Articles
- Page Path
- HOME > Endocrinol Metab > Volume 29(3); 2014 > Article
-
Original ArticleEndocrine Research Vav3, a GEF for RhoA, Plays a Critical Role under High Glucose Conditions
- Jie Sha1*, Jungsik Na2*, Jung Ok Lee1, Nami Kim1, Soo Kyung Lee1, Ji Hae Kim1, Ji Wook Moon1, Su Jin Kim1, Hye Jeong Lee1, Jong-Il Choi3, Sun Hwa Park1, Hyeon Soo Kim1
-
Endocrinology and Metabolism 2014;29(3):363-370.
DOI: https://doi.org/10.3803/EnM.2014.29.3.363
Published online: September 25, 2014
1Department of Anatomy, Korea University College of Medicine, Seoul, Korea.
2Bando Hospital, Korea.
3Division of Cardiology, Department of Internal Medicine, Korea University Anam Hospital, Korea University College of Medicine, Seoul, Korea.
- Corresponding author: Hyeon Soo Kim. Department of Anatomy, Korea University College of Medicine, 73 Inchon-ro, Seongbuk-gu, Seoul 136-705, Korea. Tel: +82-2-920-6151, Fax: +82-2-920-5696, anatomykim@korea.ac.kr
- *These authors contributed equally to this work.
• Received: November 22, 2013 • Revised: January 27, 2014 • Accepted: February 10, 2014
Copyright © 2014 Korean Endocrine Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
-
Background
- The role of small GTPase molecules is poorly understood under high glucose conditions.
-
Methods
- We analyzed the expression pattern of Vav3 in skeletal muscle C2C12 cells under high glucose culture condition with reverse transcription-polymerase chain reaction and Western blot analysis. We also measured glucose uptake using isotope-labelled glucose.
-
Results
- We showed that expression of Vav3 (a guanine nucleotide exchange factor for RhoA) increased. mRNA and protein levels in skeletal muscle C2C12 cells under high glucose conditions. The AMP-activated protein kinase (AMPK) activator AMPK agonist 5-aminoimidazole-4-carboxy-amide-1-d-ribofuranoside (AICAR) suppressed high glucose-induced Vav3 induction. In addition, exposure of cells to high glucose concentration increased the phosphorylation of PAK-1, a molecule downstream of RhoA. The phosphorylation of paxillin, a downstream molecule of PAK-1, was also increased by exposure to high glucose. Phosphorylation of these molecules was not observed in the presence of AICAR, indicating that AMPK is involved in the RhoA signal pathway under high glucose conditions. Knock down of Vav3 enhances metformin-mediated glucose uptake. Inhibition of AMPK blocked the increases of Vav3 knock down-induced glucose uptake. Metformin-mediated Glut4 translocation was also increased by Vav3 knock-down, suggesting that Vav3 is involved in metformin-mediated glucose uptake.
-
Conclusion
- These results demonstrate that Vav3 is involved in the process of metformin-mediated glucose regulation.
- Glucose, also known as d-glucose, is a simple monosaccharide and is one of the three dietary monosaccharides, along with fructose and galactose. These monosaccharides are absorbed directly into the bloodstream during digestion [1]. High blood glucose, or hyperglycemia, is a major cause of diabetes-related complications. High blood glucose occurs when the body does not produce enough insulin or when the body cannot use insulin properly. Despite its significance in the pathogenesis of metabolic diseases, such as obesity and diabetes, the molecular target of high glucose has not yet been clarified.
- Metformin is an oral antidiabetic drug of the biguanide class. Metformin originates from the French lilac (Galega officinalis), a plant known to relieve the symptoms of diabetes mellitus [2,3,4,5]. Metformin is known to have several biological activities, such as increased insulin sensitivity [6,7], decreased hepatic glucose production [8], increased peripheral glucose utilization [9], and stimulation of insulin receptor expression and tyrosine kinase activity [10,11,12]. The exact molecular mechanisms of metformin on metabolism; however, remain unclear.
- AMP-activated protein kinase (AMPK) is comprised of α, β, and γ subunits and functions as an intracellular metabolic energy sensor. It plays a critical role in the regulation of glucose and lipid metabolism and is activated when the level of cellular ATP is depleted [13]. AMPK triggers the signal which activates ATP-generating pathways, including glycolysis [14,15,16], and also switches off the signal which stimulates ATP-consuming pathways, including fatty acid synthesis [17]. The AMPK agonist 5-aminoimidazole-4-carboxy-amide-1-d-ribofuranoside (AICAR) was known to enhance insulin-mediated glucose transport in vitro [18,19] and also leads to improved insulin activity on metabolic tissues, such as muscle and liver, in rats with insulin-resistance [20]. These facts suggested that AMPK regulates the insulin-mediated signaling pathway, and thus, AMPK has become an important molecular target for the development of drugs for treating diabetes.
- The Rho family of GTPase is a family of small signaling G proteins. Three members of the family, Rac1, Cdc42, and RhoA, have been shown to regulate many kinds of cellular events, including cytoskeletal rearrangement [21]. Among them, Rac1 is dominantly expressed in mouse skeletal muscle [22] and participates in insulin-dependent glucose transporter type 4 (GLUT4) translocation [23]. Expression of dominant negative-Rac1 and knock down of Rac1 abolished insulin-stimulated GLUT4 translocation [24,25]. Overexpression of constitutively active (CA)-Rac1; however, increased the amount of surface GLUT4. These facts indicated that Rac1 plays a critical role in the insulin-dependent GLUT4 translocation process. Despite the physiological importance of GLUT4 translocation in skeletal muscle, the mechanism for GLUT4 translocation in response to insulin remains obscure at the molecular level.
- The Vav family proteins, hematopoiesis-specific signaling proteins, are cytoplasmic guanine nucleotide exchange factors (GEFs) for the Rho-family GTPases. These proteins are multidomain signaling proteins that act as adaptor proteins. The expression of Vav1 is dominant in hematopoietic cells, and Vav2 and Vav3 are ubiquitously expressed. It is not currently known if Vav proteins are associated with the dysfunction of metabolism, nor is it known whether all Vav family proteins possess similar functional activities [26]. The role of Vav proteins in muscle is not clearly defined.
- In the current study, the effects of high glucose on Vav3 expression in skeletal muscle C2C12 cells were investigated. It was shown that high glucose culture up-regulated Vav3 through AMPK, and it was further demonstrated that Vav3 was involved in metformin-mediated glucose uptake. These findings provide novel insight into the way in which AMPK contributes to glucose uptake in skeletal muscle C2C12 cells via the Vav3 pathway.
INTRODUCTION
- Reagents
- Anti-phospho-PAK and anti-phospho-paxillin antibodies were purchased from Millipore (Billerica, MA, USA). Anti-phospho-AMPK and anti-AMPK, and anti-PAK antibodies were purchased from Abcam (Cambridge, UK, USA). Anti-Vav3 and anti-β-actin antibodies were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA). Metformin and AICAR were obtained from Calbiochem (San Diego, CA, USA).
- Cell cultures and high glucose culture
- Mouse skeletal muscle C2C12 cells were maintained in Dulbecco's Modified Eagle's Medium (DMEM) supplemented with 10% fetal bovine serum (FBS) and antibiotics at 37℃ in an incubator with 5% CO2. Cells were grown in a culture medium consisting of 500 µL of DMEM (GIBCO, Auckland, NZ, USA), containing 0.584 g/L of l-glutamate and 4.5 g/L of glucose, mixed with 500 mL of F-12 medium containing 0.146 g/L of l-glutamate, 1.8 g/L of glucose, 100 µg/mL of gentamicin, 2.5 g/L of sodium carbonate, and 10% heat-inactivated FBS. For glucose concentration, 5.6 mM was regarded as the control and 35 mM was regarded as the high glucose concentration group.
- Immunoblot analysis
- Cells were grown on 10-mL plates. Following the cell treatment, the medium was aspirated and the cells were washed twice in ice-cold phosphate-buffered saline (PBS) and lysed in 100 µL of lysis buffer. The samples were then briefly sonicated, heated for 5 minutes at 95℃, and centrifuged for 5 minutes. The supernatants were electrophoresed on sodium dodecyl sulfate polyacrylamide gel electrophoresis (8%) gels and transferred to polyvinylidene difluoride membranes. The blots were incubated overnight at room temperature with primary antibodies, and then washed six times in Tris-buffered saline/0.1% Tween-20 prior to 1-hour incubation with horseradish peroxidase-conjugated secondary antibodies at room temperature. The blots were then visualized via ECL (Amersham Biosciences, Buckinghamshire, UK).
- Semi-quantitative reverse transcription-polymerase chain reaction
- First strand cDNA synthesis was performed using 1 µg of total RNA isolated from C2C12 cells at 55℃ for 20 minutes using the Thermoscript II one-step reverse transcription-polymerase chain reaction (RT-PCR) Kit (Invitrogen, Paisly, UK). Amplification of cDNA was carried out in the same tube using the Gene Amp System 9700 thermocycler (Applied Biosystems, Warrington, UK). Heating to 94℃ for 5 minutes inactivated the reverse transcriptase. The following PCR conditions were used: 27 cycles of 30 seconds at 94℃, 30 seconds at 56℃, and 30 seconds at 72℃, followed by 7 minutes at 72℃. The number of PCR cycles used was optimized to ensure amplification at the exponential phase. The 10-µL samples from each RT-PCR product were removed and analyzed by agarose gel electrophoresis. Bands were stained with ethidium bromide and visualized under ultraviolet light. Band intensity quantification was determined by a gel documentation system (Gene Genius, Syngene, UK). The following primers were used: Vav3, 5'-GGCTATCCCGAACACCAATA-3' (sense) and 5'-GAGATGGCTGACTCCACTCC-3' (antisense); and β-actin, 5'-ATT TGG TCG TAT TGG GCG CCT GGT CAC C-3' (sense) and 5' GAA GAT GGT GAT GGG ATT TC-3' (antisense).
- Vav3 silencing
- C2C12 cells were seeded in 6-well plates and grown to 70% confluence for 24 hours. Transient transfections were performed using Lipofectamine 2000 (Life Technologies, Carlsbad, CA, USA) following the manufacturer's instructions. Briefly, Vav3 siRNA was purchased from Dharmacon, and nontargeted scramble siRNA was designed and synthesized (Bioneer, Daejon, Korea). Five microlitres of siRNA and 5 µL Lipofectamine 2000 were diluted with 95 µL reduced serum medium (Opti-MEM, Life Technologies) and mixed. The transfection mixture was incubated for 30 minutes at room temperature, and then added drop-wise to each well containing 800 µL of Opti-MEM (final siRNA concentration, 100 nM). Four hours after transfection, the medium was changed to fresh complete medium.
- Glucose uptake
- Glucose uptake was measured using 2-deoxy-[3H]-d-glucose. Briefly, cells were differentiated before the glucose uptake experiment. Two days after cells reached confluence, differentiation into myotubes was induced by incubation for 6 to 7 days in an α-MEM supplemented with 2% FBS, which was changed every 2 days. The medium was replaced in the morning (serum-free medium), and the cells were exposed to the drugs for 18 hours. The cells were washed twice in warm PBS before the media and drugs were placed in DMEM devoid of glucose for 20 minutes. 2-Deoxy-[3H]-d-glucose (50 nM) was added for 15 minutes at 37℃, and the reactions were terminated by washing twice in ice-cold PBS. Cells were lysed in 10% SDS or 50 mM NaOH, and samples were transferred to scintillation vials with scintillant and allowed to sit at room temperature for 1 hour before being counted.
- Immunodetection of GLUT4myc
- Cell surface expression of GLUT4myc was quantified using an antibody-coupled colorimetric absorbance assay. Following stimulation by metformin, myoblasts stably expressing L6-GLUT4myc were incubated with polyclonal anti-myc antibody (1:1,000) for 60 minutes, fixed with 4% paraformaldehyde in PBS for 10 minutes, and incubated with horseradish-peroxidase (HRP)-conjugated goat anti-rabbit IgG (1:1,000) for 1 hour. Cells were washed six times in PBS, and incubated in 1 mL of o-phenylenediamine reagent (0.4 mg/mL) for 30 minutes. The absorbance of the supernatant was measured at 492 nm.
- Data analysis
- Values are expressed as mean±standard error of the mean. Statistical analyses were conducted using SigmaStat (SPSS Inc., Chicago, IL, USA). P values <0.05 were considered statistically significant.
METHODS
- High glucose concentration induces Vav3 levels in C2C12 cells
- To gain insight into the role of Vav3 in high glucose-mediated signaling, the effect of high glucose concentration on the expression of Vav3 was evaluated. The administration of high glucose concentrations induced a time-dependent increase in mRNA Vav3 expression in C2C12 cells (Fig. 1A). To confirm its effect on protein levels, Western blot analysis was performed. The expression of Vav3 was also increased by high glucose exposure (Fig. 1B). These results demonstrate that Vav3 is induced under high glucose culture conditions.
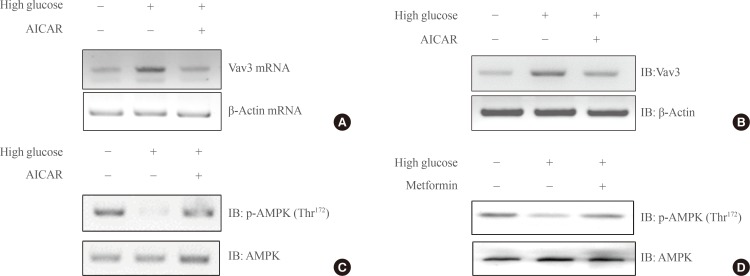
- AICAR inhibits high glucose-mediated Vav3 induction in C2C12 cells
- AICAR is known to activate AMPK. To test the hypothesis that Vav3 is regulated by AMPK, the effect of AICAR on the expression of Vav3 was investigated. The administration of AICAR blocked the high glucose-induced increase of Vav3 mRNA expression in C2C12 cells (Fig. 2A). Consistent with the decrease in mRNA level, the protein level of Vav3 also decreased after AICAR administration (Fig. 2B). Furthermore, the phosphorylation of AMPK was decreased under high glucose culture conditions. This down regulation of AMPK phosphorylation was recovered both by addition of the AMPK activator, AICAR and with treatment with the diabetes drug, metformin (Fig. 2C, D). Together, these results suggest that AMPK plays an important role in high glucose-induced Vav3 induction.
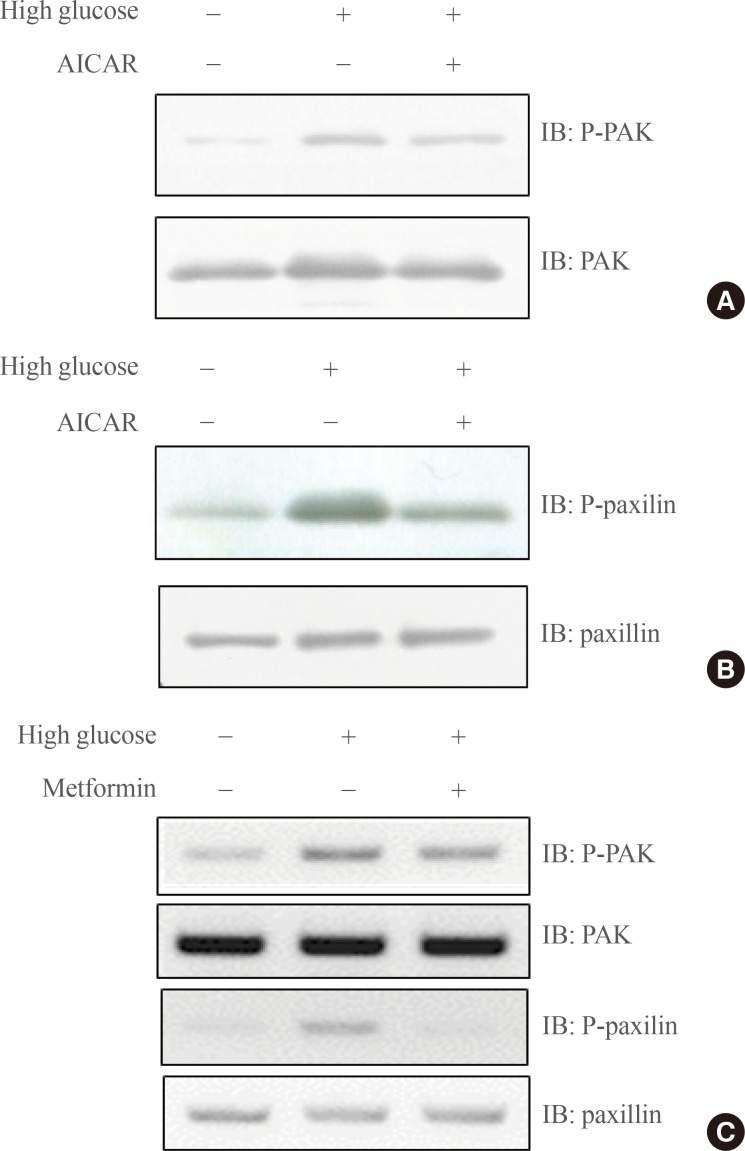
- AICAR suppresses high-glucose mediated RhoA-related signaling
- Vav3 is a GEF for RhoA, and PAK is a direct downstream molecule of RhoA. To test whether AMPK blocks high-glucose induced PAK signaling, we examined the effect of a high glucose concentration on PAK phosphorylation under AICAR. Increased phosphorylation of PAK induced by high glucose was down-regulated by the treatment of AICAR (Fig. 3A). The phosphorylation of paxillin, a downstream molecule of PAK1, was also down-regulated by AICAR (Fig. 3B). To gain insight into the physiological relevance of PAK signaling, we investigated the effect of metformin, which acts as both a diabetes drug and an AMPK activator. Increased phosphorylation of PAK and paxillin by high glucose culture was observed following metformin treatment (Fig. 3C), suggesting that PAK signaling under high glucose may be a critical molecular target for metformin treatment. These results suggest that AMPK is involved in high-glucose mediated RhoA downstream signaling.
- Knock down of Vav3 enhances metformin-mediated glucose uptake
- Vav3 was induced under a high-glucose culture condition, implying that Vav3 may be associated with high glucose-related pathophysiological conditions, such as diabetes. To gain insight into the role of Vav3 in glucose uptake, we investigated the effect of Vav3 knock down on metformin-mediated glucose uptake. First, we confirmed that the level of Vav3 was down-regulated by transient transfection with siRNA Vav3 (Fig. 4A). The increase of glucose uptake by metformin was enhanced by Vav3 knock down (Fig. 4B). This increase was not observed in the presence of the AMPK inhibitor, compound C, suggesting that AMPK plays an important role in Vav3-mediated glucose uptake regulation. HRP-conjugated secondary antibodies, in conjunction with a colorimetric assay, were used to measure cell surface localization of GLUT4myc. A further increase in plasma membrane GLUT4myc was observed in cells transfected with Vav3 siRNA (Fig. 4C). These results indicate that Vav3 may be involved in metformin-mediated glucose uptake via the translocation of GLUT4 from the cytosol to the plasma membrane.
RESULTS
- The key findings of this study were that Vav3 was induced under high-glucose culture conditions and was involved in metformin-mediated glucose uptake in skeletal muscle C2C12 cells. It was demonstrated that AMPK was instrumental in metformin-mediated signaling in these cells. The primary assertion based on this study is that AMPK mediates the activity of Vav3. These results indicate that Vav3 may play a crucial role in metformin-mediated glucose uptake through AMPK.
- Small GTPases of the Rho family control cell growth, morphogenesis, cell motility, cytokinesis, trafficking, and organization of the cytoskeleton, but they are also involved in transformation and metastasis. Vav1 is activated by two common signals generated by multiple classes of plasma membrane receptors: tyrosine phosphorylation and the phosphatidylinositol (PI) 3'-kinaseproduct, PI-3, 4, 5-P3 [27,28]. The stimulation of diverse cell surface receptors, including immune response receptors, integrins, and growth factor receptors, leads to tyrosine phosphorylation of Vav1 [29,30,31,32]. Thus, Vav proteins may function to transduce signals from diverse receptors to Rho GTPases. Vav1 was first identified by the isolation of a truncated, CA form of this protein (lacking 67 amino acids at its amino terminus) that induced oncogenic transformation of NIH 3T3 cells [33]. However, the endogenous Vav1 protein is expressed exclusively in hematopoietic cells [34,35]. Vav1 plays an important role in lymphocyte development and antigen receptor-mediated signal transduction in mice. Recently, Vav2 has been identified and is ubiquitously expressed in embryos and adult tissues [36,37].
- In this report, we showed that Vav3 was up-regulated under high glucose culture conditions. We also demonstrated that AMPK served as an upstream signal for Vav3 induction. These data revealed a novel role for AMPK as an upstream molecule of Vav3 induction involving metformin-mediated signaling. Vav3 is a Rho-specific GEF that is known to activate pathways leading to actin cytoskeletal rearrangements and transcriptional alterations. Our results showed that the phosphorylation of PAK1 was also increased by high-glucose culture concentration and that the knock down of Vav3 potentiated metformin-mediated glucose uptake, suggesting that Vav3 may become a molecular target for blood glucose regulation.
- In conclusion, we determined that a high-glucose culture condition impedes glucose uptake through Vav3 induction in skeletal muscle C2C12 cells and modulates glucose uptake. Future studies should focus on elucidating the relationship of AMPK to Vav3 within the context of a metformin-mediated glucose uptake process.
DISCUSSION
-
Acknowledgements
- This study was supported by the National Research Foundation of Korea funded by Korea government (NRF-2013R1A2A2A 05004796).
ACKNOWLEDGMENTS
- 1. Siegel Gj, Agranoff BW, Albers RW, Fisher SK, Uhler MD. Chapter 31, Circulation and energy metabolism of the brain. Basic Neurochemistry; 6th ed. Philadelphia: Lippincott-Raven; 1999. p. 637–670.
- 2. Despres JP. Potential contribution of metformin to the management of cardiovascular disease risk in patients with abdominal obesity, the metabolic syndrome and type 2 diabetes. Diabetes Metab 2003;29(4 Pt 2):6S53–6S61. ArticlePubMed
- 3. Bailey CJ, Day C. Avandamet: combined metformin-rosiglitazone treatment for insulin resistance in type 2 diabetes. Int J Clin Pract 2004;58:867–876. ArticlePubMed
- 4. Bailey CJ. Metformin: effects on micro and macrovascular complications in type 2 diabetes. Cardiovasc Drugs Ther 2008;22:215–224. ArticlePubMedPDF
- 5. Kooy A, de Jager J, Lehert P, Bets D, Wulffele MG, Donker AJ, Stehouwer CD. Long-term effects of metformin on metabolism and microvascular and macrovascular disease in patients with type 2 diabetes mellitus. Arch Intern Med 2009;169:616–625. ArticlePubMed
- 6. Borst SE, Snellen HG. Metformin, but not exercise training, increases insulin responsiveness in skeletal muscle of Sprague-Dawley rats. Life Sci 2001;69:1497–1507. ArticlePubMed
- 7. Teranishi T, Ohara T, Maeda K, Zenibayashi M, Kouyama K, Hirota Y, Kawamitsu H, Fujii M, Sugimura K, Kasuga M. Effects of pioglitazone and metformin on intracellular lipid content in liver and skeletal muscle of individuals with type 2 diabetes mellitus. Metabolism 2007;56:1418–1424. ArticlePubMed
- 8. He L, Sabet A, Djedjos S, Miller R, Sun X, Hussain MA, Radovick S, Wondisford FE. Metformin and insulin suppress hepatic gluconeogenesis through phosphorylation of CREB binding protein. Cell 2009;137:635–646. ArticlePubMedPMC
- 9. Yoshida T, Okuno A, Tanaka J, Takahashi K, Nakashima R, Kanda S, Ogawa J, Hagisawa Y, Fujiwara T. Metformin primarily decreases plasma glucose not by gluconeogenesis suppression but by activating glucose utilization in a non-obese type 2 diabetes Goto-Kakizaki rats. Eur J Pharmacol 2009;623:141–147. ArticlePubMed
- 10. Wiernsperger NF. Membrane physiology as a basis for the cellular effects of metformin in insulin resistance and diabetes. Diabetes Metab 1999;25:110–127. PubMed
- 11. Pryor PR, Liu SC, Clark AE, Yang J, Holman GD, Tosh D. Chronic insulin effects on insulin signalling and GLUT4 endocytosis are reversed by metformin. Biochem J 2000;348(Pt 1):83–91. ArticlePubMedPMCPDF
- 12. Wang Y, Li XJ, Zhang M, Wu YH, Tong NW, Zhao TY, Li J. The effect of metformin on insulin receptor protein tyrosine kinase activity of HIT-T15 cell exposed to high concentration glucose and free fatty acid. Sichuan Da Xue Xue Bao Yi Xue Ban 2007;38:819–821. PubMed
- 13. Hardie DG, Carling D. The AMP-activated protein kinase: fuel gauge of the mammalian cell? Eur J Biochem 1997;246:259–273. ArticlePubMed
- 14. Makinde AO, Gamble J, Lopaschuk GD. Upregulation of 5'-AMP-activated protein kinase is responsible for the increase in myocardial fatty acid oxidation rates following birth in the newborn rabbit. Circ Res 1997;80:482–489. ArticlePubMed
- 15. Ai H, Ihlemann J, Hellsten Y, Lauritzen HP, Hardie DG, Galbo H, Ploug T. Effect of fiber type and nutritional state on AICAR- and contraction-stimulated glucose transport in rat muscle. Am J Physiol Endocrinol Metab 2002;282:E1291–E1300. ArticlePubMed
- 16. Zong H, Ren JM, Young LH, Pypaert M, Mu J, Birnbaum MJ, Shulman GI. AMP kinase is required for mitochondrial biogenesis in skeletal muscle in response to chronic energy deprivation. Proc Natl Acad Sci U S A 2002;99:15983–15987. ArticlePubMedPMC
- 17. Henin N, Vincent MF, Gruber HE, Van den Berghe G. Inhibition of fatty acid and cholesterol synthesis by stimulation of AMP-activated protein kinase. FASEB J 1995;9:541–546. ArticlePubMed
- 18. Fisher JS, Gao J, Han DH, Holloszy JO, Nolte LA. Activation of AMP kinase enhances sensitivity of muscle glucose transport to insulin. Am J Physiol Endocrinol Metab 2002;282:E18–E23. ArticlePubMed
- 19. Ju JS, Gitcho MA, Casmaer CA, Patil PB, Han DG, Spencer SA, Fisher JS. Potentiation of insulin-stimulated glucose transport by the AMP-activated protein kinase. Am J Physiol Cell Physiol 2007;292:C564–C572. ArticlePubMed
- 20. Iglesias MA, Ye JM, Frangioudakis G, Saha AK, Tomas E, Ruderman NB, Cooney GJ, Kraegen EW. AICAR administration causes an apparent enhancement of muscle and liver insulin action in insulin-resistant high-fat-fed rats. Diabetes 2002;51:2886–2894. ArticlePubMed
- 21. Burridge K, Wennerberg K. Rho and Rac take center stage. Cell 2004;116:167–179. ArticlePubMed
- 22. Ueda S, Kitazawa S, Ishida K, Nishikawa Y, Matsui M, Matsumoto H, Aoki T, Nozaki S, Takeda T, Tamori Y, Aiba A, Kahn CR, Kataoka T, Satoh T. Crucial role of the small GTPase Rac1 in insulin-stimulated translocation of glucose transporter 4 to the mouse skeletal muscle sarcolemma. FASEB J 2010;24:2254–2261. ArticlePubMedPMC
- 23. JeBailey L, Rudich A, Huang X, Di Ciano-Oliveira C, Kapus A, Klip A. Skeletal muscle cells and adipocytes differ in their reliance on TC10 and Rac for insulin-induced actin remodeling. Mol Endocrinol 2004;18:359–372. ArticlePubMedPDF
- 24. JeBailey L, Wanono O, Niu W, Roessler J, Rudich A, Klip A. Ceramide- and oxidant-induced insulin resistance involve loss of insulin-dependent Rac-activation and actin remodeling in muscle cells. Diabetes 2007;56:394–403. ArticlePubMed
- 25. Ueda S, Kataoka T, Satoh T. Activation of the small GTPase Rac1 by a specific guanine-nucleotide-exchange factor suffices to induce glucose uptake into skeletal-muscle cells. Biol Cell 2008;100:645–657. ArticlePubMed
- 26. Moores SL, Selfors LM, Fredericks J, Breit T, Fujikawa K, Alt FW, Brugge JS, Swat W. Vav family proteins couple to diverse cell surface receptors. Mol Cell Biol 2000;20:6364–6373. ArticlePubMedPMC
- 27. Bustelo XR. Regulatory and signaling properties of the Vav family. Mol Cell Biol 2000;20:1461–1477. ArticlePubMedPMC
- 28. Han J, Luby-Phelps K, Das B, Shu X, Xia Y, Mosteller RD, Krishna UM, Falck JR, White MA, Broek D. Role of substrates and products of PI 3-kinase in regulating activation of Rac-related guanosine triphosphatases by Vav. Science 1998;279:558–560. ArticlePubMed
- 29. Cichowski K, Brugge JS, Brass LF. Thrombin receptor activation and integrin engagement stimulate tyrosine phosphorylation of the proto-oncogene product, p95vav, in platelets. J Biol Chem 1996;271:7544–7550. ArticlePubMed
- 30. Gotoh A, Takahira H, Geahlen RL, Broxmeyer HE. Cross-linking of integrins induces tyrosine phosphorylation of the proto-oncogene product Vav and the protein tyrosine kinase Syk in human factor-dependent myeloid cells. Cell Growth Differ 1997;8:721–729. PubMed
- 31. Miranti CK, Leng L, Maschberger P, Brugge JS, Shattil SJ. Identification of a novel integrin signaling pathway involving the kinase Syk and the guanine nucleotide exchange factor Vav1. Curr Biol 1998;8:1289–1299. ArticlePubMed
- 32. Zheng L, Sjolander A, Eckerdal J, Andersson T. Antibody-induced engagement of beta 2 integrins on adherent human neutrophils triggers activation of p21ras through tyrosine phosphorylation of the protooncogene product Vav. Proc Natl Acad Sci U S A 1996;93:8431–8436. ArticlePubMedPMC
- 33. Katzav S, Martin-Zanca D, Barbacid M. vav, a novel human oncogene derived from a locus ubiquitously expressed in hematopoietic cells. EMBO J 1989;8:2283–2290. ArticlePubMedPMC
- 34. Adams JM, Houston H, Allen J, Lints T, Harvey R. The hematopoietically expressed vav proto-oncogene shares homology with the dbl GDP-GTP exchange factor, the bcr gene and a yeast gene (CDC24) involved in cytoskeletal organization. Oncogene 1992;7:611–618. PubMed
- 35. Katzav S, Cleveland JL, Heslop HE, Pulido D. Loss of the amino-terminal helix-loop-helix domain of the vav proto-oncogene activates its transforming potential. Mol Cell Biol 1991;11:1912–1920. ArticlePubMedPMC
- 36. Henske EP, Short MP, Jozwiak S, Bovey CM, Ramlakhan S, Haines JL, Kwiatkowski DJ. Identification of VAV2 on 9q34 and its exclusion as the tuberous sclerosis gene TSC1. Ann Hum Genet 1995;59(Pt 1):25–37. ArticlePubMed
- 37. Schuebel KE, Bustelo XR, Nielsen DA, Song BJ, Barbacid M, Goldman D, Lee IJ. Isolation and characterization of murine vav2, a member of the vav family of proto-oncogenes. Oncogene 1996;13:363–371. PubMed
References
Fig. 1(A) Total RNA was prepared for these cells after high glucose culture, and reverse transcription-polymerase chain reaction (RT-PCR) was conducted using specific Vav3 primers. PCR products were then run on 1% agarose gels and visualized under ultraviolet light. β-Actin mRNA was employed as a positive control. (B) C2C12 cells were stimulated at the indicated times with high glucose concentrations. The cell lysates (25 µg) were analyzed via Western blotting for anti-Vav3 antibody. Blotting with anti-β-actin antibody was used as a protein loading control. The results shown are from three independent experiments.
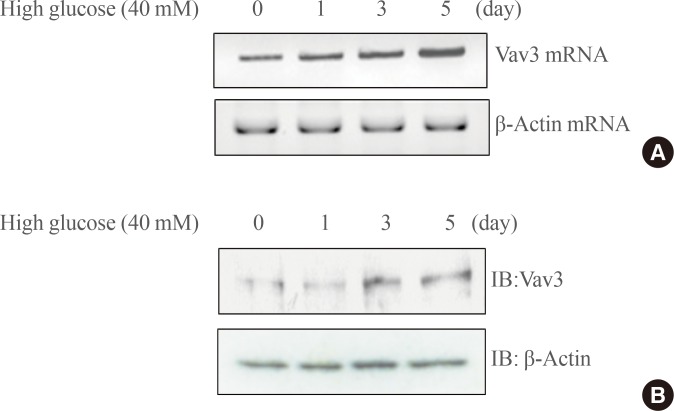

Fig. 2(A) Total RNA was cultured under high glucose culture conditions in the presence or absence of 5-aminoimidazole-4-carboxy-amide-1-d-ribofuranoside (AICAR) treatment, and reverse transcription-polymerase chain reaction (RT-PCR) was conducted using specific Vav3 primers. PCR products were then run on 1% agarose gels and visualized under ultraviolet light. β-Actin mRNA was employed as a positive control. (B) C2C12 cells were cultured under high glucose conditions in the presence or absence of AMP-activated protein kinase (AMPK) agonist 5-aminoimidazole-4-carboxy-amide-1-d-ribofuranoside (AICAR). The cell lysates (25 µg) were analyzed via Western blotting for anti-Vav3 antibody. Blotting with anti-β-actin antibody was conducted as a protein loading control. (C) C2C12 cells were cultured under high glucose conditions in the presence or absence of AICAR. The cell lysates (25 µg) were analyzed via Western blotting for anti-phospho-AMPK antibody. Blotting with anti-AMPK antibody was conducted as a protein loading control. (D) C2C12 cells were cultured under high glucose conditions in the presence or absence of metformin. The cell lysates (25 µg) were analyzed via Western blotting for anti-phospho-AMPKantibody. Blotting with anti-AMPK antibody was conducted as a protein loading control. The results shown are from three independent experiments.
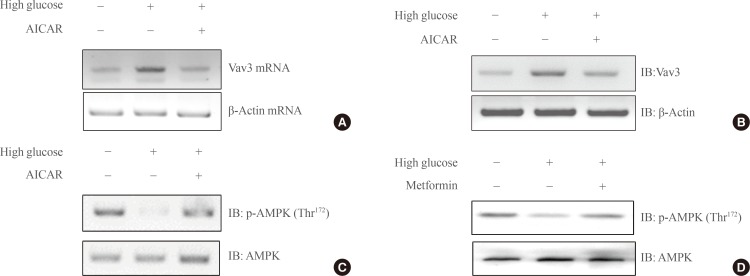

Fig. 3(A) C2C12 cells were cultured under high glucose conditions in the presence or absence of AMPK agonist 5-aminoimidazole-4-carboxy-amide-1-d-ribofuranoside (AICAR). Total cell lysates (25 µg) were analyzed via Western blotting for anti-phospho-PAK and anti-PAK antibodies. (B) C2C12 cells were cultured under high glucose conditions in the presence or absence of AICAR. The cell lysates (25 µg) were analyzed via Western blotting. Blotting with anti-phospho-paxillin and anti-paxillin antibodies was conducted as a protein loading control. (C) C2C12 cells were cultured under high glucose conditions in the presence of metformin. The cell lysates (25 µg) were analyzed via Western blotting. Blotting with anti-phospho-PAK and anti-phospho-paxillin antibodies was performed. Blotting with anti-PAK and anti-paxillin antibodies was conducted as a protein loading control. The results shown are from three independent experiments.


Fig. 4(A) C2C12 cells were transiently transfected with 50 nM siRNA Vav3 for 2 days. Total cell lysates (25 µg) were analyzed via Western blotting for anti-Vav3 and anti-β-actin antibodies. (B) C2C12 cells were allowed to differentiate for 7 days. Cells were transiently transfected with Vav3 siRNA for 48 hours and then treated with metformin for 16 hours either in the presence or absence of compound C. Glucose uptake was measured using 2-deoxy-[3H]-d-glucose. The results shown are from three independent experiments. (C) Myoblasts stably expressing L6-GLUT4myc were differentiated and were transiently transfected with Vav3 siRNA and maintained for 48 hours, then incubated with metformin for 1 hour. Cell surface expression of GLUT4myc was detected using an antibody-coupled colorimetric absorbance assay. Results are displayed as the mean±SEM from three experiments. aP<0.05 for the basal values; bP<0.05 for the metformin-treated condition; cP<0.05 for the metformin plus Vav3 siRNA; dP<0.05 compared with metformin-treated condition.
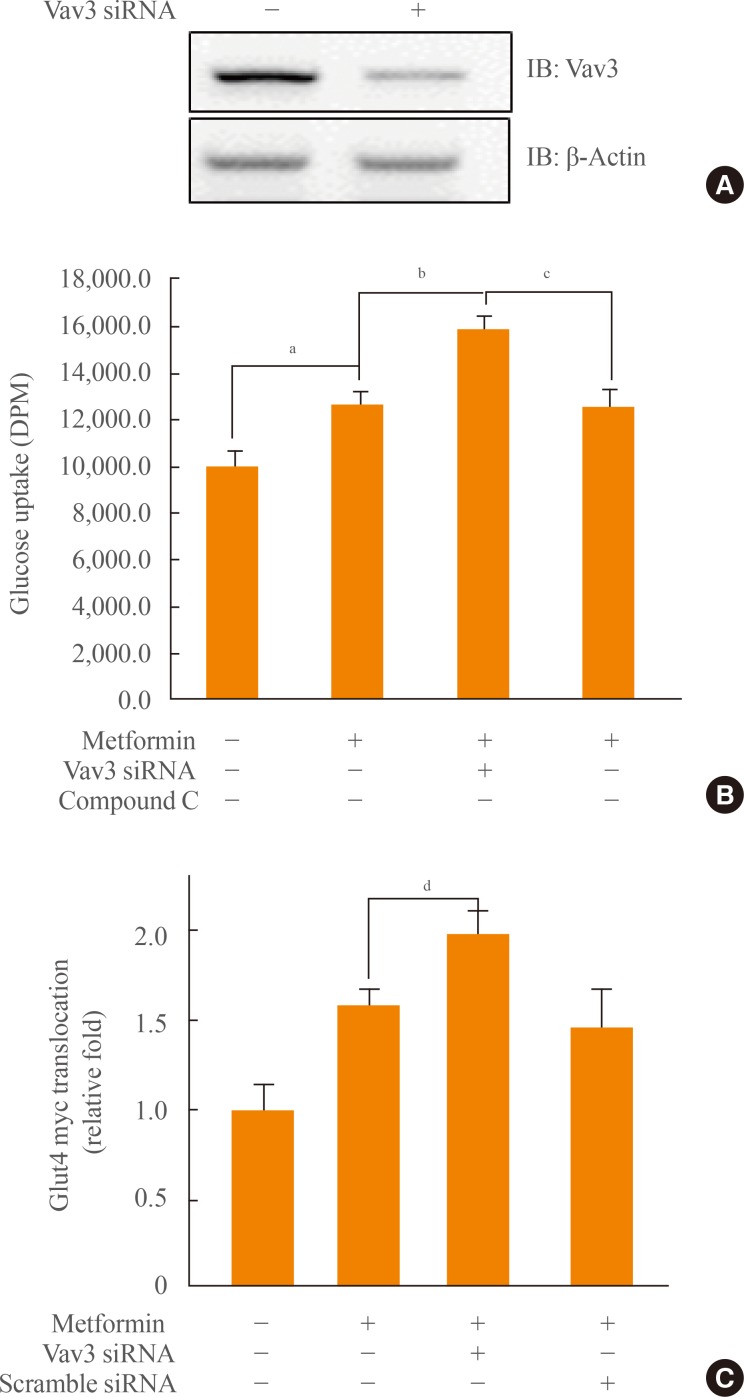

Figure & Data
References
Citations
Citations to this article as recorded by 

- Peripheral origin exosomal microRNAs aggravate glymphatic system dysfunction in diabetic cognitive impairment
Lin Zhang, Dongna Li, Pengrong Yi, Jiangwei Shi, Mengqing Guo, Qingsheng Yin, Dingbin Liu, Pengwei Zhuang, Yanjun Zhang
Acta Pharmaceutica Sinica B.2023; 13(7): 2817. CrossRef - A current overview of RhoA, RhoB, and RhoC functions in vascular biology and pathology
Robert Eckenstaler, Michael Hauke, Ralf A. Benndorf
Biochemical Pharmacology.2022; 206: 115321. CrossRef - Rho Family GTPases and Rho GEFs in Glucose Homeostasis
Polly A. Machin, Elpida Tsonou, David C. Hornigold, Heidi C. E. Welch
Cells.2021; 10(4): 915. CrossRef - Pharmacological Modulators of Small GTPases of Rho Family in Neurodegenerative Diseases
William Guiler, Addison Koehler, Christi Boykin, Qun Lu
Frontiers in Cellular Neuroscience.2021;[Epub] CrossRef - Association of VAV2 and VAV3 polymorphisms with cardiovascular risk factors
Nuria Perretta-Tejedor, Javier Fernández-Mateos, Luis García-Ortiz, Manuel A. Gómez-Marcos, José I. Recio-Rodríguez, Cristina Agudo-Conde, Emiliano Rodriguez-Sánchez, Ana I. Morales, Francisco J. López-Hernández, José M. López-Novoa, Rogelio González-Sarm
Scientific Reports.2017;[Epub] CrossRef - Articles in 'Endocrinology and Metabolism' in 2014
Won-Young Lee
Endocrinology and Metabolism.2015; 30(1): 47. CrossRef

 KES
KES
 PubReader
PubReader Cite
Cite