Search
- Page Path
- HOME > Search
Original Article
- Docosahexanoic Acid Attenuates Palmitate-Induced Apoptosis by Autophagy Upregulation via GPR120/mTOR Axis in Insulin-Secreting Cells
- Seok-Woo Hong, Jinmi Lee, Sun Joon Moon, Hyemi Kwon, Se Eun Park, Eun-Jung Rhee, Won-Young Lee
- Received August 21, 2023 Accepted November 6, 2023 Published online January 23, 2024
- DOI: https://doi.org/10.3803/EnM.2023.1809 [Epub ahead of print]
- 854 View
- 34 Download
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
Polyunsaturated fatty acids (PUFAs) reportedly have protective effects on pancreatic β-cells; however, the underlying mechanisms are unknown.
Methods
To investigate the cellular mechanism of PUFA-induced cell protection, mouse insulinoma 6 (MIN6) cells were cultured with palmitic acid (PA) and/or docosahexaenoic acid (DHA), and alterations in cellular signaling and apoptosis were examined.
Results
DHA treatment remarkably repressed caspase-3 cleavage and terminal deoxynucleotidyl transferase-mediated UTP nick end labeling (TUNEL)-positive red dot signals in PA-treated MIN6 cells, with upregulation of autophagy, an increase in microtubule- associated protein 1-light chain 3 (LC3)-II, autophagy-related 5 (Atg5), and decreased p62. Upstream factors involved in autophagy regulation (Beclin-1, unc51 like autophagy activating kinase 1 [ULK1], phosphorylated mammalian target of rapamycin [mTOR], and protein kinase B) were also altered by DHA treatment. DHA specifically induced phosphorylation on S2448 in mTOR; however, phosphorylation on S2481 decreased. The role of G protein-coupled receptor 120 (GPR120) in the effect of DHA was demonstrated using a GPR120 agonist and antagonist. Additional treatment with AH7614, a GPR120 antagonist, significantly attenuated DHA-induced autophagy and protection. Taken together, DHA-induced autophagy activation with protection against PA-induced apoptosis mediated by the GPR120/mTOR axis.
Conclusion
These findings indicate that DHA has therapeutic effects on PA-induced pancreatic β-cells, and that the cellular mechanism of β-cell protection by DHA may be a new research target with potential pharmacotherapeutic implications in β-cell protection.

Review Article
- Diabetes
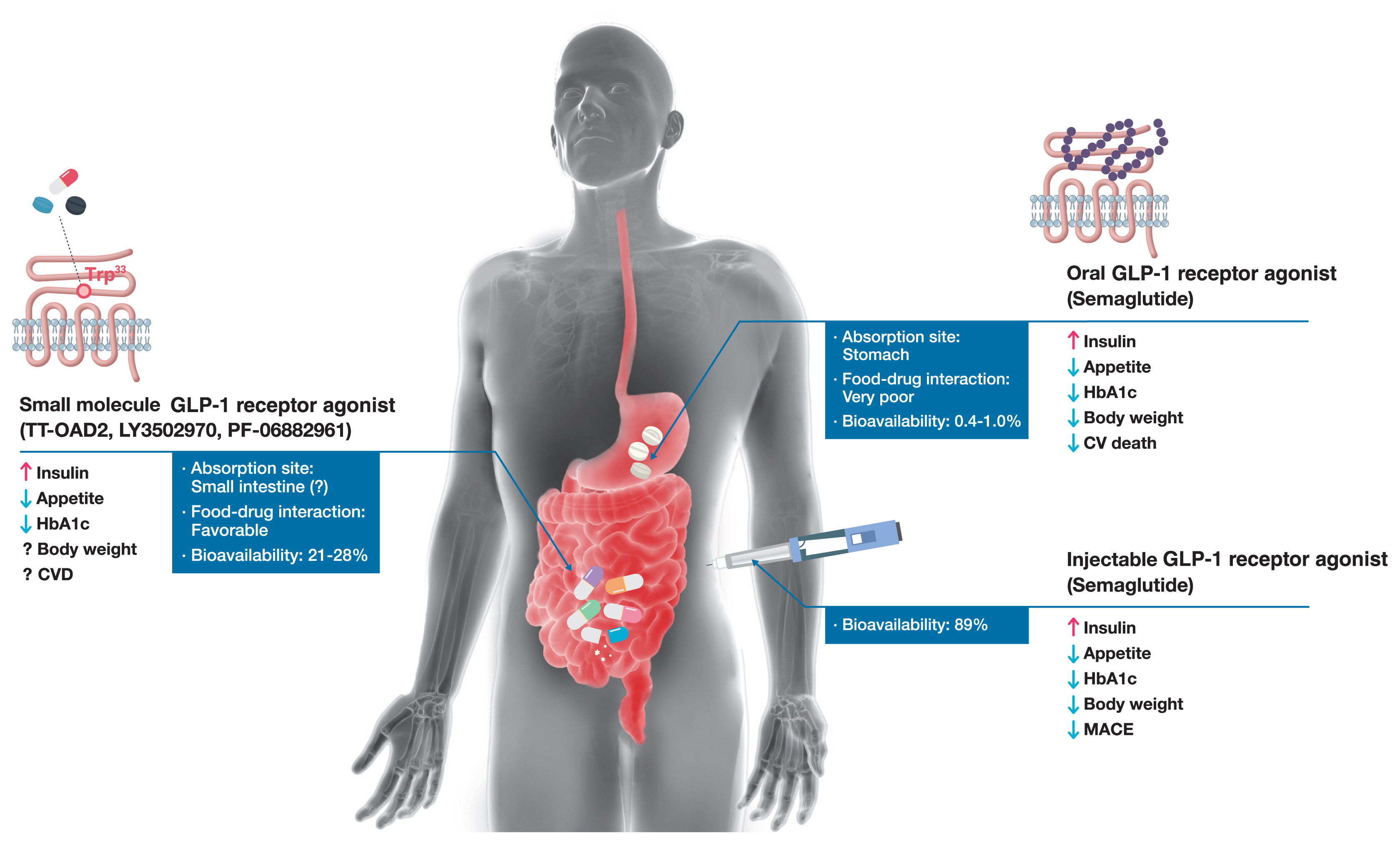
- Peptidyl and Non-Peptidyl Oral Glucagon-Like Peptide-1 Receptor Agonists
- Hun Jee Choe, Young Min Cho
- Endocrinol Metab. 2021;36(1):22-29. Published online February 24, 2021
- DOI: https://doi.org/10.3803/EnM.2021.102
- 27,836 View
- 654 Download
- 11 Web of Science
- 13 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Glucagon-like peptide-1 (GLP-1) receptor agonists are efficacious glucose-lowering medications with salient benefits for body weight and cardiovascular events. This class of medications is now recommended as the top priority for patients with established cardiovascular disease or indicators of high risk. Until the advent of oral semaglutide, however, GLP-1 receptor agonists were available only in the form of subcutaneous injections. Aversion to needles, discomfort with self-injection, or skin problems at the injection site are commonly voiced problems in people with diabetes, and thus, attempts for non-invasive delivery strategies have continued. Herein, we review the evolution of GLP-1 therapy from its discovery and the development of currently approved drugs to the unprecedented endeavor to administer GLP-1 receptor agonists via the oral route. We focus on the pharmacokinetic and pharmacodynamic properties of the recently approved oral GLP-1 receptor agonist, oral semaglutide. Small molecule oral GLP-1 receptor agonists are currently in development, and we introduce how these chemicals have addressed the challenge posed by interactions with the large extracellular ligand binding domain of the GLP-1 receptor. We specifically discuss the structure and pharmacological properties of TT-OAD2, LY3502970, and PF-06882961, and envision an era where more patients could benefit from oral GLP-1 receptor agonist therapy.
-
Citations
Citations to this article as recorded by- Sulfobetaine modification of poly (D, L-lactide-co-glycolic acid) nanoparticles enhances mucus permeability and improves bioavailability of orally delivered liraglutide
Zhenyu Zhao, Ruihuan Ding, Yumei Wang, Ranran Yuan, Houqian Zhang, Tianyang Li, Wei Zheng, Entao Chen, Aiping Wang, Yanan Shi
Journal of Drug Delivery Science and Technology.2024; 93: 105437. CrossRef - Physiology and pharmacology of glucagon-like peptide-1 receptor
D. V. Kurkin, D. A. Bakulin, E. I. Morkovin, V. I. Petrov, A. V. Strygin, K. N. Koryanova, Yu. V. Gorbunova, Yu. A. Kolosov, O. V. Ivanova, E. V. Pavlova, M. A. Dzhavakhyan, A. V. Zaborovsky, V. B. Saparova, I. E. Makarenko, R. I. Drai, A. N. Chumachenko
Pharmacy & Pharmacology.2024; 11(4): 347. CrossRef - G protein-coupled receptors driven intestinal glucagon-like peptide-1 reprogramming for obesity: Hope or hype?
Mohan Patil, Ilaria Casari, Leon N. Warne, Marco Falasca
Biomedicine & Pharmacotherapy.2024; 172: 116245. CrossRef - Glucagon-like peptide-1 analogs: Miracle drugs are blooming?
Binbin Gong, Zhihong Yao, Chenxu Zhou, Wenxi Wang, Lidan Sun, Jing Han
European Journal of Medicinal Chemistry.2024; 269: 116342. CrossRef - Opportunities and challenges of incretin-based hypoglycemic agents treating type 2 diabetes mellitus from the perspective of physiological disposition
Yaochen Xie, Qian Zhou, Qiaojun He, Xiaoyi Wang, Jincheng Wang
Acta Pharmaceutica Sinica B.2023; 13(6): 2383. CrossRef - Advances in GLP-1 receptor agonists for the treatment of type 2 diabetes
Shurui Hong, J. Xiao, Y. He
BIO Web of Conferences.2023; 61: 01006. CrossRef - Safety and efficacy of the new, oral, small-molecule, GLP-1 receptor agonists orforglipron and danuglipron for the treatment of type 2 diabetes and obesity: systematic review and meta-analysis of randomized controlled trials
Paschalis Karakasis, Dimitrios Patoulias, Konstantinos Pamporis, Panagiotis Stachteas, Konstantinos I. Bougioukas, Aleksandra Klisic, Nikolaos Fragakis, Manfredi Rizzo
Metabolism.2023; 149: 155710. CrossRef - A review of glucoregulatory hormones potentially applicable to the treatment of Alzheimer’s disease: mechanism and brain delivery
Reeju Amatya, Kyoung Ah Min, Meong Cheol Shin
Journal of Pharmaceutical Investigation.2022; 52(2): 195. CrossRef - Anti-Obesity Medications and Investigational Agents: An Obesity Medicine Association (OMA) Clinical Practice Statement (CPS) 2022
Harold E. Bays, Angela Fitch, Sandra Christensen, Karli Burridge, Justin Tondt
Obesity Pillars.2022; 2: 100018. CrossRef - Structural basis of peptidomimetic agonism revealed by small-molecule GLP-1R agonists Boc5 and WB4-24
Zhaotong Cong, Qingtong Zhou, Yang Li, Li-Nan Chen, Zi-Chen Zhang, Anyi Liang, Qing Liu, Xiaoyan Wu, Antao Dai, Tian Xia, Wei Wu, Yan Zhang, Dehua Yang, Ming-Wei Wang
Proceedings of the National Academy of Sciences.2022;[Epub] CrossRef - Improved Split TEV GPCR β-arrestin-2 Recruitment Assays via Systematic Analysis of Signal Peptide and β-arrestin Binding Motif Variants
Yuxin Wu, Isabelle von Hauff, Niels Jensen, Moritz Rossner, Michael Wehr
Biosensors.2022; 13(1): 48. CrossRef - GLP-1 Receptor Agonists: Beyond Their Pancreatic Effects
Xin Zhao, Minghe Wang, Zhitong Wen, Zhihong Lu, Lijuan Cui, Chao Fu, Huan Xue, Yunfeng Liu, Yi Zhang
Frontiers in Endocrinology.2021;[Epub] CrossRef - Recent developments in GLP‐1RA therapy: A review of the latest evidence of efficacy and safety and differences within the class
Evie K. Bain, Stephen C. Bain
Diabetes, Obesity and Metabolism.2021; 23(S3): 30. CrossRef
- Sulfobetaine modification of poly (D, L-lactide-co-glycolic acid) nanoparticles enhances mucus permeability and improves bioavailability of orally delivered liraglutide


 KES
KES

 First
First Prev
Prev



