Metabolic and Cardiovascular Implications of a Metabolically Healthy Obesity Phenotype
Article information
Abstract
Metabolically healthy obesity (MHO) is a new concept in which an individual may exhibit an obese phenotype in the absence of any metabolic abnormalities. There are a number of definitions of MHO that utilize a variety of components. The findings of clinical and basic studies indicate that subjects with MHO do not exhibit an increased mortality, an increased risk of cardiovascular disease, or an increased risk of type 2 diabetes mellitus, as compared to normal-weight controls. Although these findings imply that metabolic health is a more important factor than obesity, several studies have shown that subjects with MHO have a similar risk of metabolic or cardiovascular diseases as those with metabolically unhealthy obesity. Thus, there is still debate regarding not only the implications of the MHO phenotype but its very existence. Accordingly, future studies should focus on developing a unified definition of MHO and distinguishing subjects who will be at a high risk for metabolic and cardiovascular diseases.
INTRODUCTION
The rise in the prevalence of obesity over the past decade has become an increasingly relevant issue because this medical condition is an important risk factor for type 2 diabetes mellitus (T2DM) and cardiovascular disease (CVD), which may lead to a higher incidence of all-cause mortality [1]. Approximately 30% of obese individuals do not have metabolic syndrome and are thus at a lower risk of developing T2DM and CVD [2]; these individuals are thought to exhibit metabolically healthy obesity (MHO), which is an obese phenotype free of any metabolic abnormalities [3]. Conversely, not all non-obese individuals exhibit a healthy metabolic profile [2].
The phenotypes of MHO, metabolically unhealthy obesity (MUHO), and nonobesity have been recognized since the 1980s [3] but between the years of 1980 and 2000 epidemiological research demonstrated that individuals who were overweight or obese did not always exhibit higher rates of CVD and mortality [4,5]. Subsequently, there has been an increase in the number of studies attempting to characterize the MHO phenotype [6,7,8], which is characterized by the absence of metabolic parameters such as dyslipidemia, insulin resistance, hypertension, and unfavorable inflammatory profiles despite the presence of an elevated body mass index (BMI) [8].
Several diverse definitions of the MHO phenotype have been proposed based on the presence of various metabolic abnormalities in a subject [9]. Accordingly, the prevalence rate of MHO in obese individuals varies from 10% to 25% based on the criteria and populations that are used by a particular study [10,11]. Despite the enhanced interest and increasing number of studies investigating MHO, there are no unique criteria that can be used to define MHO and, as a result, the implications of the MHO phenotype and even its very existence are still under debate. Thus, the present review discusses the various definitions, pathophysiologies, and clinical implications of MHO as well as the debate regarding its existence.
THE MHO PHENOTYPE: DEFINITION MATTERS
Individuals with MHO display a favorable metabolic profile that is characterized by a high level of insulin sensitivity, a low incidence of hypertension, favorable lipid profiles, satisfactory fat distribution, and a low level of systemic inflammatory responses [10]. It has been suggested that the MHO phenotype exists, even though there are large variations in its prevalence rate, as a function of the criteria used to define it [12].
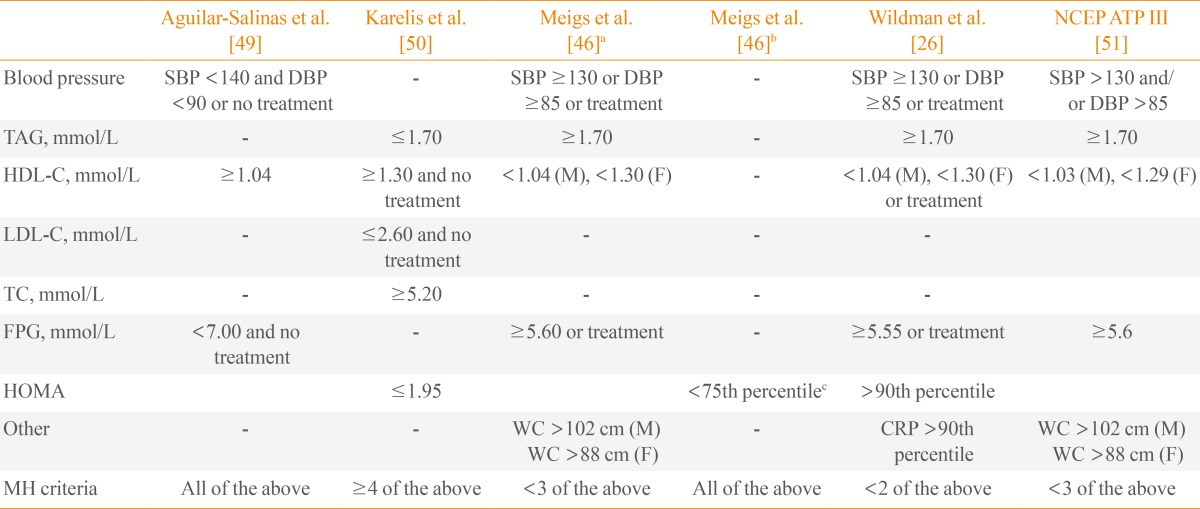
Several definitions are currently used to account for metabolic health (Table 1) and most authors define MHO using measures of blood pressure, lipid status (mainly high density lipoprotein cholesterol and/or triglycerides), glycemic and insulin resistance statuses (fasting blood glucose levels or homeostasis model assessment [HOMA]), and systemic inflammation (as defined by high-sensitivity C-reactive protein levels). However, it is difficult to compare the findings of various studies because there is no accepted standard definition of metabolic health and different studies use different inclusion criteria and/or cutoff points for metabolic measures.
Among the inclusion criteria used in various studies, there is a diversity of adiposity measures although they typically include waist circumference (WC), BMI, and body fat percentage (BF%) [9]. The prevalence of metabolic health was investigated in a population of Swedish subjects using six sets of criteria for obesity, which included measures such as BMI, abdominal obesity, and BF% [9]. Based on the different sets of criteria, the prevalence of MHO ranged between 3.3% and 43.1% in men and 11.4% and 57.5% in women. Similarly, the proportions of MHO individuals among obese participants were 6.8% when using the criteria of Aguilar-Salinas et al., 14.2% when using the criteria of Karelis et al., 23.7% when using the criteria of Wildman et al., 30.2% when using the criteria of Meigs et al., and 36.6% when using HOMA [13,14]. Furthermore, the agreement of MHO classifications among these studies was poor. Likewise, the proportions of MHO individuals in a Korean population were 24.2% when using the criteria of Meigs et al., 28.5% when using the criteria of Karelis et al., and 59.7% when using the criteria of Wildman et al. [15].
These comparative studies illustrate the considerable variability that exists in estimations of the incidence of MHO when different sets of criteria are used to define metabolic health [14]. Despite the controversy surrounding these definitions and doubts concerning the existence of the MHO phenotype, a characterization of MHO individuals represents an important tool that can be used to evaluate the contribution of various types of fat distribution to the development of metabolic diseases and CVD. Additional studies and a consensus regarding the MHO phenotype are necessary in order to determine the manner in which this tool can be used to assess the cardiometabolic status of a patient in relation to appropriate fat accumulation.
PATHOPHYSIOLOGY OF MHO
It is likely that he MHO phenotype is the result of several underlying mechanisms and the interaction between genetic, environmental, and behavioral factors (Fig. 1). Each of these factors affects abdominal fat distribution, visceral and ectopic fat accumulation, and insulin resistance, which are all important causative factors that contribute to the development of unhealthy obesity [10]. In some instances, the concept of the MHO phenotype is used interchangeably with the notion of insulin-sensitive obesity because MHO subjects display better insulin sensitivity than MUHO subjects [16].
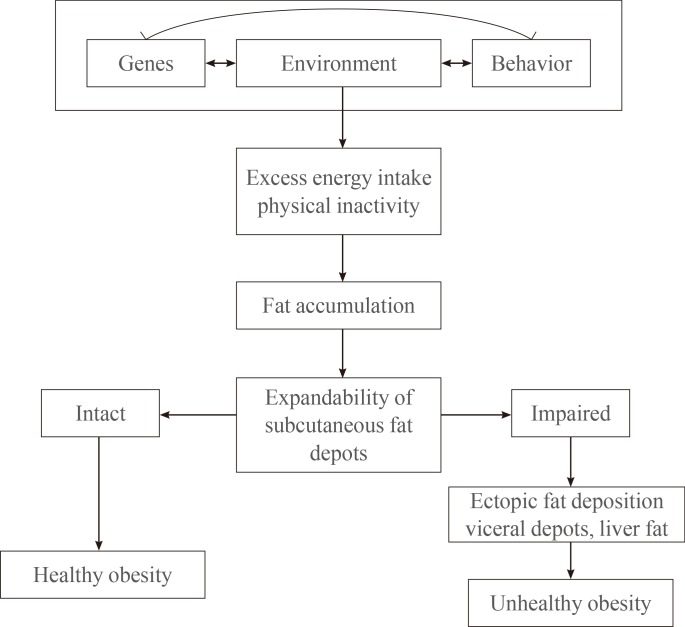
Possible pathophysiologies of metabolically healthy obesity and metabolically unhealthy obesity. The gene-environment interactions may play dual roles as both causative factors for the development of obesity and for the dissociation of obesity into subphenotypes of physiological (predominantly subcutaneous, insulin sensitive, and healthy) or pathological (visceral, ectopic, and unhealthy) fat accumulation. Adapted from Bluher, Curr Opin Lipidol 2010;21:38-43, with permission from Wolters Kluwer Health [10].
The plasticity of adipose tissue, which allows for the storage of the excessive fat that is accumulated by obese individuals, may be the primary factor that discriminates healthy obesity from unhealthy obesity. The amount of visceral fat mass in insulin-resistant obese individuals is independently associated with BMI and total body fat mass [17] while an artificial increase in the subcutaneous fat mass of rodents results in positive metabolic effects [18]. In MUHO individuals, the storage capacity of adipocytes may be exceeded and lipids can accumulate ectopically in visceral fat depots, liver and muscle cells, and pancreatic β-cells whereas the subcutaneous adipose tissue of MHO individuals possesses the (intrinsic) propensity to expand which, in turn, leads to a preservation of insulin sensitivity [10]. Additionally, MHO subjects exhibit a greater proportion of subcutaneous fat compared with MUHO subjects [19]. Therefore, the plasticity of adipose tissue that allows for expansion in response to excess fat in obese individuals with proper angiogenesis is important for healthy obesity [20].
The interaction between genes and the environment will also matter in the development of MHO. Fat mass, fat distribution and the number of adipocytes are heritable traits for which genes account for between 25% and 70% of the observed variability [21]. A number of studies have attempted to determine whether there would be a common genetic influence of these two phenotypes in addition to their gene-specific effects associated with fat mass and visceral fat. Developmental genes such as HoxA5, Gpc4, and Tbx15 exhibit changes in expression that are closely correlated with patterns of fat distribution [22], and B6J mice gain more weight, have higher levels of insulin and leptin, and show a greater degree of glucose intolerance than 129J mice on a normal diet [23]. Kulkarni et al. [24] generated mice from three genetic backgrounds (C57BL/6 [B6], 129Sv, and DBA) that were double heterozygous knockouts of the insulin receptor and insulin receptor substrate-1 and found that 90% of B6J mice developed diabetes while less than 5% of 129J mice developed diabetes. Additionally, a genome-wide scan of an intercross between these strains indicated that at least four loci on three different chromosomes are involved in this process [25]. In a study of obesity-discordant monozygotic twins (age range, 22.8 to 35.8 years), the metabolic responses to obesity differed greatly. Approximately half of the obese co-twins exhibited a typical response to obesity that was characterized by marked insulin resistance, dyslipidemia, and fatty liver tissue whereas the other half of the obese co-twins were as metabolically healthy as their lean co-twins [16]. In the same study, the MHO group exhibited a very low percentage of liver fat and the maintenance of mitochondrial function in conjunction with the absence of inflammation in subcutaneous adipose tissue.
The MHO phenotype may also be characterized by the presence of adipose tissue with reduced secretory capabilities or diminished responsiveness to the effects of adipokines [26]. It has been shown that fat distribution and differences in the adipocytokine secretory properties of fat depots are likely play an important role in the outcomes associated with the MHO phenotype [27]. Furthermore, changes during the adipose expansion process can influence the secretion of good adipokines, such as adiponectin [28], which promotes insulin sensitivity, decreases inflammation, and enhances cell survival [29]. In fact, obese co-twins who are as metabolically healthy as their lean co-twins exhibit a disproportionate increase in circulating leptin levels and a lower expression level of adiponectin in adipose tissue [16].
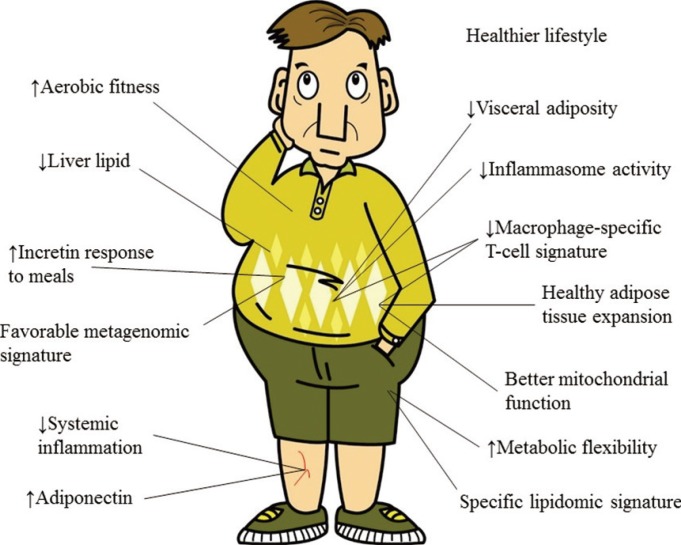
Low systemic inflammation may be another key feature of the obesity status of an individual. For example, of 44 obese (BMI >30 kg/m2) patients who underwent laparoscopic bariatric surgery, the MUHO patients had a less favorable inflammatory profile in their visceral adipose tissue that resulted from the infiltration of proinflammatory macrophages, which exhibited increased nucleotide-binding oligomerization domain-like receptor family pyrin domain-containing 3 inflammasome activity and interleukin-1 β production [30]. Adipose tissue also acquires immunological properties via the infiltration of activated macrophages, neutrophils, and T- and B-cells [31] and these immunometabolic interactions lead to low-grade inflammation. In fact, the involvement of the immune system in obesity-related metabolic diseases in humans leads to a specific T-cell signature in adipose tissue [32]. There are additional mechanisms and various differences that distinguish the MHO and MUHO phenotypes, such as improved mitochondrial function, increased aerobic fitness, and a greater degree of insulin sensitivity (Fig. 2), but the precise mechanisms supporting the MHO concept remain to be elucidated.
CLINICAL IMPLICATIONS OF MHO: MORTALITY AND CARDIOVASCULAR DISEASE
A number of studies have reported that the MHO phenotype is associated with increases in mortality, CVD, T2DM, and non-alcoholic fatty liver disease. In a prospective cohort study of 22,654 individuals between the ages of 20 and 59 years (average duration, 13.4 years; EPIC-MORGEN), the mortality risk of metabolically healthy abdominal obese (MHAO) subjects was investigated using the criteria of the Adult Treatment Panel III report of the National Cholesterol Education Program criteria and a definition of obesity based on WC [33]. These authors found that the mortality risk of MHAO individuals was approximately 40% higher than that of metabolically healthy non-abdominal obese individuals [33]. However, regardless of the definition, MHO and metabolically abnormal obese individuals have been found to have an increased risk of mortality relative to metabolically healthy normal-weight individuals [12]. A 30-year follow-up cohort study of obese men with and without metabolic syndrome identified a 2.4- and 1.7-fold higher risk of mortality, respectively, compared to normal-weight subjects without metabolic syndrome [2]. These studies suggest that the MHO phenotype may not accurately predict increased mortality.
There is also conflicting evidence regarding the association of the MHO phenotype with subclinical markers of atherosclerosis. Compared to MHO subjects, MUHO subjects exhibit a significant increase in the odds ratio (OR) for subclinical atherosclerosis as assessed by coronary artery calcium (CAC) scores [34,35]. In a similar population, MHO subjects exhibited a significantly increased OR for CAC scores, which were attenuated when metabolic risk factors were adjusted for [36]. These findings suggest that the definition of the MHO phenotype should include the absence of any metabolic components. However, when MHO individuals were followed up during longitudinal studies investigating increases in the risks of CVD and all-cause mortality, there were conflicting findings. A 7-year study found that MHO subjects were not at an increased risk of CVD or all-cause mortality compared with healthy non-obese individuals [37] while another study identified a higher prevalence and a greater severity of angiographic coronary artery disease among MUHO and normal-weight subjects, as compared to MHO and normal-weight subjects [38]. An extended follow-up period (>15 years) revealed that obese participants without metabolic syndrome at baseline had an increased risk of major CVD events compared with non-obese subjects who were healthy at baseline [39]. In a Norwegian cohort of more than 60,000 people who were free of CVD, the findings of a 12-year follow-up period revealed that there was an increased risk of acute myocardial infarction among MUHO individuals and that obesity was more important than metabolic factors in the development of heart failure [40].
CLINICAL IMPLICATIONS: T2DM
A majority of the studies investigating the relationship between the MHO phenotype and the risk of T2DM are longitudinal. During a 20-year follow-up period conducted by the Uppsala Longitudinal Study of Adult Men (ULSAM) study, the risk of diabetes was significantly increased in normal-weight individuals with metabolic syndrome (OR, 3.28), overweight subjects without metabolic syndrome (OR, 3.49), overweight subjects with metabolic syndrome (OR, 7.77), obese subjects without metabolic syndrome (OR, 11.72), and obese subjects with metabolic syndrome (OR, 10.06), as compared to normal-weight subjects without metabolic syndrome [2]. Similarly, another study found that MHO individuals were at a threefold increased risk of developing diabetes but only if they progressed to an unhealthy phenotype during the follow-up period (5.5 to 10.3 years) [41]. In a study of Mexican-Americans and non-Hispanic whites (San Antonio Heart Study) [42], both metabolically unhealthy normal-weight and MHO individuals had a 2.5-fold increased risk of developing diabetes. Likewise, middle-aged Asian MHO individuals had a higher risk of developing T2DM than their non-obese counterparts during a 5.4-year study [43]. However, in a study of 6,748 Koreans with a 4-year follow-up period, MHO subjects did not have an increased risk of T2DM compared with metabolically healthy non-obese subjects, which suggests that metabolic health is a more important determinant of the development of diabetes than obesity [44].
MYTH OF MHO
As mentioned above, a number of studies have provided strong evidence supporting the existence of the MHO phenotype. However, debate remains whether MHO individuals are truly healthy, especially because no consensus has been reached regarding an accepted definition of MHO or the influence of this phenotype on morbidity and mortality [39,40,45]. Nonetheless, the MHO-like phenotype does not appear to be associated with a significant increase in the risk for CVD [46] especially because, as in the ULSAM study, an increased risk for CVD was observed in normal-weight subjects with metabolic syndrome, obese subjects without metabolic syndrome, and obese subjects with metabolic syndrome when compared to normal-weight individuals without metabolic syndrome [39]. However, that study did not support the existence of an MHO phenotype when defining this concept based on the absence of metabolic syndrome.
Very recently, a systematic review of eight studies (n=61,836 subjects) investigated the associations of BMI and metabolic status with total mortality and cardiovascular events [47] and found that MHO individuals have an increased risk of cardiovascular events, as compared to metabolically healthy normal-weight individuals. This study also revealed that all metabolically unhealthy subjects, including normal-weight, overweight, and obese individuals, had a similarly elevated risk of cardiovascular events. The researchers concluded that obese individuals are at increased risk for adverse long-term outcomes, even in the absence of metabolic abnormalities, compared with metabolically healthy normal-weight individuals and emphasized that there is no healthy pattern of increased weight.
However, the findings of the present meta-analysis should be interpreted with caution as there are several limitations inherent in this study. The majority of studies included in this review provided relatively inadequate information regarding the health behaviors of the participants, they did not present data concerning weight gain in the subjects, they tended to focus only on total mortality and cardiovascular events, and they did not include older participants. According to a very recent editorial by Hill and Wyatt [48], along with the urgent focus to treat obesity regardless of the presence or absence of metabolic risk factors, it is important to reduce the long-term risk of death and cardiovascular events, as with any other chronic disease. However, the findings of the present meta-analysis indicate that any interventions or attempts to treat obesity should target not only the reduction of weight but also the type and proportion of fat that should be reduced. Additionally, the manner in which this is achieved is important because the treatment of obesity without any consideration of the metabolic risk factors is not likely to improve the lives of patients.
CONCLUSIONS
The implications and very existence of the MHO phenotype are still associated with controversy due to the discrepancies among studies regarding the criteria used to define obesity, the experimental designs of these studies, and the ethnicity of the subjects included in these studies. Although the clinical implications of MHO, such as cardiovascular mortality, CVD, and the development of diabetes, cannot be conclusively determined, the concept of a MHO phenotype is important as a tool for the stratification of patients at a high risk for metabolic and CVDs. However, a more unified set of criteria that can be used to define metabolic health needs to be established and clinical efforts aiming to treat obese patients should target reductions in body weight as well as improvements in metabolic risks. If this goal cannot be accomplished, it would be like running from Athens to Marathon without carrying the message box.
Notes
No potential conflict of interest relevant to this article was reported.