made to identify genetic risk factors for diabetic microvascular complications using candidate gene approach, linkage analysis and the recent genome-wide association studies (GWASs).
Based on biological or positional plausibility, numerous candidate genes were selected for genetic association studies. The most thoroughly investigated genes include vascular endothelial growth factor A (VEGFA) [9], aldo-keto reductase family 1, member B1 (AKR1B1) [10], and erythropoietin (EPO) [11] for diabetic retinopathy and angiotensin 1 converting enzyme (ACE) [12], protein kinase C β (PRKCB) [13], and erythropoietin (EPO) [11] for diabetic nephropathy. Although significant associations were initially reported, subsequent studies frequently showed inconsistent results. Most of the candidate gene association studies were conducted using a small sample size and had less stringent statistical thresholds. In addition, a meta-analysis showed that these candidate variants were not significantly associated on a genome-wide basis [14].
The advances in genotyping technology and publicly available databases of reference genomes and human genetic variations, including the International HapMap project [15], have contributed to understanding the genetic risk factors of common metabolic disorders using GWASs. In GWASs, hundreds of thousands or more of single-nucleotide variants are genotyped and tested for association with a disease or a continuous trait in several hundred or more subjects [16]. GWASs do not rely on previous knowledge and thus, are free from bias. Recently, GWASs have increased the number of genetic markers to more than one million by imputation methods and the sample size has increased to more than one hundred thousand by using meta-GWASs. The first successful GWAS on T2DM was published in 2007 [17]. Since then, at least 77 confirmed genetic loci for T2DM have been identified, [18] providing a better understanding of diabetes pathophysiology and there are ongoing efforts to use this genetic information in risk prediction and tailoring of individualized therapy [19]. In parallel, attempts have been made to unravel the genetic risk factors for diabetic microvascular complications using GWASs. In this article, we reviewed the recent GWASs on diabetic retinopathy, nephropathy and neuropathy and discussed their limitations and future directives.
DIABETIC RETINOPATHY
Diabetic retinopathy is clinically defined by the retinal microvascular lesions in diabetic patients and broadly classified into nonproliferative diabetic retinopathy (NPDR) and PDR. Diabetic macular edema can result in moderate visual loss and can be present at any stage of diabetic retinopathy, although more common in advanced retinopathy. The gold standard for classification of diabetic retinopathy severity is derived from the Early Treatment of Diabetic Retinopathy Study [20]. Diabetic retinopathy increases as the duration of diabetes increases. According to the Wisconsin Epidemiologic Study of Diabetic Retinopathy in T2DM patients, diabetic retinopathy is present in 20% of patients at the time of diagnosis, which increases to 60% to 85% after 15 years [21]. Whether the pathophysiology of retinopathy differs between type 1 diabetes mellitus (T1DM) and T2DM remains unknown. All heterogeneity factors-including the severity, duration, and type of diabetes-should be considered to understand the genetic risk factors for diabetic retinopathy.
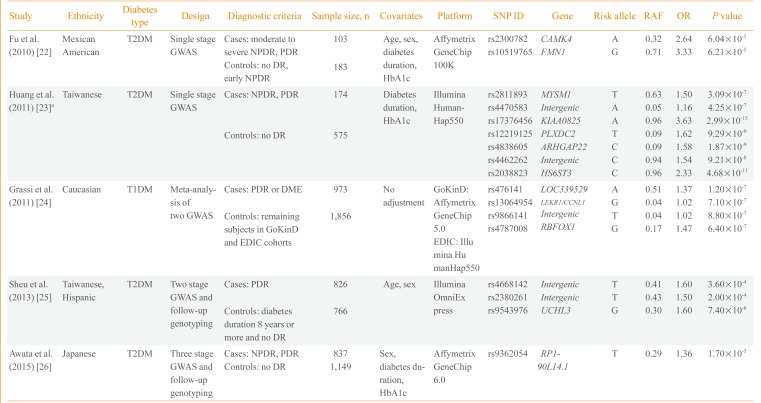
Currently, five GWASs on diabetic retinopathy have been published. The first GWAS performed included 283 Mexican-American T2DM retinopathy patients and controls (Table 1) [2223242526]. Fu et al. [22] found two potential loci in the introns of calcium/calmodulin-dependent protein kinase IV (CAMK4) and formin 1 (FMN1) genes. Huang et al. [23] reported a GWAS on T2DM diabetic retinopathy including 749 Taiwanese patients and found seven independent loci with potential significance (P<1.0×10-6). However, they reported the lowest P value among the six genetic models (genotype, allele, trend, additive, dominant, and recessive) and did not adjust for multiple comparisons. The third study is the largest GWAS conducted to date and is a meta-analysis of two GWASs, Genetics of Kidneys in Diabetes (GoKinD) and Epidemiology of Diabetes Interventions and Complications (EDIC) studies [24]. This study by Grassi et al. [24] involved 2,829 European subjects with T1DM. The most significant variant was rs476141 located in a long non-coding RNA (LOC339529) in chromosome 1 with P values of 1.20×10-7. The study by Sheu et al. [25] used a two-stage GWAS with follow-up genotyping in an independent population. Although they found three potential genetic variants in stage 1 GWAS in Taiwanese subjects, these findings were not replicated in the Hispanic population. The latest GWAS on diabetic retinopathy was a three-stage design performed by Awata et al. [26] in Japanese subjects. Among the eight variants that were followed-up to the third stage, none reached a genome-wide significance threshold. The most significant variant was located in RP1-90L14.1, a long non-coding RNA gene, with a P value of 1.70×10-5 in the meta-analysis of the three-stage results.
Overall, three studies were performed with Asian subjects, one with Mexican-American and one with European subjects. One study included T1DM patients and the remaining four studies included T2DM patients. The earliest two studies performed by Fu et al. [22] and Huang et al. [23] were single-stage GWASs with a small sample of fewer than 1,000 subjects. The findings were not replicated in an independent cohort using a different genotyping method. The majority of the genetic variants reported from the five studies did not pass the conventional significance threshold of P<5.0×10-8, except for several variants in the study by Huang et al. [23]. However, the latter study reported the best P value among various genetic models and did not correct for multiple comparisons. None of the genetic variants reported overlapped among the five studies; however, cases and controls were defined differently, which could be a crucial point when performing a genetic study. Regarding case groups, several studies included subjects with either NPDR or PDR, whereas others included only subjects with PDR. Regarding the control group, only in the study by Sheu et al. [25] the subjects were limited to those with a diabetes duration of more than 8 years without any diabetic retinopathy. The heterogeneity in study design and relatively small sample sizes could explain the inconsistencies in the genetic variants identified in the five GWASs on diabetic microvascular complications. A clear and rational definition of cases and controls is necessary to enhance genetic contrast as well as a large sample size to ensure sufficient statistical power.
DIABETIC NEPHROPATHY
Diabetic nephropathy is clinically defined as an increase in urinary albumin excretion and a decrease in kidney function. Classification of diabetic nephropathy using the Kidney Disease: Improving Global Outcomes group criteria is based on estimated glomerular filtration rate (eGFR) and the degree of proteinuria. The eGFR is generally calculated using the Modification of Diet in Renal Disease formula and is divided into five stages. The degree of proteinuria is used for substaging diabetic nephropathy. The eGFR reflects the current kidney function and proteinuria reflects the extent of pathological kidney damage. Proteinuria is a hallmark of diabetic nephropathy and precedes the decline in kidney function, but is not a prerequisite in some cases. A large body of evidence indicates that treatments to prevent or delay its progression should include intensive glycemic and blood pressure control. In the Action in Diabetes and Vascular Disease (ADVANCE) trial, intensive glycemic control resulted in the risk reduction for microalbuminuria (30 to 300 mg/g), macroalbuminuria (>300 mg/g) and ESRD, by 9%, 30%, and 65%, respectively [27]. In the pivotal study of Diabetes Control and Complications Trial (DCCT), intensive glucose control in T1DM patients resulted in 39% reduced occurrence of microalbuminuria [28]. However, 25% of participants in the intensive treatment group eventually developed microalbuminuria during the 6.5-year follow-up period. Therefore, individual variations in the risk of diabetic nephropathy exist and genetic factors likely play an important role.
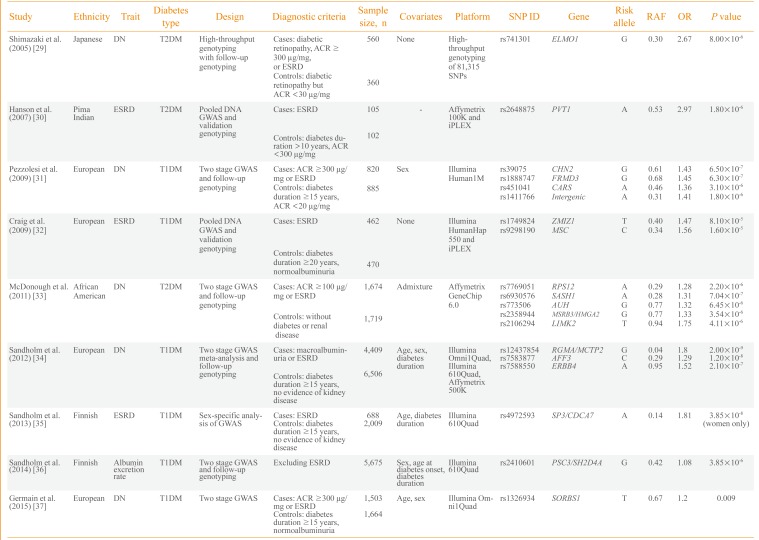
Consequently, efforts have been made to understand the genetic risk factors for diabetic nephropathy. Nine GWASs on diabetic nephropathy have been published (Table 2) [293031323334353637]. The first large-scale genotyping of more than 80,000 gene-based single nucleotide polymorphisms was performed in 2005 by Shimazaki et al. [29] in 920 Japanese T2DM patients. They identified an intronic variant, rs741301, of the engulfment and cell motility 1 (ELMO1) gene to be significantly associated with diabetic nephropathy. In vitro experiments suggested its role in the overaccumulation of extracellular matrix proteins and progression of glomerulosclerosis. Subsequently, a GWAS by Hanson et al. [30] used pooled DNA and validated the top signals using individual genotyping in 207 Pima Indian T2DM ESRD cases and controls. A variant in plasmacytoma variant translocation (PVT1) gene was suggestively associated with ESRD.
The first standard GWAS on diabetic nephropathy was published in 2009 by Pezzolesi et al. [31] and included the GoKinD study participants. They used two-stage GWAS with follow-up genotyping in 1,705 T1DM cases and controls. The four loci having a potential association signal of P<1.0×10-5 included FERM domain containing 3 (FRMD3), cysteinyl-tRNA synthetase (CARS), chimerin 2 (CHN2), and carboxypeptidase (CPVL). The rs1888747 variant in FRMD3 and rs451041 variant in CARS showed associations with time to onset of diabetic nephropathy in an independent cohort of DCCT/EDIC study with P<0.05. Craig et al. [32] reported a GWAS using pooled DNA of 932 T1DM GoKinD participants. They reported suggestive loci for ESRD on zinc finger, MIZ-type containing 1 (ZMIZ1) and musculin (MSC) genes as well as the association signals in six previously reported genetic loci of diabetic nephropathy (P≤0.0006). A relatively large-scale GWAS involving 3,393 African-American T2DM subjects was performed by McDonough et al. [33] in 2011. However, it was uncommon for African-Americans to have normal albuminuria after a diabetes duration of 10 years and the authors used nondiabetic controls for the control group. Therefore, analysis was performed to discriminate association signals between T2DM-associated ESRD, T2DM and all-cause ESRD using additional genotyping in African-Americans. Although none of the genetic variants reached genome-wide significance, several genes-including SAM and SH3 domain containing 1 (SASH1), ribosomal protein S12 gene (RPS12), and LIM kinase 2 (LIMK2)-were suggested as strong candidates for diabetic nephropathy in T2DM patients.
As previous studies were not fully powered for GWAS and the phenotype definition was different, a collaborative effort was made to conduct the Genetics of Nephropathy: An International Effort (GENIE) study on T1DM diabetic nephropathy [34]. This study was performed in two stages by Sandholm et al. [34]. In the first stage, a GWAS was meta-analyzed in three cohorts with a sample size of 5,783 subjects. In the second stage, de novo genotyping was performed in 5,873 participants from nine cohorts. The study was adequately powered and two variants were found associated with ESRD in AF4/FMR2 family, member 3 (AFF3) and in the intergenic region between repulsive guidance molecule family member A (RGMA) and multiple C2 domains, transmembrane 2 (MCTP2) that reached a genome wide significance threshold of P<5.0×10-8. In a GWAS on Finnish Diabetic Nephropathy study and GENIE consortium, Sandholm et al. [35] identified a gender-specific variant, rs4972593, which was associated with the risk of T1DM ESRD only in females. A GWAS was also conducted on urinary albumin excretion rate in 5,675 T1DM patients. This study identified rs2410601 in the intergenic region between pleckstrin and Sec7 domain containing 3 (PSD3) and SH2 domain containing 4A (SH2D4A) as the most significantly associated with albumin excretion rate (P=3.85×10-6) [36].
Among the nine GWASs, six included participants of European origin and one study each of Japanese subjects, Pima Indians and African-Americans. Only three studies were performed on T2DM, and the remaining six on T1DM subjects. Most of the studies included both ESRD and macroalbuminuria groups as cases. Only a few studies, including those from the GENIE consortium, had sufficient statistical power for GWAS and most of the earlier studies were limited in terms of sample size. Genetic variants in earlier reports, such as in ELMO1, were analyzed in subsequent studies and an association with diabetic nephropathy was confirmed in several, but not all, studies. Whether the pathophysiology of diabetic nephropathy differs between T1DM and T2DM patients remains unknown. Nonglycemic factors, such as insulin resistance and dyslipidemia, in T2DM may modulate the development of diabetic nephropathy and certain genetic risk factors could be involved in this process. Most of the well-powered GWASs were performed on T1DM patients and the genetic variants of diabetic nephropathy in T2DM patients should be elucidated.
DIABETIC NEUROPATHY
Diabetic peripheral neuropathy is classified as generalized symmetric polyneuropathies and focal and multifocal neuropathies [38]. Diabetic sensorimotor polyneuropathy is one of the most common complications in diabetic patients with an estimated lifetime prevalence of up to 50% [38]. In 2009, the Toronto Consensus Panel on Diabetic Neuropathies updated its definition and diagnostic criteria for diabetic polyneuropathy [39]. Diagnosis of distal symmetric polyneuropathy is categorized into possible, probable, confirmed, and subclinical, according to the certainty based on symptoms and signs. The confirmation of neuropathy requires typical symptoms, signs, and positive nerve conduction studies.
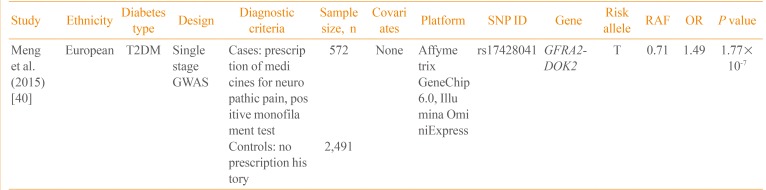
Currently, only one GWAS on neuropathic pain in diabetic patients has been published (Table 3) [40]. Using the Genetics of Diabetes Audit and Research Tayside (GoDARTS) study, Meng et al. [40] performed a GWAS on 3,063 T2DM patients. The case control status was defined based on the prescription of medications frequently used for diabetic sensorimotor polyneuropathy, including duloxetine, gabapentin, pregabalin, capsaicin, and lidocaine patch. In a single-stage GWAS without follow-up genotyping, rs17428041 located in the intergenic region between the GDNF family receptor alpha 2 (GFRA2) and docking protein 2 (DOK2) was potentially associated with neuropathic pain (P=1.77×10-7). In addition, the narrow sense heritability of diabetic neuropathic pain was 11%, excluding the effect of gene-gene and gene-environment interactions. Further studies are required to replicate this finding and to identify additional genetic variants of diabetic neuropathy.
CONCLUSIONS
During the past several years, the identification of genetic risk factors for diabetic microvascular complications has improved. However, most of the studies were not fully powered for GWASs, with the exception of the GENIE study. Therefore, most of the results associated with the genetic risk factors were below the genome-wide significance threshold and inconsistent among studies. In addition, the definition of cases and controls differed, thereby introducing significant heterogeneity. Based on the findings reported, these genetic association results should be validated in other populations. In addition, a collaborative effort to harmonize phenotype definitions and to increase sample size is necessary.
Whether certain microvascular complications are caused by specific genetic risk factors, or common genetic risk factors are shared by different microvascular complications should be clarified. Additionally, a possible difference in genetic risk factors for microvascular complications between T1DM and T2DM patients should be explored. Whether confirmed genetic variants for T1DM or T2DM per se have significant effects on the development of microvascular complications remains unclear. Finally, a metabolic memory or legacy effect, as shown by the DCCT/EDIC trial, should be considered; this might be mediated by epigenetic change. Compared to T2DM, genetic studies on diabetic microvascular complications are still in the early stages and have further challenges to overcome. Further genetic studies of microvascular complications will enhance understanding of their pathogenesis and facilitate the development of effective preventive and therapeutic measures.
ACKNOWLEDGMENTS
This research was supported by a grant of Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health and Welfare, Republic of Korea (grant number: HI14C0060).
Notes
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.